Abstract
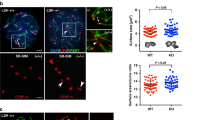
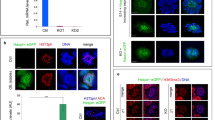
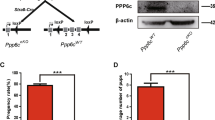
Histone phosphorylation is sometimes associated with mitosis and meiosis. We have recently identified a phosphorylation of the 127th threonine on TH2A (pTH2A), a germ cell-specific H2A variant, in condensed spermatids and mitotic early preimplantation embryos of mice. Here, we further report the existence of pTH2A at the centromeres in metaphase I spermatocytes and oocytes. Moreover, we identified Haspin, a known kinase for the 3rd threonine on H3, is responsible for pTH2A in vivo. In contrast to the severe meiotic defect in oocytes treated with a Haspin inhibitor, pTH2A-deficient mice, in which the 127th threonine was replaced by alanine, maintained the fertility and exhibited no obvious defect in both oocytes and spermatogenesis. Interestingly, pTH2A was significantly decreased in aged oocytes, suggesting that its accumulation is regulated by centromeric cohesins. Collectively, our study proposes a new set of kinase-histone pair at meiotic centromere, which is highly coordinated during meiosis.






Similar content being viewed by others
References
Balboula AZ, Schindler K (2014) Selective disruption of aurora C kinase reveals distinct functions from aurora b kinase during meiosis in mouse oocytes. Plos Genet 10:e1004194. doi:10.1371/journal.pgen.1004194
Burkhardt S et al (2016) Chromosome cohesion established by Rec8-Cohesin in fetal oocytes is maintained without detectable turnover in oocytes arrested for months in mice. Curr Biol 26:678–685. doi:10.1016/j.cub.2015.12.073
Carretero M, Ruiz-Torres M, Rodriguez-Corsino M, Barthelemy I, Losada A (2013) Pds5B is required for cohesion establishment and aurora B accumulation at centromeres. EMBO J 32:2938–2949. doi:10.1038/emboj.2013.230
Chiang T, Duncan FE, Schindler K, Schultz RM, Lampson MA (2010) Evidence that weakened centromere cohesion is a leading cause of age-related aneuploidy in oocytes. Curr Biol 20:1522–1528. doi:10.1016/j.cub.2010.06.069
Choi YC, Chae CB (1993) Demethylation of somatic and testis-specific histone H2A and H2B genes in F9 embryonal carcinoma cells. Mol Cell Biol 13:5538–5548
Dai J, Sultan S, Taylor SS, Higgins JMG (2005) The kinase haspin is required for mitotic histone H3 Thr 3 phosphorylation and normal metaphase chromosome alignment. Genes Dev 19:472–488. doi:10.1101/gad.1267105
Eswaran J et al (2009) Structure and functional characterization of the atypical human kinase haspin. Proc Natl Acad Sci USA 106:20198–20203. doi:10.1073/pnas.0901989106
Ghenoiu C, Wheelock MS, Funabiki H (2013) Autoinhibition and polo-dependent multisite phosphorylation restrict activity of the histone H3 kinase Haspin to mitosis. Mol Cell 52:734–745. doi:10.1016/j.molcel.2013.10.002
Goto Y, Yamagishi Y, Shintomi-Kawamura M, Abe M, Tanno Y, Watanabe Y (2017) Pds5 regulates sister-chromatid cohesion and chromosome bi-orientation through a conserved protein interaction module. Curr Biol 27:1005–1012. doi:10.1016/j.cub.2017.02.066
Hada M, Masuda K, Yamaguchi K, Shirahige K, Okada Y (2017) Identification of a variant-specific phosphorylation of TH2A during spermiogenesis. Sci Rep 7:46228. doi:10.1038/srep46228
Higgins JMG (2001a) Haspin-like proteins: a new family of evolutionarily conserved putative eukaryotic protein kinases. Protein Sci 10:1677–1684. doi:10.1110/ps.49901
