Abstract
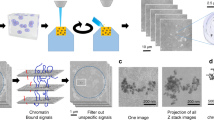
The nuclear architecture is considered an important contributor to genome function. Although the fine structural features of the cell nucleus have been investigated extensively by means of ultrastructural cytochemistry, mainly on ultrathin sections in two dimensions (2D), there was a of lack routine methods for a rapid reconstruction of three-dimensional (3D) distribution of different structural constituents throughout the nuclear volume. We have now filled this gap by the application of a novel approach associating a pre-embedding selective visualization of nuclear components with a method making use of ultramicrotomy combined with scanning electron microscopy (microtome serial block face scanning electron microscopy—‘3View’). We have been able to apply this method to the study of DNA distribution within the nuclear volume and reconstruction of 3D chromatin arrangement in nuclei of rat hepatocytes and endothelial cells. Our observations demonstrate that while chromatin appears to occupy the interior of nuclei rather sparsely on 2D images, once reconstructed in 3D from a series of sequential 2D images it gives the impression of considerably filling the nuclear volume. However, quantitative evaluation of the nuclear volume occupied by DNA in the above two types of nuclei leaves a significant part to the interchromatin space (66.2% for hepatic cells and 41.7% for endothelial cells, including nuclear space occupied by nucleoli). Detailed analysis of the reconstructed nuclei reveals a high degree of superposition of chromatin domains, giving rise to a false impression that they fill a much larger part of the nuclear volume than they really do. Our results show the importance of the contribution of such reconstruction techniques to our understanding of the nuclear architecture.





Similar content being viewed by others
Abbreviations
- 2D:
-
two-dimensional
- 3D:
-
three-dimensional
- 3D EM:
-
three-dimensional electron microscopy
- CT:
-
chromosome territory
- DNA-FISH:
-
DNA-fluorescent in-situ hybridization
- IC:
-
interchromatin space/compartment
- PAS:
-
periodic acid-Schiff
- SBFSEM:
-
serial block face scanning electron microscopy
- SEM:
-
scanning electron microscopy
- TEM:
-
transmission electron microscopy
References
Albiez H, Cremer M, Tiberi C et al (2006) Chromatin domains and the interchromatin compartment form structurally defined and functionally interacting nuclear networks. Chromosome Res 14:707–733
Biggiogera M, Fakan S (2008) Visualization of nuclear organization by ultrastructural cytochemistry. In: Allen T (ed) Methods in cell biology. Elsevier, New York, pp. 433–449
Branco MR, Pombo A (2006) Intermingling of chromosome territories in interphase suggests role in translocations and transcription-dependent associations. PLoS Biol 4:e138
Cmarko D, Verschure PJ, Martin TE et al (1999) Ultrastructural analysis of transcription and splicing in the cell nucleus after bromo-UTP microinjection. Mol Biol Cell 10:211–223
Cremer T, Cremer M, Dietzel S, Muller S, Solovei I, Fakan S (2006) Chromosome territories—a functional nuclear landscape. Curr Opin Cell Biol 18:307–316
Dechat T, Pfleghaar K, Sengupta K et al (2008) Nuclear lamins: major factors in the structural organization and function of the nucleus and chromatin. Genes Dev 22:832–853
Denk W, Horstmann H (2004) Serial block-face scanning electron microscopy to reconstruct three-dimensional tissue nanostructure. PLoS Biol 2:e329
Derenzini M (1979) Fine structure of chromatin as visualized in thin sections with the Gautier selective stain for DNA. J Ultrastruct Res 69:239–248
Dupuy-Coin AM, Moens P, Bouteille M (1986) Three-dimensional analysis of given cell structures: nucleolus, nucleoskeleton and nuclear inclusions. Methods Achiev Exp Pathol 12:1–25
Esquivel C, Rovira P, Echeverria O, Vazquez-Nin GH (1987) A simple staining method for chromatin in electron microscopy compatible with serial sectioning. Ultramicroscopy 21:103–109
Esquivel C, Vazquez-Nin GH, Echeverria O (1989) Evidence of repetitive patterns of chromatin distribution in cell nuclei of rat liver. Acta Anat (Basel) 136:94–98
Fakan S (2004) Ultrastructural cytochemical analyses of nuclear functional architecture. Eur J Histochem 48:5–14
Fakan S, Van Driel R (2007) The perichromatin region: a functional compartment in the nucleus that determines large-scale chromatin folding. Semin Cell Dev Biol 18:676–681
Hozak P, Novak JT, Smetana K (1989) Three-dimensional reconstructions of nucleolus-organizing regions in PHA-stimulated human lymphocytes. Biol Cell 66:225–33
