Abstract
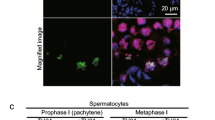
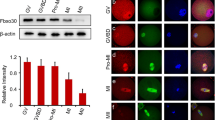
During meiosis in male mammals, X and Y chromosomes undergo the process of meiotic sex chromosome inactivation (MSCI). A crucial role in MSCI has recently been reported for BRCA1, ATR kinase, and phosphorylated histone H2AX, but the exact mechanism remains to be determined. Small ubiquitin-like modifier (SUMO) proteins have recently been shown to localize to the sex body in mouse meiotic spermatocytes, but the role they play during MSCI is unknown. In this study, in order to better understand the molecular events of MSCI, we followed dynamic changes in γH2AX and SUMO localization patterns during MSCI. Using confocal laser scanning microscopy (CLSM) as an analytical tool for visualizing numerous spermatocytes from the same development stage and for consecutively following the meiotic progression, we were able to demonstrate a very early appearance of SUMO-1, which preceded γH2AX accumulation on the sex chromosomes during their meiotic inactivation. In contrast to SUMO-1, SUMO-2/3 was undetectable in zygotene spermatocytes, suggesting a possible specific role for SUMO-1 in the initiation of MSCI.



Similar content being viewed by others
Abbreviations
- ATR:
-
ATM and Rad3-related
- BRCA1:
-
breast cancer 1, early onset
- BSA:
-
bovine serum albumin
- CLSM:
-
confocal laser scanning microscopy
- CY:
-
cyanine
- DSB:
-
double strand breaks
- E sp:
-
elongating spermatids
- FITC:
-
fluorescein isothiocyanate
- γH2AX:
-
phosphorylated histone H2AX
- MSCI:
-
meiotic sex chromosome inactivation
- MSUC:
-
meiotic silencing of unsynapsed chromatin
- PAR:
-
pseudoautosomal region
- PBS:
-
phosphate-buffered saline
- PI:
-
propidium iodide
- PL:
-
preleptotene spermatocytes
- Le:
-
leptotene spermatocytes
- Meio:
-
meiotic figures
- PS:
-
pachytene spermatocytes
- RS:
-
round spermatids
- Se:
-
Sertoli cells
- Sg:
-
spermatogonia
- SC:
-
synaptonemal complex
- SUMO:
-
small ubiquitin-like modifier(s)
- Zyg:
-
zygotene spermatocytes
References
Ayaydin F, Dasso M (2004) Distinct in vivo dynamics of vertebrate SUMO paralogues. Mol Biol Cell 15:5208–5218
Baarends WM, Wassenaar E, van der Laan R et al (2005) Silencing of unpaired chromatin and histone H2A ubiquitination in mammalian meiosis. Mol Cell Biol 25(3):1041–1053
Bellani MA, Romanienko PJ, Cairatti DA, Camerini-Otero RD (2005) SPO11 is required for sex-body formation, and Spo11 heterozygosity rescues the prophase arrest of Atm−/− spermatocytes. J Cell Sci 118:3233–3245
Bohren KM, Nadkarni V, Song JH, Gabbay KH, Owerbach D (2004) M55V polymorphism in a novel SUMO gene (SUMO-4) differentially activates heat shock transcription factors and is associated with susceptibility to type I diabetes mellitus. J Biol Chem 279(26):27233–27238
Cheng CH, Lo YH, Liang SS et al (2006) SUMO modifications control assembly of synaptonemal complex and polycomplex in meiosis of Saccharomyces cerevisiae. Genes Dev 20(15):2067–2081
Cohen PE, Pollack SE, Pollard JW (2006) Genetic analysis of chromosome pairing, recombination, and cell cycle control during first meiotic prophase in mammals. Endocr Rev 27(4):398–426
Fernandez-Capetillo O, Mahadevaiah SK, Celeste A et al (2003) H2AX is required for chromatin remodeling and inactivation of sex chromosomes in male mouse meiosis. Dev Cell 4:497–508
