Abstract
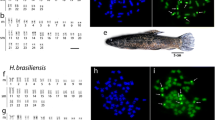
Astyanax has been the subject of extensive cytogenetic studies due to its wide karyotypic diversity. This genus comprises species complexes, namely groups of fish of difficult morphological differentiation, such as the bimaculatus complex, which includes the characids with a rounded humeral spot. Thence, the present study proposed to accomplish a cytogenetic characterization of two species of this complex: A. asuncionensis and A. altiparanae, aiming to find chromosomal markers that differentiate these species, as well as achieve a better understanding of the karyotype evolution in the genus. For this we used different techniques of chromosome banding as C-banding, impregnation by silver nitrate, fluorochrome staining and FISH with 18S rDNA probe. This is the first cytogenetic study in A. asuncionensis, from Miranda river, which presented 2n = 50 and 18 m + 22sm + 6st + 4a (FN = 96) and single NORs. The populations of A. altiparanae also presented 2n = 50, but with different karyotypic formulae: the population of the Quexada river presented 16 m + 24sm + 4st + 6a (FN = 94) and the Esperança stream and Jacutinga river showed 16 m + 20sm + 4st + 10a (FN = 90). All analyzed populations showed an interindividual variation in the number and location of the nucleolar organizer regions (NORs). Single and multiple NORs were detected either by impregnation with silver nitrate or by FISH with 18S rDNA probe. After C-banding, the two species differed in relation to the composition and heterochromatin distribution. The meiotic cells of A. altiparanae male individuals were also analyzed, showing that, despite the high karyotype variability, chromosome pairing occurs normally. The data show that A. altiparanae and A. asuncionensis share some characteristics with other species of the bimaculatus complex, suggesting a close phylogenetic relationship among those species. However, some features can be used as differentiation chromosomal markers in altiparanae/asuncionensis morphotypes, which could result from a natural speciation process.






Similar content being viewed by others
References
Almeida-Toledo LF, Ozouf-Costaz C, Foresti F, Bonilo C, Porto-Foresti F, Daniel-Silva MFZ (2002) Conservation of the 5S-bearing chromosome pair and co-localization with major rDNA clusters in five species of Astyanax (Pisces, Characidae). Cytogen Genome Res 97:229–233
Bertaco VA, Garutti V (2007) New Astyanax from the upper rio Tapajós drainage, Central Brazil (Characiformes, Characidae). Neotrop Ichthyol 5:25–30
Bertollo LAC, Takahashi CS, Moreira-Filho O (1978) Cytotaxonomic considerations on Hoplias lacerdae (Pisces, Erythrinidae). Brazil J Gen 1:103–120
Domingues MS, Vicari MV, Abilhoa V, Wamser JP, Cestari MM, Bertollo LAC, Almeida MC, Artoni RF (2007) Cytogenetic and comparative morphology of two allopatric populations of Astyanax altiparanae Garutti & Britski, 2000 (Teleostei: characidae) from upper rio Paraná basin. Neotrop Ichthyol 5:37–44
Eschmeyer WN (2014) Catalog of fishes. http://research.calacademy.org. Accessed 12 Sept 2014
Fenocchio AS, Venere PC, Cesar ACG, Dias AL, Bertollo LAC (1991) Short term culture from solid tissues of fishes. Caryologia 44:161–166
Fernandes CA, Martins-Santos IC (2004) Cytogenetic studies in two populations of Astyanax altiparanae (Pisces, Characiformes). Hereditas 141:328–332
Fernandes CA, Martins-Santos IC (2006a) Mapping of the 18S and 5S ribosomal RNA genes in Astyanax altiparanae Garutti & Britski, 2000 (Teleostei, Characidae) from the upper Paraná river basin, Brazil. Gen Mol Biol 29:464–468
Fernandes CA, Martins-Santos IC (2006b) Chromosomal location of 5S and 18S rRNA genes in three sympatric cytotypes of Astyanax scabripinnis (Characiformes, Characidae) from the Ivaí river basin, state of Paraná, Brazil. Caryologia 59:253–259
Ferreira Neto M, Vicari MR, Camargo EF, Artoni RF, Moreira-Filho O (2009) Comparative cytogenetics among populations of Astyanax altiparanae (Characiformes, Characidae, Incertae sedis). Gen Mol Biol 32:792–796
Garutti V (1995) Revisão taxonômica dos Astyanax (Pisces, Characidae) com mancha umeral ovalada e mancha no pedúnculo caudal, estendendo-se à extremidade dos raios caudais medianos, das bacias do Paraná. Thesis, Universidade Estadual Paulista, São Francisco e Amazônica
Garutti V, Britski HÁ (2000) Descrição de uma espécie nova de Astyanax (Teleostei: Characidae) da bacia do alto Rio Paraná e considerações sobre as demais espécies do gênero na bacia. Comunicações do Museu de Ciências e Tecnologia da PUCRS: Série Zoologia 13:65–88
Hashimoto DT, Gonçalves VR, Bortolozzi J, Foresti F, Porto-Foresti F (2008) First report of a B chromosome in a natural population of Astyanax altiparanae (Characiformes, Characidae). Gen Mol Biol 31:275–278
Hatanaka T, Galetti PM Jr (2004) Mapping of the 18S and 5S ribosomal RNA genes in the fish Prochilodus argenteus, Agassiz, 1829 (Characiformes, Prochilodontidae). Genetica 122:239–244
