The products of the one-pot, three-component synthesis of [1,3]thiazolo-[4,3-b][1, 3, 4]thiadiazoles from aromatic aldehydes, thioglycolic acid, and compounds containing a C(=S)–N–NH2 fragment (thiosemicarbazide or 4-amino-2,4-dihydro-3H-1,2,4-triazole-3-thiones) are not condensed hetero-cycles (as reported by several researchers), but are thiosemicarbazones or triazolylimines of the aldehydes used.
Similar content being viewed by others
Notes
Here and further, for the AA'XX' system of the para-substituted phenyl ring, instead of the true 3 J AX values, the distances between maxima of the corresponding doublet-like multiplets are indicated.
References
H. Malipeddi, A. A. Karigar, V. R. Malipeddi, and M. S. Sikarwar, Trop. J. Pharm. Res., 11, 611 (2012).
M. Himaja, A. Karigar, M. V. Ramana, D. Munirajasekhar, and M. S. Sikarwar, Lett. Drug Des. Discovery, 9, 611 (2012).
A. A. Karigar, M. Himaja, S. V. Mali, K. P. Jagadeesh, and M. S. Sikarwar, Int. Res. J. Pharm., 2, 153 (2011).
M. A. Kukaniev, M. M. Akbarova, Z. G. Sangov, S. Sh. Safarov, and D. M. Osimov, Khim. Geterotsikl. Soedin., 761 (2010). [Chem. Heterocycl. Compd., 46, 605 (2010)].
A. R. Saundane, Y. Manjunatha, and P. Walmik, Heterocycl. Commun., 15, 303 (2009).
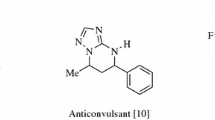
T. E. Ali, S. A. Abdel-Aziz, H. M. El-Shaaer, F. I. Hanafy, and A. Z. El-Fauomy, Phosphorus, Sulfur Silicon Relat. Elem., 183, 2139 (2008).
V. N. Yarovenko, A. S. Nikitina, I. V. Zavarzin, M. M. Krayushkin, and L. V. Kovalenko, Zh. Org. Khim., 43, 1368 (2007). [Russ. J. Org. Chem., 43, 1364 (2007)].
Nizamuddin and A. Singh, Indian J. Chem., 43B, 901 (2004).
L. D. S. Yadav and S. Singh, Indian J. Chem., 40B, 440 (2001).
L. D. S. Yadav, A. Vaish, and S. Sharma, J. Agric. Food Chem., 42, 811 (1994).
K. Singh, N. Tiwari, and Nizamuddin, Indian J. Chem., 32B, 1086 (1993).
B. El-S. Bayoumy and L. Skulski, Bull. Pol. Acad. Sci., Chem., 39, 449 (1991).
N. Tiwari and Nizamuddin, J. Indian Chem. Soc., 68, 144 (1991).
S. Sh. Shukurov, M. A. Kukaniev, and A. M. Alibaeva, Izv. Akad. Nauk, Ser. Khim., 763 (1996). [Russ. Chem. Bull., 724 (1996)].
H. Rajak, P. Parmar, B. S. Thakur, A. Agarawal, R. Veerasamy, P. C. Sharma, and M. D. Kharya, Bioorg. Med. Chem. Lett., 21, 5735 (2011).
W. Yi, S. Yahiaoui, R. Haudecoeur, A. Boumendjel, C. Dubois, R. Hardre, M. Reglier, C. Belle, and H. Song, Eur. J. Med. Chem., 46, 4330 (2011).
M. S. Alam, Y.-E. Lee, D.-U. Lee, and L. Liu, Chem. Pharm. Bull., 59, 568 (2011).
B. Puetzer, W. E. Hamlin, and L. Katz, J. Am. Chem. Soc., 73, 2958 (1951).
M. M. Akbarova, Dissertations of Candidates in Chemical Sciences, Dushanbe (2007).
S. Sh. Shukurov, M. A. Kukaniev, A. M. Alibaeva, and B. M. Bobogaribov, Khim. Geterotsikl. Soedin., 271 (1996). [Chem. Heterocycl. Compd., 32, 243 (1996)].
H. E. Gottlieb, V. Kotlyar, and A. Nudelman, J. Org. Chem., 62, 7512 (1997).
I. Guben, Methods in Organic Chemistry [Russian translation], Vol. 4, Book 1, GNTI KhimLit, Moscow-Leningrad (1949), p. 277.
H. Beyer and C. F. Kroger, Liebigs Ann. Chem., 637, 144 (1960).
J. R. Raid and N. D. Heindel, J. Heterocycl. Chem., 13, 925 (1976).
F. P. Invidiata, G. Furno, I. Lampronti, and D. Simone, J. Heterocycl. Chem., 34, 1255 (1997).
Author information
Authors and Affiliations
Corresponding author
Additional information
Translated from Khimiya Geterotsiklicheskikh Soedinenii, No. 10, pp. 1572-1577, October, 2013.
Rights and permissions
About this article
Cite this article
Kuznetsov, M.A., Bespalov, A.Y. One-pot, Three-component Synthesis of [1,3]thiazolo[4,3-b][1,3,4]thiadiazoles: Correct Structure of the Products. Chem Heterocycl Comp 49, 1458–1463 (2014). https://doi.org/10.1007/s10593-014-1396-4
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10593-014-1396-4