Abstract
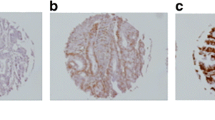
Cancer of unknown primary (CUP) is a heterogeneous entity, managed on the basis of “one size fits all” therapeutic concepts; insights into the molecular biology of CUP are urgently needed. We retrospectively examined the immunohistochemical (IHC) expression of Notch1, 2, 3, Jagged1, cMET, and pMAPK biomolecules in 100 CUP tumors using tissue microarrays, aiming to study their correlation to clinicopathologic characteristics and prognostic utility for patient outcome. Notch3 and pMAPK were most frequently expressed (97 and 91 %, respectively). A linear correlation of Notch3 and cMET expression was found (p = 0.001), while pMAPK emerged as the major adverse prognostic factor (median overall survival OS 9 vs. 17 months, p = 0.016), carrying also a significantly positive predictive value (p = 0.02). Our study indicated a favorable prognostic impact of cMET expression in CUP, both in univariate (median OS 15 vs. 9 months, p = 0.05) and in multivariate analysis (Relative Risk RR for death 0.48, p = 0.025). cMET and Notch3 expression were found to be statistically more frequent in squamous carcinomas (positive in 90 % of cases), associated with a unique metastatic IHC pattern (cMET-high in soft tissue/lymph node metastases, p < 0.001, Notch3-high in visceral, peritoneal/pleural and soft tissue/lymph node metastases, p < 0.001). Our study points to the MAPK and cMET axes as crucial in defining cancer progression and outcome in CUP patients and, if validated, could justify attempts at their therapeutic modulation.




Similar content being viewed by others
References
Golfinopoulos V, Pentheroudakis G, Salanti G, Nearchou AD, Ioannidis JP, Pavlidis N (2009) Comparative survival with diverse chemotherapy regimens for cancer of unknown primary site: multiple-treatments meta-analysis. Cancer Treat Rev 35:570–573
Borggrefe T, Oswald F (2009) The Notch signaling pathway: transcriptional regulation at Notch target genes. Cell Mol Life Sci 66:1631–1646
Miele L (2006) Notch signaling. Clin Cancer Res 12:1074–1079
Miele L, Miao H, Nickoloff BJ (2006) NOTCH signaling as a novel cancer therapeutic target. Curr Cancer Drug Targets 6:313–323
Wang Z, Li Y, Kong D, Sarkar FH (2010) The role of Notch signaling pathway in epithelial-mesenchymal transition (EMT) during development and tumor aggressiveness. Curr Drug Targets 11:745–751
Wang Z, Li Y, Banerjee S, Sarkar FH (2009) Emerging role of Notch in stem cells and cancer. Cancer Lett 279:8–12
McCubrey JA, Steelman LS, Chappell WH, Abrams SL, Wong EW, Chang F, Lehmann B, Terrian DM, Milella M, Tafuri A, Stivala F, Libra M, Basecke J, Evangelisti C, Martelli AM, Franklin RA (2007) Roles of the Raf/MEK/ERK pathway in cell growth, malignant transformation and drug resistance. Biochim Biophys Acta 1773:1263–1284
Davis JM, Navolanic PM, Weinstein-Oppenheimer CR, Steelman LS, Hu W, Konopleva M, Blagosklonny MV, McCubrey JA (2003) Raf-1 and Bcl-2 induce distinct and common pathways that contribute to breast cancer drug resistance. Clin Cancer Res 9:1161–1170
Birchmeier C, Birchmeier W, Gherardi E, Vande Woude GF (2003) Met, metastasis, motility and more. Nat Rev Mol Cell Biol 4:915–925
Pavlidis N, Briasoulis E, Hainsworth J, Greco FA (2003) Diagnostic and therapeutic management of cancer of an unknown primary. Eur J Cancer 39:1990–2005
