Abstract
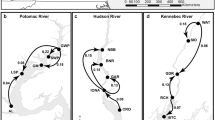
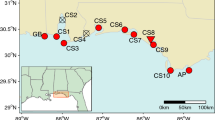
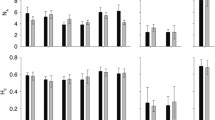
Submersed aquatic macrophyte beds provide important ecosystem services, yet their distribution and extent has declined worldwide in aquatic ecosystems. Effective restoration of these habitats will require, among other factors, reintroduction of genetically diverse source material that can withstand short- and long-term environmental fluctuations in environmental conditions. We examined patterns of genetic diversity in Vallisneria americana because it is a cosmopolitan freshwater submersed aquatic macrophyte and is commonly used for restoring freshwater habitats. We sampled 26 naturally occurring populations of V. americana in the Chesapeake Bay estuary and its tributaries and found that the majority of populations have high genotypic diversity and are not highly inbred. Fourteen of the populations had high allelic and genotypic diversity and could serve as source sites for restoration material. However, substantial geographic structuring of genetic diversity suggests that caution should be used in moving propagules to locations distant from their source. In particular, we suggest that propagules at least be limited within four primary geographic areas that correspond to freshwater tidal and non-tidal, oligohaline, and seasonally mesohaline areas of the Chesapeake Bay.




Similar content being viewed by others
References
Allendorf FW, Luikart G (2007) Conservation and the genetics of populations. Blackwell Publishing, Malden
Antonovics J (2006) Evolution in closely adjacent plant populations X: long-term persistence of prereproductive isolation at a mine boundary. Heredity 97:33–37
Antonovics J, Bradshaw AD (1970) Evolution in closely adjacent plant populations. VIII. Clinal patterns at a mine boundary. Heredity 25:349–362
Arnaud-Haond S, Belkhir K (2007) GENCLONE: a computer program to analyse genotypic data, test for clonality and describe spatial clonal organization. Mol Ecol Notes 7:15–17
Arnaud-Haond S, Duarte CM, Alberto F, Serrao EA (2007) Standardizing methods to address clonality in population studies. Mol Ecol 16:5115–5139
Arnaud-Haond S, Marba N, Diaz-Almela E, Serrao EA, Duarte CM (2010) Comparative analysis of stability-genetic diversity in seagrass (Posidonia oceanica) meadows yields unexpected results. Estuar Coast 33:878–889
Barko J, Hardin D, Matthews M (1982) Growth and morphology of submersed freshwater macrophytes in relation to light and temperature. Can J Bot 60:877–887
Barrett RDH, Schluter D (2008) Adaptation from standing genetic variation. Trends Ecol Evol 23:38–44
Becheler R, Diekmann O, Hily C, Moalic Y, Arnaud-Haond S (2010) The concept of population in clonal organisms: mosaics of temporally colonized patches are forming highly diverse meadows of Zostera marina in Brittany. Mol Ecol 19:2394–2407
Beerli P (2006) Comparison of Bayesian and maximum-likelihood inference of population genetic parameters. Bioinformatics 22:341–345
Beerli P, Felsenstein J (1999) Maximum-likelihood estimation of migration rates and effective population numbers in two populations using a coalescent approach. Genetics 152:763–773
Beerli P, Felsenstein J (2001) Maximum likelihood estimation of a migration matrix and effective population sizes in n subpopulations by using a coalescent approach. Proc Natl Acad Sci USA 98:4563–4568
Berryman AA (2002) Population: a central concept for ecology? Oikos 97:439–442
Blum MJ, Knapke E, McLachlan JS, Snider SB, Saunders CJ (2010) Hybridization between Schoenoplectus sedges across Chesapeake Bay marshes. Conserv Genet 11:1885–1898