Howell WM, Black DA (1980) Controlled silver staining of nucleolus organizer regions with a protective colloidal developer: a 1-step method. Experientia 36:1014–1015
Kantek DLZ, Noleto RB, Fenochio AS, Cestari MM (2007) Cytotaxonomy, heterochromatic polymorphism and natural triploidy of a species of Astyanax (Pisces, Characidae) endemic to the Iguaçu river basin. Brazil Arch Biol Technol 50:67–74
Kavalco KF, Moreira-Filho O (2003) Cytogenetical analyses in four species of the genus Astyanax (Pisces, Characidae) from Paraíba do Sul River Basin. Caryologia 56:453–461
Kavalco KF, Pazza R, Brandão KO, Garcia C, Almeida-Toledo LF (2011) Comparative Cytogenetics and Molecular Phylogeography in the Group Astyanax altiparanae—Astyanax aff. bimaculatus (Teleostei, Characidae). Cytogenet Genome Res 134:108–119
Kligerman AD, Bloom SE (1977) Rapid chromosome preparations from solide tissues of fishes. J Fish Res Board Can 34:266–269
Levan A, Fredga K, Sandberg AA (1964) Nomenclature for centromeric position on chromosomes. Hereditas 52:201–220
Lima FCT, Malabarba LR, Buckup PA, Silva JFP, Vari RP, Harold A, Benine R, Oyakawa OT, Pavanelli CS, Menezes NA, Lucena CAS, Malabarba MCSL, Lucena ZMS, Reis RE, Langeani F, Cassati L, Bertaco VA, Moreira C, Lucinda PHF (2003) Genera Incertae sedis in Characidae. In: Reis RE, Kullander SO, Ferraris Jr CJ (eds) Check list of the Freshwater Fishes of South and Central America, EDIPUCRS, Porto Alegre, pp 106–113
Mantovani M, Abel LDS, Moreira-Filho O (2005) Conserved 5S and variable 45S DNAr chromosomal localization revealed by FISH in Astyanax scabripinnis (Pisces, Characidae). Genetica 123:211–216
Mendes MM, Rosa R, Giuliano-Caetano L, Dias AL (2011) Karyotype diversity of four species of the Incertae sedis group (Characidae) from different hydrographic basins: analysis of AgNORs, CMA3 and 18S rDNA. Gen Mol Res 10:3596–3608
Pacheco RB, Giuliano-Caetano L, Julio HF Jr, Dias AL (2010) Cytogenetic data on Astyanax jacuhiensis (Characidae) in the lago Guaíba and tributaries, Brazil. Neotrop Ichthyol 8:667–671
Pacheco RB, Rosa R, Giuliano-Caetano L, Júlio HF Jr, Dias AL (2011) Cytogenetic comparison between two allopatric different populations of Astyanax altiparanae Garutti et Britski, 2000 (Teleostei, Characidae), with emphasis on the localization of 18S and 5S rDNA. Comparative Cytogenet 5:237–246
Paiz LM, Baumgärtner L, Graça WJ, Margarido VP (2015) Basic cytogenetics and physical mapping of ribosomal genes in four Astyanax species (Characiformes, Characidae) collected in Middle Paraná River, Iguassu National Park: considerations on taxonomy and systematics of the genus. Comp Cytogenetics 9:54–65
Peres WAM, Bertollo LAC, Moreira-Filho O (2008) Physical mapping of the 18S and 5S ribosomal genes in nine Characidae species (Teleostei, Characiformes). Gen Mol Biol 31:222–226
Peres WAM, Bertollo LAC, Buckup PA, Blanco DR, Kantek DLZ, Moreira-Filho O (2012) Invasion, dispersion and hybridization of fish associated to river transposition: karyotypic evidence in Astyanax ‘‘bimaculatus group’’ (Characiformes: Characidae). Rev Fish Biol Fish 22:519–526
Pinkel D, Straume T, Gray JW (1986) Cytogenetic analysis using quantitative, high-sensitivity, fluorescence hybridization. Proc Natl Acad Sci USA 83:2934–2938
Prioli AJ, Carlo VA, Soria TV, Prioli SMAP, Pavanelli CS, Prioli RA, Gomes VN, Júlio HF Jr, Prioli LM (2012) Mitochondrial D-loop nucleotide diversity in Astyanax (Osteichthyes, Characidae) from the upper Paraná and upper Paraguay River basins. Gen Mol Res 11:1064–1074
Schweizer D (1980) Simultaneous fluorescent staining of R bands and specific heterochromatic regions (DAPI bands) in human chromosomes. Cytogenet Cell Gen 27:190–193
Schweizer D, Loidl J (1987) A model for heterochromatin dispersion and the evolution of C-band pattern. In: Stahl A, Luciani JM, Vagner-Capodano AM (eds) Chromosome today. Allen & Unwin, New York, pp 61–74
Silva LLL, Giuliano-Caetano L, Dias AL (2012) Chromosome studies of Astyanax jacuhiensis Cope, 1894 (Characidae) from the Tramandai River Basin, Brazil, using in situ hybridization with 18S rDNA probe, DAPI and CMA3 staining. Folia Biol (Krákow) 60:3–4
Sumner AT (1972) A simple technique for demonstrating centromeric heterochromatin. Exp Cell Res 75:304–306
Acknowledgments
The authors are grateful to Fábio Hiroshi Takagui and Natália Bortholazzi Venturelli by collecting specimens of Astyanax asuncionensis. This research was supported by a grant from Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (CAPES). The researcher received permission from Instituto Brasileiro do Meio Ambiente e dos Recursos Naturais Renováveis (IBAMA) to collect fish specimens.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Da Silva, L.L.L., Dos Santos, A.R., Giuliano-Caetano, L. et al. Chromosomal characterization in two species of an Astyanax bimaculatus complex (Characidae, Characiformes) using different techniques of chromosome banding. Cytotechnology 68, 1277–1286 (2016). https://doi.org/10.1007/s10616-015-9888-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10616-015-9888-3