McShane LM, Altman DG, Sauerbrei W, Taube SE, Gion M, Clark GM (2005) Reporting recommendations for tumor marker prognostic studies. J Clin Oncol 23:9067–9072
Hainsworth JD, Lennington WJ, Greco FA (2000) Overexpression of Her-2 in patients with poorly differentiated carcinoma or poorly differentiated adenocarcinoma of unknown primary site. J Clin Oncol 18:632–635
Pavlidis N, Briassoulis E, Bai M, Fountzilas G, Agnantis N (1995) Overexpression of C-myc, Ras and C-erbB-2 oncoproteins in carcinoma of unknown primary origin. Anticancer Res 15:2563–2567
Dova L, Pentheroudakis G, Georgiou I, Malamou-Mitsi V, Vartholomatos G, Fountzilas G, Kolaitis N, Kitsiou E, Pavlidis N (2007) Global profiling of EGFR gene mutation, amplification, regulation and tissue protein expression in unknown primary carcinomas: to target or not to target? Clin Exp Metastasis 24:79–86
Dova L, Pentheroudakis G, Golfinopoulos V, Malamou-Mitsi V, Georgiou I, Vartholomatos G, Ntemou A, Fountzilas G, Pavlidis N (2008) Targeting c-KIT, PDGFR in cancer of unknown primary: a screening study for molecular markers of benefit. J Cancer Res Clin Oncol 134:697–704
Briasoulis E, Tsokos M, Fountzilas G, Bafaloukos D, Kosmidis P, Samantas E, Skarlos D, Nicolaides C, Pavlidis N (1998) Bcl2 and p53 protein expression in metastatic carcinoma of unknown primary origin: biological and clinical implications. A Hellenic Co-operative Oncology Group study. Anticancer Res 18:1907–1914
Karavasilis V, Malamou-Mitsi V, Briasoulis E, Tsanou E, Kitsou E, Kalofonos H, Fountzilas G, Fotsis T, Pavlidis N (2005) Angiogenesis in cancer of unknown primary: clinicopathological study of CD34, VEGF and TSP-1. BMC Cancer 5:25
Hainsworth JD, Spigel DR, Rubin MS, Boccia RV, Fox EP, Firdaus I, Erlander MG, Schnabel C, Greco FA (2010) Treatment of carcinoma of unknown primary site (CUP) directed by molecular profiling diagnosis: a prospective, phase II trial. J Clin Oncol 28
Dang TP, Gazdar AF, Virmani AK, Sepetavec T, Hande KR, Minna JD, Roberts JR, Carbone DP (2000) Chromosome 19 translocation, overexpression of Notch3, and human lung cancer. J Natl Cancer Inst 92:1355–1357
Haruki N, Kawaguchi KS, Eichenberger S, Massion PP, Olson S, Gonzalez A, Carbone DP, Dang TP (2005) Dominant-negative Notch3 receptor inhibits mitogen-activated protein kinase pathway and the growth of human lung cancers. Cancer Res 65:3555–3561
Radtke F, Wilson A, Mancini SJ, MacDonald HR (2004) Notch regulation of lymphocyte development and function. Nat Immunol 5:247–253
Harrison H, Farnie G, Howell SJ, Rock RE, Stylianou S, Brennan KR, Bundred NJ, Clarke RB (2010) Regulation of breast cancer stem cell activity by signaling through the Notch4 receptor. Cancer Res 70:709–718
Grugan KD, Miller CG, Yao Y, Michaylira CZ, Ohashi S, Klein-Szanto AJ, Diehl JA, Herlyn M, Han M, Nakagawa H, Rustgi AK (2010) Fibroblast-secreted hepatocyte growth factor plays a functional role in esophageal squamous cell carcinoma invasion. Proc Natl Acad Sci USA 107:11026–11031
Seiwert TY, Jagadeeswaran R, Faoro L, Janamanchi V, Nallasura V, El Dinali M, Yala S, Kanteti R, Cohen EE, Lingen MW, Martin L, Krishnaswamy S, Klein-Szanto A, Christensen JG, Vokes EE, Salgia R (2009) The MET receptor tyrosine kinase is a potential novel therapeutic target for head and neck squamous cell carcinoma. Cancer Res 69:3021–3031
