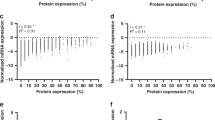
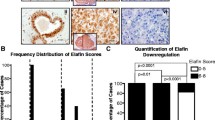
Abstract
Epithelial ovarian cancer is the most lethal gynecologic cancer with a 5 years survival rate of 30–40% in patients diagnosed with high-grade invasive disease (TOV). This is in stark contrast to the 95% 5 years survival rate in ovarian cancer patients diagnosed with low malignant potential (LMP) disease. The progression from localized tumor to invasive metastasis involves matrix proteolysis. Protease inhibitors are thought to play a key role by limiting this process. Using the Affymetrix HG-U133A GeneChip array, we have studied all serine protease inhibitors and found several serpin family members that are differentially expressed between LMP and TOV serous tumors. SERPINA1 was selected for further study due to its high expression in the majority of LMP tumors and its low expression in TOV tumors; observations that were also validated by quantitative-PCR (Q-PCR). To study the effects of its over expression on different tumorigenic parameters, SERPINA1 was cloned in the pcDNA3.1+ plasmid which was subsequently used to derive stable clones from two invasive ovarian cancer cell lines, TOV-112D and TOV-1946. We found no effect of SERPINA1 over expression on tumor growth in SCID mice although cell migration and invasion were affected in in vitro assays. There was also no association between patient survival and SERPINA1 immunostaining, however, SERPINA1 localization was different in LMP (nuclear) and TOV (cytoplasmic) tumors. SERPINA1 remains an interesting candidate since protein homeostasis, regulated by proteases and their inhibitors, should be studied holistically in order to assess their full impact in tumor progression.






Similar content being viewed by others

Abbreviations
- AAT/SERPINA1:
-
Alpha 1-antitrypsin/Serpin peptidase inhibitor, clade A, member 1
- BOV:
-
Benign ovarian tumor
- DAB:
-
Diaminobenzidine
- ELISA:
-
Enzyme-linked immunosorbent assay
- ERK1:
-
Extracellular signal-regulated kinase 1
- FBS:
-
Fetal bovine serum
- FIGO:
-
International Federation of Gynecology and Obstetrics
- G:
-
Tumor grade
- ICC:
-
Intraclass correlation coefficient
- LMP:
-
Low malignant potential tumor
- OSE:
-
Ovarian surface epithelium
- ROC:
-
Receiver Operating Characteristic
- SCID:
-
Severe combined immunodeficiency
- S2N:
-
Signal-to-noise ratio
- TOV:
-
Invasive ovarian tumor
References
Permuth-Wey J, Sellers TA (2009) Epidemiology of ovarian cancer. Methods Mol Biol 472:413–437
Colombo N, Van Gorp T, Parma G et al (2006) Ovarian cancer. Crit Rev Oncol Hematol 60:159–179
Bell DA (2005) Origins and molecular pathology of ovarian cancer. Mod Pathol 18(Suppl 2):S19–S32
Crum CP, Drapkin R, Kindelberger D et al (2007) Lessons from BRCA: the tubal fimbria emerges as an origin for pelvic serous cancer. Clin Med Res 5:35–44
Crum CP, Drapkin R, Miron A et al (2007) The distal fallopian tube: a new model for pelvic serous carcinogenesis. Curr Opin Obstet Gynecol 19:3–9
Benedet JL (2006) Staging classifications and clinical practice guidelines for gynaecologic cancers. In: Publications of the international federation of gynecology and obstetrics. Available via http://www.figo.org/publications
Bell DA, Longacre TA, Prat J et al (2004) Serous borderline (low malignant potential, atypical proliferative) ovarian tumors: workshop perspectives. Hum Pathol 35:934–948
Singer G, Oldt R III, Cohen Y et al (2003) Mutations in BRAF and KRAS characterize the development of low-grade ovarian serous carcinoma. J Natl Cancer Inst 95:484–486
Singer G, Stohr R, Cope L et al (2005) Patterns of p53 mutations separate ovarian serous borderline tumors and low- and high-grade carcinomas and provide support for a new model of ovarian carcinogenesis: a mutational analysis with immunohistochemical correlation. Am J Surg Pathol 29:218–224