Boesch DF, Brinsfield RB, Magnien RE (2001) Chesapeake Bay eutrophication: scientific understanding, ecosystem restoration, and challenges for agriculture. J Environ Qual 30:303–320
Boustany RG, Thomas CM, Rebecca FM (2010) Effects of salinity and light on biomass and growth of Vallisneria americana from Lower St. Johns River, FL, USA. Wetl Ecol Manag 18:203–217
Brix H, Schierup HH (1989) The use of aquatic macrophytes in water-pollution control. Ambio 18:100–107
Broadhurst LM, Lowe A, Coates DJ, Cunningham SA, McDonald M, Vesk PA, Yates C (2008) Seed supply for broadscale restoration: maximizing evolutionary potential. Evol Appl 1:587–597
Burnett RK, Lloyd MW, Engelhardt KAM, Neel MC (2009) Development of 11 polymorphic microsatellite markers in a macrophyte of conservation concern, Vallisneria americana Michaux (Hydrocharitaceae). Mol Ecol Resour 9:1427–1429
Caisse M, Antonovics J (1978) Evolution in closely adjacent plant populations IX. Evolution of reproductive isolation in clinal populations. Heredity 40:371–384
Campanella JJ, Bologna PAX, Smith SM, Rosenzweig EB, Smalley JV (2010) Zostera marina population genetics in Barnegat Bay, New Jersey, and implications for grass bed restoration. Popul Ecol 52:181–190
Catling PM, Spicer KW, Biernacki M, Doust JL (1994) The biology of Canadian weeds.103. Vallisneria americana Michx. Can J Plant Sci 74:883–897
Chao A, Jost L, Chiang SC, Jiang YH, Chazdon RL (2008) A two-stage probabilistic approach to multiple-community similarity indices. Biometrics 64:1178–1186
Chen L, Xu LM, Huang HW (2007) Genetic diversity and population structure in Vallisneria spinulosa (Hydrocharitaceae). Aquat Bot 86:46–52
Clausen J, Keck DD, Hiesey WM (1941) Regional differentiation in plant species. Am Nat 75:231–250
Cooper SR (1995) Chesapeake Bay watershed historical land-use-impact on water-quality and diatom communities. Ecol Appl 5:703–723
Cornuet JM, Luikart G (1996) Description and power analysis of two tests for detecting recent population bottlenecks from allele frequency data. Genetics 144:2001–2014
Costanza R, Greer J (1995) The Chesapeake Bay and its watershed: a model for sustainable ecosystem management? In: Gunderson LH, Holling CS, Light SS (eds) Barriers and bridges to the renewal of ecosystems and institutions. Columbia University Press, New York, pp 169–213
Crain CM, Silliman BR, Bertness SL, Bertness MD (2004) Physical and biotic drivers of plant distribution across estuarine salinity gradients. Ecology 85:2539–2549
Crawford NG (2009) SMOGD: software for the measurement of genetic diversity. Mol Ecol Resour. doi:10.1111/j.1755-0998.2009.02801.x
Dennison WC, Orth RJ, Moore KA, Stevenson JC, Carter V, Kollar S, Bergstrom PW, Batiuk RA (1993) Assessing water-quality with submersed aquatic vegetation. Bioscience 43:86–94
Di Rienzo A, Peterson AC, Garza JC, Valdes AM, Slatkin M, Freimer NB (1994) Mutational processes of simple-sequence repeat loci in human populations. Proc Natl Acad Sci USA 91:3166–3170
Doebeli M, Dieckmann U (2003) Speciation along environmental gradients. Nature 421:259–264
Doering PH, Chamberlain RH, McMunigal JM (2001) Effects of simulated saltwater intrusions on the growth and survival of wild celery, Vallisneria americana, from the Caloosahatchee estuary (south Florida). Estuaries 24:894–903
Doust JL, Laporte G (1991) Population sex-ratios, population mixtures and fecundity in a clonal dioecious macrophyte, Vallisneria americana. J Ecol 79:477–489