Knott G, Marchman H, Wall D, Lich B (2008) Serial section scanning electron microscopy of adult brain tissue using focused ion beam milling. J Neurosci 28:2959–2964
Kostova NN, Srebreva L, Markov DV, Rundquist I (2004) Histone H1 and chromatin interactions in human fibroblast nuclei after H1 depletion and reconstitution with H1 subfractions. Cytometry A 58:132–139
Lanctot C, Cheutin T, Cremer M, Cavalli G, Cremer T (2007) Dynamic genome architecture in the nuclear space: regulation of gene expression in three dimensions. Nat Rev Genet 8:104–115
Liu DF, El-Alfy M, Leblond CP (1992) Visualization of DNA within mitochondria by osmium-ammine staining of mouse duodenal crypt cells. J Cell Sci 101(Pt 4):785–793
Lopez-Velazquez G, Marquez J, Ubaldo E, Corkidi G, Echeverria O, Vazquez Nin GH (1996) Three-dimensional analysis of the arrangement of compact chromatin in the nucleus of G0 rat lymphocytes. Histochem Cell Biol 105:153–161
Meaburn KJ, Misteli T (2007) Cell biology: chromosome territories. Nature 445:379–381
Mikhaylova VT, Markov DV (1994) An alternative method for preparation of Schiff-like reagent from osmium-ammine complex for selective staining of DNA on thin Lowicryl sections. J Histochem Cytochem 42:1643–1649
Misteli T (2007) Beyond the sequence: cellular organization of genome function. Cell 128:787–800
Nash RE, Puvion E, Bernhard W (1975) Perichromatin fibrils as components of rapidly labeled extranucleolar RNA. J Ultrastruct Res 53:395–405
Solovei I, Cavallo A, Schermelleh L et al (2002) Spatial preservation of nuclear chromatin architecture during three-dimensional fluorescence in-situ hybridization (3D-FISH). Exp Cell Res 276:10–23
Spector DL (2003) The dynamics of chromosome organization and gene regulation. Annu Rev Biochem 72:573–608
Stelzer EHK (1995) The intermediate optical system of laser-scanning confocal microscopes. In: Pawley JB (ed) Handbook of biological confocal microscopy. Plenum Press, New York, pp. 139–154
Taddei A (2007) Active genes at the nuclear pore complex. Curr Opin Cell Biol 19:305–310
Taddei A, Hediger F, Neumann FR, Gasser SM (2004) The function of nuclear architecture: a genetic approach. Annu Rev Genet 38:305–345
Testillano PS, Sanchez-Pina MA, Olmedilla A, Ollacarizqueta MA, Tandler CJ, Risueno MC (1991) A specific ultrastructural method to reveal DNA: the NAMA-Ur. J Histochem Cytochem 39:1427–1438
Vazquez-Nin GH, Biggiogera M, Echeverria OM (1995) Activation of osmium ammine by SO2-generating chemicals for EM Feulgen-type staining of DNA. Eur J Histochem 39:101–106
Visser AE, Jaunin F, Fakan S, Aten JA (2000) High resolution analysis of interphase chromosome domains. J Cell Sci 113:2585–2593
Von Schack ML, Fakan S (1994) Retention of glycogen in cryosubstituted mouse liver. Histochem Cell Biol 102:451–455
Zankel A, Kraus B, Poelt P, Schaffer M, Ingolic E (2009) Ultramicrotomy in the ESEM, a versatile method for materials and life sciences. J Microsc 233:140–148
Acknowledgements
We thank Ernestina Ubaldo for her remarkable technical skill, Jitka Fakan for expert technical assistance with the initial experiments, and Patrick Schwarb for his help with image analysis. We are indebted to Drs Boris Joffe and Irina Solovei for critical reading of the manuscript and helpful suggestions and to Professor Dr Gerhard Wanner for stimulating discussions. This work was supported by the Deutsche Forschungsgemeinschaft, the Munich Center of Integrated Protein Science (CIPSM), the LMU Bioimaging Network (BIN), and PAPIIT IN203308 and Conacyt 81213 (Mexico). Transmission electron microscopy at the LMU was supported by SFB 646 (Roland Beckmann).
Author information
Authors and Affiliations
Corresponding author
Additional information
Responsible Editor: Hans-Joachim Lipps.
Electronic Supplementary Material
Below is the link to the electronic supplementary material.
Supplementary Movie S1
A complete stack of images reconstructed from one hepatocyte nucleus (MOV 6.46 mb)
Supplementary Movie S2
A model reconstruction of the median portion (2.8 × 9.2 × 8.2 μm) of an image stack of a hepatocyte nucleus. The observation from different angles gives the possibility to better appreciate the real extent of the interchromatin space occurring between chromatin domains (green). (MOV 6.78 mb)
Rights and permissions
About this article
Cite this article
Rouquette, J., Genoud, C., Vazquez-Nin, G.H. et al. Revealing the high-resolution three-dimensional network of chromatin and interchromatin space: A novel electron-microscopic approach to reconstructing nuclear architecture. Chromosome Res 17, 801–810 (2009). https://doi.org/10.1007/s10577-009-9070-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10577-009-9070-x