Bonome T, Lee JY, Park DC et al (2005) Expression profiling of serous low malignant potential, low-grade, and high-grade tumors of the ovary. Cancer Res 65:10602–10612
Chen VW, Ruiz B, Killeen JL et al (2003) Pathology and classification of ovarian tumors. Cancer 97:2631–2642
Chambers AF, Groom AC, MacDonald IC (2002) Dissemination and growth of cancer cells in metastatic sites. Nat Rev Cancer 2:563–572
Friedl P, Wolf K (2003) Tumour-cell invasion and migration: diversity and escape mechanisms. Nat Rev Cancer 3:362–374
Aznavoorian S, Murphy AN, Stetler-Stevenson WG et al (1993) Molecular aspects of tumor cell invasion and metastasis. Cancer 71:1368–1383
Duffy MJ (1992) The role of proteolytic enzymes in cancer invasion and metastasis. Clin Exp Metastasis 10:145–155
Duffy MJ (1996) Proteases as prognostic markers in cancer. Clin Cancer Res 2:613–618
Schmitt M (1992) Tumor associated proteases. Fibrinolysis 6:3–26
Gettins PG (2002) Serpin structure, mechanism, and function. Chem Rev 102:4751–4804
van Gent D, Sharp P, Morgan K et al (2003) Serpins: structure, function and molecular evolution. Int J Biochem Cell Biol 35:1536–1547
de Koning PJ, Bovenschen N, Leusink FK et al (2009) Downregulation of SERPINB13 expression in head and neck squamous cell carcinomas associates with poor clinical outcome. Int J Cancer 125:1542–1550
Croucher DR, Saunders DN, Lobov S et al (2008) Revisiting the biological roles of PAI2 (SERPINB2) in cancer. Nat Rev Cancer 8:535–545
Bailey CM, Khalkhali-Ellis Z, Seftor EA et al (2006) Biological functions of maspin. J Cell Physiol 209:617–624
Zou Z, Anisowicz A, Hendrix MJ et al (1994) Maspin, a serpin with tumor-suppressing activity in human mammary epithelial cells. Science 263:526–529
Sieben NL, Oosting J, Flanagan AM et al (2005) Differential gene expression in ovarian tumors reveals Dusp 4 and Serpina 5 as key regulators for benign behavior of serous borderline tumors. J Clin Oncol 23:7257–7264
Tamura K, Furihata M, Tsunoda T et al (2007) Molecular features of hormone-refractory prostate cancer cells by genome-wide gene expression profiles. Cancer Res 67:5117–5125
Warrenfeltz S, Pavlik S, Datta S et al (2004) Gene expression profiling of epithelial ovarian tumours correlated with malignant potential. Mol Cancer 3:27
Carlson JA, Rogers BB, Sifers RN et al (1988) Multiple tissues express alpha 1-antitrypsin in transgenic mice and man. J Clin Invest 82:26–36
Carrell RW (1986) alpha 1-Antitrypsin: molecular pathology, leukocytes, and tissue damage. J Clin Invest 78:1427–1431
Kalsheker N, Morley S, Morgan K (2002) Gene regulation of the serine proteinase inhibitors alpha1-antitrypsin and alpha1-antichymotrypsin. Biochem Soc Trans 30:93–98
Kohnlein T, Welte T (2008) Alpha-1 antitrypsin deficiency: pathogenesis, clinical presentation, diagnosis, and treatment. Am J Med 121:3–9
Law RH, Zhang Q, McGowan S et al (2006) An overview of the serpin superfamily. Genome Biol 7:216
Lisowska-Myjak B (2005) AAT as a diagnostic tool. Clin Chim Acta 352:1–13
Silverman GA, Bird PI, Carrell RW et al (2001) The serpins are an expanding superfamily of structurally similar but functionally diverse proteins. Evolution, mechanism of inhibition, novel functions, and a revised nomenclature. J Biol Chem 276:33293–33296
Travis J, Salvesen GS (1983) Human plasma proteinase inhibitors. Annu Rev Biochem 52:655–709
Ouellet V, Zietarska M, Portelance L et al (2008) Characterization of three new serous epithelial ovarian cancer cell lines. BMC Cancer 8:152
Provencher DM, Lounis H, Champoux L et al (2000) Characterization of four novel epithelial ovarian cancer cell lines. In Vitro Cell Dev Biol Anim 36:357–361