Dudash M (1990) Relative fitness of selfed and outcrossed progeny in a self-compatible, protandrous species, Sabatia angularis L. (Gentianaceae): a comparison in three environments. Evolution 44:1129–1139
Ellstrand NC, Roose ML (1987) Patterns of genotypic diversity in clonal plant-species. Am J Bot 74:123–131
Fér T, Hroudová Z (2008) Detecting dispersal of Nuphar lutea in river corridors using microsatellite markers. Freshw Biol 53:1409–1422
Frankel OH (1974) Genetic conservation—our evolutionary responsibility. Genetics 78:53–65
Frankham R (1995a) Conservation genetics. Annu Rev Genet 29:305–327
Frankham R (1995b) Inbreeding and extinction—a threshold effect. Conserv Biol 9:792–799
Frankham R (1996) Relationship of genetic variation to population size in wildlife. Conserv Biol 10:1500–1508
Frankham R (2005) Stress and adaptation in conservation genetics. J Evol Biol 18:750–755
Gigord L, Lavigne C, Shykoff JA (1998) Partial self-incompatibility and inbreeding depression in a native tree species of La Réunion (Indian Ocean). Oecologia 117:342–352
Hammerli A, Reusch TBH (2003) Inbreeding depression influences genet size distribution in a marine angiosperm. Mol Ecol 12:619–629
Hedrick PW (2005) A standardized genetic differentiation measure. Evolution 59:1633–1638
Hedrick PW, Fredrickson R (2010) Genetic rescue guidelines with examples from Mexican wolves and Florida panthers. Conserv Genet 11:615–626
Holsinger KE, Weir BS (2009) Genetics in geographically structured populations: defining, estimating and interpreting Fst. Nat Rev Genet 10:639–650
Huelsenbeck JP, Andolfatto P (2007) Inference of population structure under a Dirichlet process model. Genetics 175:1787–1802
Hufford KM, Mazer SJ (2003) Plant ecotypes: genetic differentiation in the age of ecological restoration. Trends Ecol Evol 18:147–155
Hughes AR, Stachowicz JJ (2004) Genetic diversity enhances the resistance of a seagrass ecosystem to disturbance. Proc Natl Acad Sci USA 101:8998–9002
Hughes AR, Stachowicz JJ (2009) Ecological impacts of genotypic diversity in the clonal seagrass Zostera marina. Ecology 90:1412–1419
Hughes AR, Inouye BD, Johnson MTJ, Underwood N, Vellend M (2008) Ecological consequences of genetic diversity. Ecol Lett 11:609–623
Hunt G (1963) Wild celery in the lower Detroit River. Ecology 44:360–370
Hutchison DW, Templeton AR (1999) Correlation of pairwise genetic and geographic distance measures: inferring the relative influences of gene flow and drift on the distribution of genetic variability. Evolution 53:1898–1914
Jarvis JC, Moore KA (2008) Influence of environmental factors on Vallisneria americana seed germination. Aquat Bot 88:283–294
Jensen JL, Bohonak AJ, Kelley ST (2005) Isolation by distance, web service. BMC Genet 6:13
Jones AT, Hayes MJ, Hamilton NRS (2001) The effect of provenance on the performance of Crataegus monogyna in hedges. J Appl Ecol 38:952–962
Jost L (2008) G st and its relatives do not measure differentiation. Mol Ecol 17:4015–4026
Kalinowski ST (2004) Counting alleles with rarefaction: private alleles and hierarchical sampling designs. Conserv Genet 5:539–543
Kalinowski ST (2005) HP-RARE 1.0: a computer program for performing rarefaction on measures of allelic richness. Mol Ecol Notes 5:187–189
Keeley JE (1979) Population differentiation along a flood frequency gradient: physiological adaptations to flooding in Nyssa sylvatica. Ecol Monogr 49:89–108
Kemp WM, Boynton WR, Stevenson JC, Twilley RR, Means JC (1983) The decline of submerged vascular plants in upper Chesapeake Bay: summary of results concerning possible causes. Mar Technol Soc J 17:78–89
