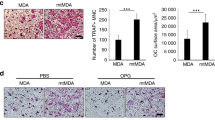
Abstract
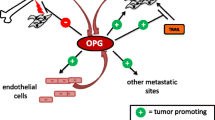
The bone microenvironment plays a critical role in tumor-induced osteolysis and osteolytic metastasis through tumor–bone (TB)-interaction. Receptor activator of nuclear factor-κB (RANK) ligand (RANKL) is one of the critical signaling molecules involved in osteolysis and bone metastasis. However, the regulation and functional significance of RANKL at the TB-interface in tumor-induced osteolysis remains unclear. In this report, we examined the role of tumor–stromal interaction in the regulation of RANKL expression and its functional significance in tumor-induced osteolysis. Using a novel mammary tumor model, we identified that RANKL expression was upregulated at the TB-interface as compared to the tumor alone area. We demonstrate increased generation of sRANKL at the TB-interface, which is associated with tumor-induced osteolysis. The ratio of RANKL to osteoprotegrin (OPG), a decoy receptor for RANKL, at the TB-interface was also increased. Targeting RANKL expression with antisense oligonucleotides (RANKL-ASO), significantly abrogated tumor-induced osteolysis, decreased RANKL expression and the RANKL:OPG ratio at the TB-interface. Together, these results demonstrate that upregulation of RANKL expression and sRANKL generation at the TB-interface potentiates tumor-induced osteolysis.






Similar content being viewed by others
References
Jemal A, Siegel R, Ward E, Hao Y, Xu J, Murray T et al (2008) Cancer statistics, 2008. CA Cancer J Clin 58(2):71–96
Mundy GR (2002) Metastasis to bone: causes, consequences and therapeutic opportunities. Nat Rev Cancer 2(8):584–593
Boyce BF, Yoneda T, Guise TA (1999) Factors regulating the growth of metastatic cancer in bone. Endocr Relat Cancer 6(3):333–347
Coleman RE (1997) Skeletal complications of malignancy. Cancer 80(8 Suppl):1588–1594
Kakonen SM, Mundy GR (2003) Mechanisms of osteolytic bone metastases in breast carcinoma. Cancer 97(3 Suppl):834–839
Roodman GD (2004) Mechanisms of bone metastasis. N Engl J Med 350(16):1655–1664
Lacey DL, Tan HL, Lu J, Kaufman S, Van G, Qiu W et al (2000) Osteoprotegerin ligand modulates murine osteoclast survival in vitro and in vivo. Am J Pathol 157(2):435–448
Dougall WC, Chaisson M (2006) The RANK/RANKL/OPG triad in cancer-induced bone diseases. Cancer Metastasis Rev 25(4):541–549
Morrissey C, Kostenuik P, Brown L, Vessella R, Corey E (2007) Host-derived RANKL is responsible for osteolysis in a C4-2 human prostate cancer xenograft model of experimental bone metastases. BMC Cancer 7(1):148
Dallas SL, Rosser JL, Mundy GR, Bonewald LF (2002) Proteolysis of latent transforming growth factor-beta (TGF-beta)-binding protein-1 by osteoclasts. A cellular mechanism for release of TGF-beta from bone matrix. J Biol Chem 277(24):21352–21360
Lacey DL, Timms E, Tan H-L, Kelley MJ, Dunstan CR, Burgess T et al (1998) Osteoprotegerin ligand is a cytokine that regulates osteoclast differentiation and activation. Cell 93(2):165–176
Kitazawa S, Kitazawa R (2002) RANK ligand is a prerequisite for cancer-associated osteolytic lesions. J Pathol 198(2):228–236
Tanaka S, Nakamura K, Takahasi N, Suda T (2005) Role of RANKL in physiological and pathological bone resorption and therapeutics targeting the RANKL-RANK signaling system. Immunol Rev 208:30–49
Roodman GD, Dougall WC (2008) RANK ligand as a therapeutic target for bone metastases and multiple myeloma. Cancer Treat Rev 34(1):92–101
Zhang J, Dai J, Qi Y, Lin DL, Smith P, Strayhorn C et al (2001) Osteoprotegerin inhibits prostate cancer-induced osteoclastogenesis and prevents prostate tumor growth in the bone. J Clin Invest 107(10):1235–1244
Zhang J, Dai J, Yao Z, Lu Y, Dougall W, Keller ET (2003) Soluble receptor activator of nuclear factor kappaB Fc diminishes prostate cancer progression in bone. Cancer Res 63(22):7883–7890