Ouellet V, Provencher DM, Maugard CM et al (2005) Discrimination between serous low malignant potential and invasive epithelial ovarian tumors using molecular profiling. Oncogene 24:4672–4687
Puiffe ML, Le Page C, Filali-Mouhim A et al (2007) Characterization of ovarian cancer ascites on cell invasion, proliferation, spheroid formation, and gene expression in an in vitro model of epithelial ovarian cancer. Neoplasia 9:820–829
Novak JP, Sladek R, Hudson TJ (2002) Characterization of variability in large-scale gene expression data: implications for study design. Genomics 79:104–113
Golub TR, Slonim DK, Tamayo P et al (1999) Molecular classification of cancer: class discovery and class prediction by gene expression monitoring. Science 286:531–537
Tusher VG, Tibshirani R, Chu G (2001) Significance analysis of microarrays applied to the ionizing radiation response. Proc Natl Acad Sci U S A 98:5116–5121
Peant B, Diallo JS, Dufour F et al (2009) Over-expression of IkappaB-kinase-epsilon (IKKepsilon/IKKi) induces secretion of inflammatory cytokines in prostate cancer cell lines. Prostate 69:706–718
Wolf K, Wu YI, Liu Y et al (2007) Multi-step pericellular proteolysis controls the transition from individual to collective cancer cell invasion. Nat Cell Biol 9:893–904
Hanahan D, Weinberg RA (2000) The hallmarks of cancer. Cell 100:57–70
Ludwig T (2005) Local proteolytic activity in tumor cell invasion and metastasis. Bioessays 27:1181–1191
Sato T, Takahashi S, Mizumoto T et al (2006) Neutrophil elastase and cancer. Surg Oncol 15:217–222
Kobel M, Kalloger SE, Boyd N et al (2008) Ovarian carcinoma subtypes are different diseases: implications for biomarker studies. PLoS Med 5:e232
Li W, Savinov AY, Rozanov DV et al (2004) Matrix metalloproteinase-26 is associated with estrogen-dependent malignancies and targets alpha1-antitrypsin serpin. Cancer Res 64:8657–8665
O’Neill CJ, McBride HA, Connolly LE et al (2007) High-grade ovarian serous carcinoma exhibits significantly higher p16 expression than low-grade serous carcinoma and serous borderline tumour. Histopathology 50:773–779
Ouellet V, Ling TH, Normandin K et al (2008) Immunohistochemical profiling of benign, low malignant potential and low grade serous epithelial ovarian tumors. BMC Cancer 8:346
Schmeler KM, Gershenson DM (2008) Low-grade serous ovarian cancer: a unique disease. Curr Oncol Rep 10:519–523
Wolf K, Mazo I, Leung H et al (2003) Compensation mechanism in tumor cell migration: mesenchymal-amoeboid transition after blocking of pericellular proteolysis. J Cell Biol 160:267–277
Wong SY, Hynes RO (2006) Lymphatic or hematogenous dissemination: how does a metastatic tumor cell decide? Cell Cycle 5:812–817
Taetle R, Aickin M, Yang JM et al (1999) Chromosome abnormalities in ovarian adenocarcinoma: I. Nonrandom chromosome abnormalities from 244 cases. Genes Chromosomes Cancer 25:290–300
Bagdasarian A, Wheeler J, Stewart GJ et al (1981) Isolation of alpha 1-protease inhibitor from human normal and malignant ovarian tissue. J Clin Invest 67:281–291
Ny T, Wahlberg P, Brandstrom IJ (2002) Matrix remodeling in the ovary: regulation and functional role of the plasminogen activator and matrix metalloproteinase systems. Mol Cell Endocrinol 187:29–38
Allgayer H, Babic R, Grutzner KU et al (1998) Tumor-associated proteases and inhibitors in gastric cancer: analysis of prognostic impact and individual risk protease patterns. Clin Exp Metastasis 16:62–73
Karashima S, Kataoka H, Itoh H et al (1990) Prognostic significance of alpha-1-antitrypsin in early stage of colorectal carcinomas. Int J Cancer 45:244–250
Higashiyama M, Doi O, Kodama K et al (1992) An evaluation of the prognostic significance of alpha-1-antitrypsin expression in adenocarcinomas of the lung: an immunohistochemical analysis. Br J Cancer 65:300–302
Kloth JN, Gorter A, Fleuren GJ et al (2008) Elevated expression of SerpinA1 and SerpinA3 in HLA-positive cervical carcinoma. J Pathol 215:222–230