Kemp WM, Batiuk R, Bartleson R, Bergstrom P, Carter V, Gallegos CL, Hunley W, Karrh L, Koch EW, Landwehr JM, Moore KA, Murray L, Naylor M, Rybicki NB, Stevenson JC, Wilcox DJ (2004) Habitat requirements for submerged aquatic vegetation in Chesapeake Bay: water quality, light regime, and physical-chemical factors. Estuaries 27:363–377
Kemp WM, Boynton WR, Adolf JE, Boesch DF, Boicourt WC, Brush G, Cornwell JC, Fisher TR, Glibert PM, Hagy JD, Harding LW, Houde ED, Kimmel DG, Miller WD, Newell RIE, Roman MR, Smith EM, Stevenson JC (2005) Eutrophication of Chesapeake Bay: historical trends and ecological interactions. Mar Ecol Prog Ser 303:1–29
Kingman JFC (1982a) The coalescent. Stoch Process Appl 13:235–248
Kingman JFC (1982b) On the genealogy of large populations. J Appl Probab 19A:27–43
Korschgen C, Green W (1988) American wild celery (Vallisneria americana): ecological considerations for restoration. Technical report 19. US Fish and Wildlife Service, Washington, DC
Korschgen CE, Green WL, Kenow KP (1997) Effects of irradiance on growth and winter bud production by Vallisneria americana and consequences to its abundance and distribution. Aquat Bot 58:1–9
Kreiling RM, Yin Y, Gerber DT (2007) Abiotic influences on the biomass of Vallisneria americana Michx. in the Upper Mississippi River. River Res Appl 23:343–349
Krull JN (1970) Aquatic plant-macroinvertebrate associations and waterfowl. J Wildl Manag 34:707–718
Lande R, Shannon S (1996) The role of genetic variation in adaptation and population persistence in a changing environment. Evolution 50:434–437
Langeland KA (1996) Hydrilla verticillata (L.F.) Royle (Hydrocharitaceae), “The perfect aquatic weed”. Castanea 61:293–304
Laushman RH (1993) Population genetics of hydrophilous angiosperms. Aquat Bot 44:147–158
Lavergne S, Molofsky J (2007) Increased genetic variation and evolutionary potential drive the success of an invasive grass. Proc Natl Acad Sci USA 104:3883–3888
Lewis PO, Zaykin D (2001) Genetic data analysis: computer program for the analysis of alleleic data. Free program distributed by the authors over the internet from http://ewis.eeb.unconn.edu/lewishome/software.html
Lokker C, Susko D, Lovett-Doust L, Lovett-Doust J (1994) Population genetic structure of Vallisneria americana, a dioecious clonal macrophyte. Am J Bot 81:1004–1012
Linhart YB, Grant MC (1996) Evolutionary significance of local genetic differentiation in plants. Annu Rev Ecol Syst 27:237–277
Madsen JD, Chambers PA, James WF, Koch EW, Westlake DF (2001) The interaction between water movement, sediment dynamics and submersed macrophytes. Hydrobiologia 444:71–84
Mank JE, Avise JC (2003) Microsatellite variation and differentiation in North Atlantic eels. J Hered 94:310–314
McFarland DG, Shafer DJ (2008) Factors influencing reproduction in American wild celery: a synthesis. J Aquat Plant Manage 46:129–144
McKay JK, Christian CE, Harrison S, Rice KJ (2005) “How local is local?”—a review of practical and conceptual issues in the genetics of restoration. Restor Ecol 13:432–440
Meirmans PG (2006) Using the AMOVA framework to estimate a standardized genetic differentiation measure. Evolution 60:2399–2402
Meirmans PG, Van Tienderen PH (2004) GENOTYPE and GENODIVE: two programs for the analysis of genetic diversity of asexual organisms. Mol Ecol Notes 4:792–794
Mitton JB, Grant MC (1996) Genetic variation and the natural history of quaking aspen. Bioscience 46:25–31
Montalvo A, Ellstrand N (2001) Nonlocal transplantation and outbreeding depression in the subshrub Lotus scoparius (Fabaceae). Am J Bot 88:258–269