Higgins JMG (2001b) The Haspin gene: location in an intron of the integrin alpha E gene, associated transcription of an integrin alpha E-derived RNA and expression in diploid as well as haploid cells. Gene 267:55–69. doi:10.1016/s0378-1119(01)00387-0
Huynh LM, Shinagawa T, Ishii S (2016) Two histone variants TH2A and TH2B enhance human induced pluripotent stem cell generation. Stem Cells Dev 25:251–258. doi:10.1089/scd.2015.0264
Ishizuka Y et al (2013) The influence of reduced glutathione in fertilization medium on the fertility of in vitro-matured C57BL/6 mouse oocytes. Theriogenology 80:421–426. doi:10.1016/j.theriogenology.2013.07.002
Kawashima SA, Yamagishi Y, Honda T, Ishiguro K-I, Watanabe Y (2010) Phosphorylation of H2A by Bub1 prevents chromosomal instability through localizing Shugoshin. Science 327:172–177. doi:10.1126/science.1180189
Kelly AE, Ghenoiu C, Xue JZ, Zierhut C, Kimura H, Funabiki H (2010) Survivin reads phosphorylated histone H3 threonine 3 to activate the mitotic kinase aurora B. Science 330:235–239. doi:10.1126/science.1189505
Kim J et al (2015) Meikin is a conserved regulator of meiosis-I-specific kinetochore function. Nature 517:466–471. doi:10.1038/nature14097
Kimmins S et al (2007) Differential functions of the aurora-B and aurora-C kinases in mammalian spermatogenesis. Mol Endocrinol 21:726–739. doi:10.1210/me.2006-0332
Kitajima TS, Ohsugi M, Ellenberg J (2011) Complete kinetochore tracking reveals error-prone homologous chromosome biorientation in mammalian oocytes. Cell 146:568–581. doi:10.1016/j.cell.2011.07.031
Liu H, Qu QH, Warrington R, Rice A, Cheng NY, Yu HT (2015) Mitotic transcription installs Sgo1 at centromeres to coordinate chromosome segregation. Mol Cell 59:426–436. doi:10.1016/j.molcel.2015.06.018
Maiolica A et al (2014) Modulation of the chromatin phosphoproteome by the haspin protein kinase. Mol Cell Proteomics 13:1724–1740. doi:10.1074/mcp.M113.034819
Markaki Y, Christogianni A, Politou AS, Georgatos SD (2009) Phosphorylation of histone H3 at Thr3 is part of a combinatorial pattern that marks and configures mitotic chromatin. Journal of Cell Science 122:2809–2819. doi:10.1242/jcs.043810
Montellier E et al (2013) Chromatin-to-nucleoprotamine transition is controlled by the histone H2B variant TH2B. Genes Dev 27:1680–1692. doi:10.1101/gad.220095.113
Nguyen AL, Gentilello AS, Balboula AZ, Shrivastava V, Ohring J, Schindler K (2014) Phosphorylation of threonine 3 on histone H3 by haspin kinase is required for meiosis I in mouse oocytes. J Cell Sci 127:5066–5078. doi:10.1242/jcs.158840
Padavattan S, Shinagawa T, Hasegawa K, Kumasaka T, Ishii S, Kumarevel T (2015) Structural and functional analyses of nucleosome complexes with mouse histone variants TH2a and TH2b, involved in reprogramming. Biochem Biophys Res Commun 464:929–935. doi:10.1016/j.bbrc.2015.07.070
Revenkova E, Herrmann K, Adelfalk C, Jessberger R (2010) Oocyte cohesin expression restricted to predictyate stages provides full fertility and prevents aneuploidy. Curr Biol 20:1529–1533. doi:10.1016/j.cub.2010.08.024
Sakakibara Y, Hashimoto S, Nakaoka Y, Kouznetsova A, Hoog C, Kitajima TS (2015) Bivalent separation into univalents precedes age-related meiosis I errors in oocytes. Nat Commun 6:7550. doi:10.1038/ncomms8550
Shibuya H, Ishiguro K-I, Watanabe Y (2014) The TRF1-binding protein TERB1 promotes chromosome movement and telomere rigidity in meiosis. Nat Cell Biol 16:145–156. doi:10.1038/ncb2896
Shimada M et al (2016) Essential role of autoactivation circuitry on aurora B-mediated H2AX-pS121 in mitosis. Nat Commun 7:11. doi:10.1038/ncomms12059