Morony S, Capparelli C, Sarosi I, Lacey DL, Dunstan CR, Kostenuik PJ (2001) Osteoprotegerin inhibits osteolysis and decreases skeletal tumor burden in syngeneic and nude mouse models of experimental bone metastasis. Cancer Res 61(11):4432–4436
Wilson TJ, Nannuru KC, Futakuchi M, Sadanandam A, Singh RK (2008) Cathepsin G enhances mammary tumor-induced osteolysis by generating soluble receptor activator of nuclear factor-{kappa}B ligand. Cancer Res 68(14):5803–5811
Wilson TJ, Singh RK (2008) Proteases as modulators of tumor–stromal interaction: primary tumors to bone metastases. Biochimica et Biophysica Acta (BBA) Rev Cancer 1785(2):85–95
Lynch CC, Hikosaka A, Acuff HB, Martin MD, Kawai N, Singh RK et al (2005) MMP-7 promotes prostate cancer-induced osteolysis via the solubilization of RANKL. Cancer Cell 7(5):485–496
Chesneau V, Becherer JD, Zheng Y, Erdjument-Bromage H, Tempst P, Blobel CP (2003) Catalytic properties of ADAM19. J Biol Chem 278(25):22331–22340
Schlondorff J, Lum L, Blobel CP (2001) Biochemical and pharmacological criteria define two shedding activities for TRANCE/OPGL that are distinct from the tumor necrosis factor alpha convertase. J Biol Chem 276(18):14665–14674
Aslakson CJ, Miller FR (1992) Selective events in the metastatic process defined by analysis of the sequential dissemination of subpopulations of a mouse mammary tumor. Cancer Res 52(6):1399–1405
Chen Z, Varney ML, Backora MW, Cowan K, Solheim JC, Talmadge JE et al (2005) Down-regulation of vascular endothelial cell growth factor-C expression using small interfering RNA vectors in mammary tumors inhibits tumor lymphangiogenesis and spontaneous metastasis and enhances survival. Cancer Res 65(19):9004–9011
Heppner GH, Miller FR, Shekhar PM (2000) Nontransgenic models of breast cancer. Breast Cancer Res 2(5):331–334
Huang X, Wong MK, Yi H, Watkins S, Laird AD, Wolf SF et al (2002) Combined therapy of local and metastatic 4T1 breast tumor in mice using SU6668, an inhibitor of angiogenic receptor tyrosine kinases, and the immunostimulator B7.2-IgG fusion protein. Cancer Res 62(20):5727–5735
Lelekakis M, Moseley JM, Martin TJ, Hards D, Williams E, Ho P et al (1999) A novel orthotopic model of breast cancer metastasis to bone. Clin Exp Metastasis 17(2):163–170
Murphy BO, Joshi S, Kessinger A, Reed E, Sharp JG (2002) A murine model of bone marrow micrometastasis in breast cancer. Clin Exp Metastasis 19(7):561–569
Sloan EK, Stanley KL, Anderson RL (2004) Caveolin-1 inhibits breast cancer growth and metastasis. Oncogene 23(47):7893–7897
Bennett CF, Cowsert LM (1999) Antisense oligonucleotides as a tool for gene functionalization and target validation. Biochim Biophys Acta 1489(1):19–30
Henry SP, Geary RS, Yu R, Levin AA (2001) Drug properties of second-generation antisense oligonucleotides: how do they measure up to their predecessors? Curr Opin Invest Drugs 2(10):1444–1449
Kingsley LA, Fournier PGJ, Chirgwin JM, Guise TA (2007) Molecular biology of bone metastasis. Mol Cancer Ther 6(10):2609–2617
Siclari VA, Guise TA, Chirgwin JM (2006) Molecular interactions between breast cancer cells and the bone microenvironment drive skeletal metastases. Cancer Metastasis Rev 25(4):621–633
Blair JM, Zhou H, Seibel MJ, Dunstan CR (2006) Mechanisms of disease: roles of OPG, RANKL and RANK in the pathophysiology of skeletal metastasis. Nat Clin Pract Oncol 3(1):41–49
Kuperwasser C, Dessain S, Bierbaum BE, Garnet D, Sperandio K, Gauvin GP et al (2005) A mouse model of human breast cancer metastasis to human bone. Cancer Res 65(14):6130–6138
Reddi AH, Roodman D, Freeman C, Mohla S (2003) Mechanisms of tumor metastasis to the bone: challenges and opportunities. J Bone Miner Res 18(2):190–194
Varney ML, Singh S, Backora M, Chen Z, Singh RK (2008) Lymphangiogenesis and anti-tumor immune responses. Curr Mol Med (In Press)