Kim CH, Kim J, Kahng H, Choi EC (2007) Change of E-cadherin by hepatocyte growth factor and effects on the prognosis of hypopharyngeal carcinoma. Ann Surg Oncol 14:1565–1574
Yu J, Shi R, Zhang D, Wang E, Qiu X (2011) Expression of integrin-linked kinase in lung squamous cell carcinoma and adenocarcinoma: correlation with E-cadherin expression, tumor microvessel density and clinical outcome. Virchows Arch 458:99–107
Stoyianni A, Goussia A, Pentheroudakis G, Siozopoulou V, Ioachim E, Krikelis D, Golfinopoulos V, Cervantes A, Bobos M, Fotsis T, Bellou S, Fountzilas G, Malamou-Mitsi V, Pavlidis N (2012) Immunohistochemical study of the epithelial-mesenchymal transition phenotype in cancer of unknown primary: incidence, correlations and prognostic utility. Anticancer Res
Hu YC, Lam KY, Law S, Wong J, Srivastava G (2001) Profiling of differentially expressed cancer-related genes in esophageal squamous cell carcinoma (ESCC) using human cancer cDNA arrays: overexpression of oncogene MET correlates with tumor differentiation in ESCC. Clin Cancer Res 7:3519–3525
Crosswell HE, Dasgupta A, Alvarado CS, Watt T, Christensen JG, De P, Durden DL, Findley HW (2009) PHA665752, a small-molecule inhibitor of c-Met, inhibits hepatocyte growth factor-stimulated migration and proliferation of c-Met-positive neuroblastoma cells. BMC Cancer 9:411
Todaro M, Iovino F, Eterno V, Cammareri P, Gambara G, Espina V, Gulotta G, Dieli F, Giordano S, De Maria R, Stassi G (2010) Tumorigenic and metastatic activity of human thyroid cancer stem cells. Cancer Res 70:8874–8885
Stella GM, Benvenuti S, Gramaglia D, Scarpa A, Tomezzoli A, Cassoni P, Senetta R, Venesio T, Pozzi E, Bardelli A, Comoglio PM (2011) MET mutations in cancers of unknown primary origin (CUPs). Hum Mutat 32:44–50
Radtke F, Raj K (2003) The role of Notch in tumorigenesis: oncogene or tumour suppressor? Nat Rev Cancer 3:756–767
Sundaram MV (2005) The love-hate relationship between Ras and Notch. Genes Dev 19:1825–1839
Klotz R, Zeimet AG, Reimer D, Muller-Holzner E, Chamson M, Marth C (2008) Activated p38-MAPK and gemcitabine sensitivity in recurrent ovarian cancer. Anticancer Res 28:2975–2980
Inno A, Salvatore MD, Cenci T, Martini M, Orlandi A, Strippoli A, Ferrara AM, Bagala C, Cassano A, Larocca LM, Barone C (2011) Is there a role for IGF1R and c-MET pathways in resistance to cetuximab in metastatic colorectal cancer? Clin Colorectal Cancer 10:325–332
Gu F, Ma Y, Zhang Z, Zhao J, Kobayashi H, Zhang L, Fu L (2010) Expression of Stat3 and Notch1 is associated with cisplatin resistance in head and neck squamous cell carcinoma. Oncol Rep 23:671–676
Rudiger T, Hofler H, Kreipe HH, Nizze H, Pfeifer U, Stein H, Dallenbach FE, Fischer HP, Mengel M, von Wasielewski R, Muller-Hermelink HK (2002) Quality assurance in immunohistochemistry: results of an interlaboratory trial involving 172 pathologists. Am J Surg Pathol 26:873–882
Conflict of interest
The authors declare that they have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Krikelis, D., Pentheroudakis, G., Goussia, A. et al. Profiling immunohistochemical expression of NOTCH1–3, JAGGED1, cMET, and phospho-MAPK in 100 carcinomas of unknown primary. Clin Exp Metastasis 29, 603–614 (2012). https://doi.org/10.1007/s10585-012-9474-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10585-012-9474-4