Montalvo AM, Williams SL, Rice KJ, Buchmann SL, Cory C, Handel SN, Nabhan GP, Primack R, Robichaux RH (1997) Restoration biology: a population biology perspective. Restor Ecol 5:277–290
Moore KA (2004) Influence of seagrasses on water quality in shallow regions of the lower Chesapeake Bay. J Coastal Res 45:162–178
Moore KA, Wilcox DJ, Orth RJ (2000) Analysis of the abundance of submersed aquatic vegetation communities in the Chesapeake Bay. Estuaries 23:115–127
Morrell PL, Lundy KE, Clegg MT (2003) Distinct geographic patterns of genetic diversity are maintained in wild barley (Hordeum vulgare ssp spontaneum) despite migration. Proc Natl Acad Sci USA 100:10812–10817
Orth RJ, Carruthers TJB, Dennison WC, Duarte CM, Fourqurean JW, Heck KL, Hughes AR, Kendrick GA, Kenworthy WJ, Olyarnik S, Short FT, Waycott M, Williams SL (2006) A global crisis for seagrass ecosystems. Bioscience 56:987–996
Orth RJ, Wilcox DJ, Whiting JR, Nagey LS, Owens AL, Kenne AK (2008) 2007 Distribution of submerged aquatic vegetation in the Chesapeake Bay and coastal bays. VIMS special scientific report number 151. Final report to US EPA, Chesapeake Bay Program, Annapolis, MD. http://webvimsedu/bio/sav/sav07/indexhtml
Parks JC, Werth CR (1993) A study of spatial features of clones in a population of bracken fern, Pteridium aquilinum (Dennstaedtiaceae). Am J Bot 80:537–544
Pleasants JM, Wendel JF (1989) Genetic diversity in a clonal narrow endemic, Erythronium propullans, and in its widespread progenitor, Erythronium albidum. Am J Bot 76:1136–1151
Pollux BJA, Jong MDE, Steegh A, Verbruggen E, Van Groenendael JM, Ouborg NJ (2007) Reproductive strategy, clonal structure and genetic diversity in populations of the aquatic macrophyte Sparganium emersum in river systems. Mol Ecol 16:313–325
Potts BM, Barbour RC, Hingston AB, Vaillancourt RE (2003) Genetic pollution of native eucalypt gene pools—identifying the risks. Aust J Bot 51:1–25
Pritchard DW (1952) Salinity distribution and circulation in the Chesapeake Bay estuarine system. J Mar Res 11:106–123
Pritchard JK, Stephens M, Donnelly P (2000) Inference of population structure using multilocus genotype data. Genetics 155:945–959
Procaccini G, Piazzi L (2001) Genetic polymorphism and transplantation success in the mediterranean seagrass Posidonia oceanica. Restor Ecol 9:332–338
Procaccini G, Olsen JL, Reusch TBH (2007) Contribution of genetics and genomics to seagrass biology and conservation. J Exp Mar Biol Ecol 350:234–259
Ray N (2005) PATHMATRIX: a GIS tool to compute effective distances among samples. Mol Ecol Notes 5:177–180
Reed DH, Frankham R (2003) Correlation between fitness and genetic diversity. Conserv Biol 17:230–237
Reusch TBH, Bostrom C, Stam WT, Olsen JL (1999a) An ancient eelgrass clone in the Baltic. Mar Ecol 183:301–304
Reusch TBH, Stam WT, Olsen JL (1999b) Microsatellite loci in eelgrass Zostera marina reveal marked polymorphism within and among populations. Mol Ecol 8:317–321
Reusch TBH, Stam WT, Olsen JL (2000) A microsatellite-based estimation of clonal diversity and population subdivision in Zostera marina, a marine flowering plant. Mol Ecol 9:127–140
Reusch TBH, Ehlers A, Hammerli A, Worm B (2005) Ecosystem recovery after climatic extremes enhanced by genotypic diversity. Proc Natl Acad Sci USA 102:2826–2831
Rhode JM, Duffy JE (2004) Relationships between bed age, bed size, and genetic structure in Chesapeake Bay (Virginia, USA) eelgrass (Zostera marina L.). Conserv Genet 5:661–671
Rogstad SH, Keane B, Beresh J (2002) Genetic variation across VNTR loci in central North American Taraxacum surveyed at different spatial scales. Plant Ecol 161:111–121