Grimaud E, Soubigou L, Couillaud S, Coipeau P, Moreau A, Passuti N et al (2003) Receptor activator of nuclear factor kappaB ligand (RANKL)/osteoprotegerin (OPG) ratio is increased in severe osteolysis. Am J Pathol 163(5):2021–2031
Jung K, Stephan C, Semjonow A, Lein M, Schnorr D, Loening SA (2003) Serum osteoprotegerin and receptor activator of nuclear factor-kappa B ligand as indicators of disturbed osteoclastogenesis in patients with prostate cancer. J Urol 170(6 Pt 1):2302–2305
Seidel C, Hjertner O, Abildgaard N, Heickendorff L, Hjorth M, Westin J et al (2001) Serum osteoprotegerin levels are reduced in patients with multiple myeloma with lytic bone disease. Blood 98(7):2269–2271
Terpos E, Szydlo R, Apperley JF, Hatjiharissi E, Politou M, Meletis J et al (2003) Soluble receptor activator of nuclear factor kappaB ligand-osteoprotegerin ratio predicts survival in multiple myeloma: proposal for a novel prognostic index. Blood 102(3):1064–1069
Kostenuik PJ, Singh G, Suyama KL, Orr FW (1992) Stimulation of bone resorption results in a selective increase in the growth rate of spontaneously metastatic Walker 256 cancer cells in bone. Clin Exp Metastasis 10(6):411–418
Schneider A, Kalikin LM, Mattos AC, Keller ET, Allen MJ, Pienta KJ et al (2005) Bone turnover mediates preferential localization of prostate cancer in the skeleton. Endocrinology 146(4):1727–1736
Brown JE, Cook RJ, Major P, Lipton A, Saad F, Smith M et al (2005) Bone turnover markers as predictors of skeletal complications in prostate cancer, lung cancer, and other solid tumors. JNCI J Natl Cancer Inst 97(1):59–69
Costa L, Demers LM, Gouveia-Oliveira A, Schaller J, Costa EB, de Moura MC et al (2002) Prospective evaluation of the peptide-bound collagen type I cross-links N-telopeptide and C-telopeptide in predicting bone metastases status. J Clin Oncol 20(3):850–856
Sasaki A, Boyce BF, Story B, Wright KR, Chapman M, Boyce R et al (1995) Bisphosphonate risedronate reduces metastatic human breast cancer burden in bone in nude mice. Cancer Res 55(16):3551–3557
van der Pluijm G, Que I, Sijmons B, Buijs JT, Lowik CWGM, Wetterwald A et al (2005) Interference with the microenvironmental support impairs the de novo formation of bone metastases in vivo. Cancer Res 65(17):7682–7690
Buijs JT, Que I, Lowik CW, Papapoulos SE, Van der Pluijm G (2009) Inhibition of bone resorption and growth of breast cancer in the bone microenvironment. Bone 44(2):380–386
Phadke PA, Mercer RR, Harms JF, Jia Y, Frost AR, Jewell JL et al (2006) Kinetics of metastatic breast cancer cell trafficking in bone. Clin Cancer Res 12(5):1431–1440
Michaelson MD, Smith MR (2005) Bisphosphonates for treatment and prevention of bone metastases. J Clin Oncol 23(32):8219–8224
Saarto T, Vehmanen L, Virkkunen P, Blomqvist C (2004) Ten-year follow-up of a randomized controlled trial of adjuvant clodronate treatment in node-positive breast cancer patients. Acta Oncol 43(7):650–656
Acknowledgments
This work was supported in part by grants CA72781 (R.K.S.) and Cancer Center Support Grant (P30CA036727) from National Cancer Institute, National Institutes of Health, Susan G. Komen for the Cure grant KG090860 and Nebraska Research Initiative Program in Cancer Glycobiology (R.K.S.), Department of Defense Breast Cancer Research Program Predoctoral traineeship award (K.C.N) and the Howard Hughes Medical Institute Research Training Fellowship (T.J.W.). We thank Dr. James Eudy and the UNMC DNA Microarray Core Facility for help in microarray analysis.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Nannuru, K.C., Futakuchi, M., Sadanandam, A. et al. Enhanced expression and shedding of receptor activator of NF-κB ligand during tumor–bone interaction potentiates mammary tumor-induced osteolysis. Clin Exp Metastasis 26, 797–808 (2009). https://doi.org/10.1007/s10585-009-9279-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10585-009-9279-2