Gill G (2004) SUMO and ubiquitin in the nucleus: different functions, similar mechanisms? Genes Dev 18:2046–2059
Handel MA (2004) The XY body: a specialized meiotic chromatin domain. Exp Cell Res 296:57–63
Hoyer-Fender S (2003) Molecular aspects of XY body formation. Cytogenet Genome Res 103:245–255
Johnson ES (2004) Protein modification by SUMO. Annu Rev Biochem 73:355–382
La Salle S, Sun F, Zhang XD, Matunis MJ, Handel MA (2008) Developmental control of sumoylation pathway proteins in mouse male germ cells. Dev Biol 321(1):227–237
Mahadevaiah SK et al (2001) Recombinational DNA double-strand breaks in mice precede synapsis. Nat Genet 27:271–276
Mahadevaiah SK, Bourc'his D, de Rooij DG, Bestor TH, Turner JM, Burgoyne PS (2008) Extensive meiotic asynapsis in mice antagonises meiotic silencing of unsynapsed chromatin and consequently disrupts meiotic sex chromosome inactivation. J Cell Biol 182(2):263–276
Martin RH (2008) Meiotic errors in human oogenesis and spermatogenesis. Reprod Biomed Online 16(4):523–531
Muller S, Ledl A, Schmidt D (2004) SUMO: a regulator of gene expression and genome integrity. Oncogene 23:1998–2008
Russell L, Ettlin R, Sinha Hikim A, Clegg E (eds) (1990) Histological and Histopathological evaluation of the testis. Cache River, Clearwater, FL
Sacher M, Pfander B, Hoege C, Jentsch S (2006) Control of Rad52 recombination activity by double-strand break-induced SUMO modification. Nat Cell Biol 11:1284–1290
Schwartz DC, Hochstrasser M (2003) A superfamily of protein tags: ubiquitin, SUMO and related modifiers. Trends Biochem Sci 28:321–328
Seeler JS, Dejean A (2003) Nuclear and unclear functions of SUMO. Nat Rev Mol Cell Biol 4:690–699
Turner JM (2007) Meiotic sex chromosome inactivation. Development 134(10):1823–1831
Turner JM, Aprelikova O, Xu X et al (2004) BRCA1, histone H2AX phosphorylation and male meiotic sex chromosome inactivation. Curr Biol 14:2135–2142
Turner JM, Mahadevaiah SK, Fernandez-Capetillo O et al (2005) Silencing of unsynapsed meiotic chromosomes in the mouse. Nat Genet 37(1):41–47
Vertegaal AC, Ogg SC, Jaffray E et al (2004) A proteomic study of SUMO-2 target proteins. J Biol Chem 279(32):33791–33798
Vigodner M, Morris PL (2005) Testicular expression of small ubiquitin-related modifier-1 (SUMO-1) supports multiple roles in spermatogenesis: silencing of sex chromosomes in spermatocytes, spermatid microtubule nucleation, and nuclear reshaping. Dev Biol 282:480–492
Vigodner M, Lewin LM, Glaser T, Shochat L, Mittelman L, Golan R (2002) Use of confocal microscopy for the study of spermatogenesis. Methods Cell Sci 24(4):169–180
Acknowledgments
The author thanks Stern College for Women, Yeshiva University and Joseph Alexander Foundation for supporting this research project; Dr. Alison North and other staff of The Rockefeller University Bio-Imaging Resource Center kindly provided help with the use of the facility’s equipment.
Author information
Authors and Affiliations
Corresponding author
Additional information
Responsible Editor: Herbert McGregor.
Rights and permissions
About this article
Cite this article
Vigodner, M. Sumoylation precedes accumulation of phosphorylated H2AX on sex chromosomes during their meiotic inactivation. Chromosome Res 17, 37–45 (2009). https://doi.org/10.1007/s10577-008-9006-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10577-008-9006-x