Rosenberg NA, Nordborg M (2002) Genealogical trees, coalescent theory and the analysis of genetic polymorphisms. Nat Rev Genet 3:380–390
Rozas LP, Minello TJ (2006) Nekton use of Vallisneria americana Michx. (wild celery) beds and adjacent habitats in coastal Louisiana. Estuar Coast 29:297–310
Rozas LP, Odum WE (1987) Fish and macrocrustacean use of submerged plant beds in tidal fresh-water marsh creeks. Mar Ecol 38:101–108
Rozas LP, Odum WE (1988) Occupation of submerged aquatic vegetation by fishes—testing the roles of food and refuge. Oecologia 77:101–106
Ruggiero MV, Turk R, Procaccini G (2002) Genetic identity and homozygosity in North-Adriatic populations of Posidonia oceanica: an ancient, post-glacial clone? Conserv Genet 3:71–74
Saccheri I, Kuussaari M, Kankare M, Vikman P, Fortelius W, Hanski I (1998) Inbreeding and extinction in a butterfly metapopulation. Nature 392:491–494
Sand-Jensen K (1998) Influence of submerged macrophytes on sediment composition and near-bed flow in lowland streams. Freshw Biol 39:663–679
Schwartz MK, McKelvey KS (2009) Why sampling scheme matters: the effect of sampling scheme on landscape genetic results. Conserv Gent 10:441–452
Short FT, Wyllie-Echeverria S (1996) Natural and human-induced disturbance of seagrasses. Environ Conserv 23:17–27
Slatkin M (1981) Estimating levels of gene flow in natural populations. Genetics 99:323–335
Slatkin M (1985) Gene flow in natural populations. Annu Rev Ecol Syst 16:393–430
Slatkin M (1987) Gene flow and the geographic structure of natural populations. Science 236:787–792
Slatkin M (1995) A measure of population subdivision based on microsatellite allele frequencies. Genetics 139:457–462
Society for Ecological Restoration International Science & Policy Working Group (2004) The SER international primer on ecological restoration. Society for Ecological Restoration International, Tucson. www.ser.org
Sork VL, Nason J, Campbell DR, Fernandez JF (1999) Landscape approaches to historical and contemporary gene flow in plants. Trends Ecol Evol 14:219–224
Stanton ML, Galen C, Shore J (1997) Population structure along a steep environmental gradient: consequences of flowering time and habitat variation in the snow buttercup, Ranunculus adoneus. Evolution 51:79–94
Stevenson JC, Confer NM (1978) Summary of available information on Chesapeake Bay submerged vegetation. Fish and Wildlife Service, Washington, DC, pp 283–335
Takamura N, Kadono Y, Fukushima M, Nakagawa M, Kim BHO (2003) Effects of aquatic macrophytes on water quality and phytoplankton communities in shallow lakes. Ecol Res 18:381–395
Templeton AR (1997) Coadaptation and outbreeding depression. In: Soule M (ed) Conservation biology the science of scarcity and diversity. Sinauer Associates, Inc., Sunderland, pp 105–116
Thuillet AC, Bru D, David J, Roumet P, Santomi S, Sourdille P, Bataillon T (2002) Direct estimation of mutation rate for 10 microsatellite loci in durum wheat, Triticum turgidum (L.) Thell. ssp durum desf. Mol Biol Evol 19:122–125
Till-Bottraud I, Poncet BN, Rioux D, Girel J (2010) Spatial structure and clonal distribution of genotypes in the rare Typha minima Hoppe (Typhaceae) along a river system. Bot Helv 120:53–62
Titus JE, Adams MS (1979) Coexistence and the comparative light relations of the submersed macrophytes Myriophyllum spicatum L. and Vallisneria americana Michx. Oecologia 40:273–286
Titus JE, Hoover DT (1991) Toward predicting reproductive success in submersed freshwater angiosperms. Aquat Bot 41:111–136
Titus JE, Stephens MD (1983) Neighbor influences and seasonal growth-patterns for Vallisneria americana in a mesotrophic lake. Oecologia 56:23–29