Sood AK, Fletcher MS, Gruman LM et al (2002) The paradoxical expression of maspin in ovarian carcinoma. Clin Cancer Res 8:2924–2932
Keppler D, Markert M, Carnal B et al (1996) Human colon carcinoma cells synthesize and secrete alpha 1-proteinase inhibitor. Biol Chem Hoppe Seyler 377:301–311
Yavelow J, Tuccillo A, Kadner SS et al (1997) Alpha 1-antitrypsin blocks the release of transforming growth factor-alpha from MCF-7 human breast cancer cells. J Clin Endocrinol Metab 82:745–752
Lah TT, Duran Alonso MB, Van Noorden CJ (2006) Antiprotease therapy in cancer: hot or not? Expert Opin Biol Ther 6:257–279
Acknowledgments
We are grateful to Jason Madore, Kim Leclerc Desaulniers, Liliane Meunier, Lise Portelance, Marie-Line Puiffe, and Louise Champoux for technical assistance. We would like to thank laboratory members for thoughtful discussion, and Luke Masson, Jason Madore and Jennifer Kendall-Dupont for careful reading of the paper. We are also grateful to the members of the Department of Gynecologic Oncology and the Department of pathology of the CHUM for providing specimens. This research was supported by grants from Génome Canada/Génome Québec and the Canadian Institutes of Health Research (CIHR) to Anne-Marie Mes-Masson, Patricia N. Tonin, and Diane M. Provencher. Tumor banking was supported by the Banque de tissus et de données du Réseau de recherche sur le cancer des Fonds de la Recherche en Santé du Québec (FRSQ), affiliated to the Canadian Tumor Repository Network (CTRNet). Karine Normandin was supported by studentships from the Canderel Fund of the Institut du cancer de Montréal and the molecular biology department of the Faculté des études supérieures de l’Université de Montréal. Véronique Ouellet was supported by studentships from the CIHR and the Canderel Fund of the Institut du cancer de Montréal.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
10585_2009_9303_MOESM1_ESM.ppt
Supplementary Table 1 Clinical characteristics of tumors used for RT-Q-PCR analysis. No patient received chemotherapy prior to the surgery. (PPT 49 kb)
10585_2009_9303_MOESM2_ESM.ppt
Supplementary Figure 1 SERPINA1 mRNA and protein expression in TOV-112D and TOV-1946 derived clones. a Expression of SERPINA1 mRNA in transfected clones. After 30 rounds of SERPINA1 and ERK1 amplification, PCR products were separated on a 2% agarose gel. ERK1 was used as a reference gene. b Overexposure of low level expressing clones. Total protein extracts (50 μg) were separated on a 10% polyacrylamide gel and transferred onto PVDF membrane. Equal loading was evaluated using a β-actin antibody. (PPT 236 kb)
10585_2009_9303_MOESM3_ESM.ppt
Supplementary Figure 2 High variability of tumor formation in SCID mice injected with TOV-1946 derived clones. (PPT 193 kb)
10585_2009_9303_MOESM4_ESM.ppt
Supplementary Figure 3 In situ expression of SERPINA1 in ovarian tissues. a Specificity of SERPINA1 antibody tested by immunocytochemistry on TOV-1946 over expressing SERPINA1 cells and the parental TOV-1946 cell line. b Staining of both cell lines (TOV-112D and TOV-1946); and the different subcellular localizations of SERPINA1 in LMP and TOV tumors obtained by immunohistochemistry on the ovarian cancer tissue microarray. (PPT 7721 kb)
10585_2009_9303_MOESM5_ESM.ppt
Supplementary Figure 4 Cellular morphology of the SERPINA1 over expressing clone S112D-1, the TOV-112D cell line, and the control clone C112D-B. (PPT 617 kb)
Rights and permissions
About this article
Cite this article
Normandin, K., Péant, B., Le Page, C. et al. Protease inhibitor SERPINA1 expression in epithelial ovarian cancer. Clin Exp Metastasis 27, 55–69 (2010). https://doi.org/10.1007/s10585-009-9303-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10585-009-9303-6