van Dijk JK, van Tussenbroek BI, Jimenez-Duran K, Marquez-Guzman GJ, Ouborg J (2009) High levels of gene flow and low population genetic structure related to high dispersal potential of a tropical marine angiosperm. Mar Ecol 390:67–77
van Katwijk M, Bos A, de Jonge V, Hanssen L, Hermus D, de Jong D (2009) Guidelines for seagrass restoration: Importance of habitat selection and donor population, spreading of risks, and ecosystem engineering effects. Mar Pollut Bull 58:179–188
Vigouroux Y, Jaqueth JS, Matsuoka Y, Smith OS, Beavis WF, Smith JSC, Doebley J (2002) Rate and pattern of mutation at microsatellite loci in maize. Mol Biol Evol 19:1251–1260
Wang B, Song Z, Liu G, Lu F, Li W (2010) Comparison of the extent of genetic variation of Vallisneria natans and its sympatric congener V. spinulosa in lakes of the middle-lower reaches of the Yangtze River. Aquat Bot. doi:10.1016/j.aquabot.2009.1012.1006
Weir BS, Cockerham CC (1984) Estimating F-statistics for the analysis of population structure. Evolution 38:1358–1370
Westemeier RL, Brawn JD, Simpson SA, Esker TL, Jansen RW, Walk JW, Kershner EL, Bouzat JL, Paige KN (1998) Tracking the long-term decline and recovery of an isolated population. Science 282:1695–1698
Wilder GJ (1974) Symmetry and development of pistillate Vallisneria americana (Hydrocharitaceae). Am J Bot 61:846–866
Williams SL (2001) Reduced genetic diversity in eelgrass transplantations affects both population growth and individual fitness. Ecol Appl 11:1472–1488
Williams SL, Davis CA (1996) Population genetic analyses of transplanted eelgrass (Zostera marina) beds reveal reduced genetic diversity in southern California. Restor Ecol 4:163–180
Wright S (1931) Evolution in Mendelian populations. Genetics 16:97–159
Wyda JC, Deegan LA, Hughes JE, Weaver MJ (2002) The response of fishes to submerged aquatic vegetation complexity in two ecoregions of the mid-atlantic bight: Buzzards Bay and Chesapeake Bay. Estuaries 25:86–100
Yeh FC, Yang R-C, Boyle TBJ, Ye Z-H, Mao JX (1997) POPGENE, the user-friendly shareware for population genetic analysis. Molecular Biology and Biotechnology Centre, University of Alberta, Canada
Young A, Boyle T, Brown T (1996) The population genetic consequences of habitat fragmentation for plants. Trends Ecol Evol 11:413–418
Zipperle AM, Coyer JA, Reise K, Gitz E, Stam WT, Olsen JL (2009) Clonal architecture in an intertidal bed of the dwarf eelgrass Zostera noltii in the Northern Wadden Sea: persistence through extreme physical perturbation and the importance of a seed bank. Mar Biol 156:2139–2148
Acknowledgments
The authors gratefully acknowledge the assistance of a number of organizations and individuals that were instrumental in assistance with location of sites and collection of samples: Maryland Department of Natural Resources, North Bay Camp, Jason Granberg, Peter Bergstrom, Lee Karrh, Mark Lewandowski, Stan Kollar, Nancy Rybicki, Todd Beser, and Jason Jullian. We thank Paul Widmeyer for creating the study area map. Our funding was provided through NOAA Sea Grant Maryland, and University of Maryland Department of Plant Science and Landscape Architecture and Maryland Agricultural Experiment Station.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Lloyd, M.W., Burnett, R.K., Engelhardt, K.A.M. et al. The structure of population genetic diversity in Vallisneria americana in the Chesapeake Bay: implications for restoration. Conserv Genet 12, 1269–1285 (2011). https://doi.org/10.1007/s10592-011-0228-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10592-011-0228-7