Shinagawa T et al (2014) Histone variants enriched in oocytes enhance reprogramming to induced pluripotent stem cells. Cell Stem Cell 14:217–227. doi:10.1016/j.stem.2013.12.015
Shinagawa T et al (2015) Disruption of Th2a and Th2b genes causes defects in spermatogenesis. Development 142:1287–1292. doi:10.1242/dev.121830
Shires A, Carpenter MP, Chalkley R (1976) Cysteine-containing H2B-like histone found in mature mammalian testis. J Biol Chem 251:4155–4158
Tanaka H et al (1999) Identification and characterization of a haploid germ cell-specific nuclear protein kinase (Haspin) in spermatid nuclei and its effects on somatic cells. J Biol Chem 274:17049–17057. doi:10.1074/jbc.274.24.17049
Trostleweige PK, Meistrich ML, Brock WA, Nishioka K, Bremer JW (1982) Isolation and characterization of TH2A, a germ cell-specific variant of histone-2A in rat testis. J Biol Chem 257:5560–5567
Tseng TC, Chen SH, Hsu YPP, Tang TK (1998) Protein kinase profile of sperm and eggs: cloning and characterization of two novel testis-specific protein kinases (AIE1, AIE2) related to yeast and fly chromosome segregation regulators. DNA Cell Biol 17:823–833. doi:10.1089/dna.1998.17.823
Tsukahara T, Tanno Y, Watanabe Y (2010) Phosphorylation of the CPC by Cdk1 promotes chromosome bi-orientation. Nature 467:719–U111. doi:10.1038/nature09390
Wang F et al (2010) Histone H3 Thr-3 phosphorylation by Haspin positions aurora B at centromeres in mitosis. Science 330:231–235. doi:10.1126/science.1189435
Wang FW, Higgins JMG (2013) Histone modifications and mitosis: countermarks, landmarks, and bookmarks. Trends Cell Biol 23:175–184. doi:10.1016/j.tcb.2012.11.005
Wang FW, Ulyanova NP, van der Waal MS, Patnaik D, Lens SMA, Higgins JMG (2011) A positive feedback loop involving haspin and aurora B promotes CPC accumulation at centromeres in mitosis. Curr Biol 21:1061–1069. doi:10.1016/j.cub.2011.05.016
Wang Q et al (2016) H3 Thr3 phosphorylation is crucial for meiotic resumption and anaphase onset in oocyte meiosis. Cell Cycle 15:213–224. doi:10.1080/15384101.2015.1121330
Yamagata K, Yamazaki T, Yamashita M, Hara Y, Ogonuki N, Ogura A (2005) Noninvasive visualization of molecular events in the mammalian zygote. Genesis 43:71–79. doi:10.1002/gene.20158
Yamagishi Y, Honda T, Tanno Y, Watanabe Y (2010) Two histone marks establish the inner centromere and chromosome Bi-orientation. Science 330:239–243. doi:10.1126/science.1194498
Yoshida S, Kaido M, Kitajima TS (2015) Inherent instability of correct kinetochore-microtubule attachments during meiosis I in oocytes. Dev Cell 33:589–602. doi:10.1016/j.devcel.2015.04.020
Yun Y, Holt JE, Lane SIR, McLaughlin EA, Merriman JA, Jones KT (2014) Reduced ability to recover from spindle disruption and loss of kinetochore spindle assembly checkpoint proteins in oocytes from aged mice. Cell Cycle 13:1938–1947. doi:10.4161/cc.28897
Zhou L et al (2017) The N-terminal non-kinase-domain-mediated binding of Haspin to Pds5B protects centromeric cohesion in mitosis. Curr Biol 27:992–1004. doi:10.1016/j.cub.2017.02.019
Zhou LL, Tian XY, Zhu CL, Wang FW, Higgins JMG (2014) Polo-like kinase-1 triggers histone phosphorylation by Haspin in mitosis. EMBO Rep 15:273–281. doi:10.1002/embr.201338080
Acknowledgments
We would like to give a special thanks to the members of MBL antibody production team for generating anti-pTH2A antibody. We also thank Dr. Y. Makino and Mr. I. Tsuchiya from the laboratory of Pathology, Drs. H. Shibuya, Y. Tanno, and T. Sakuno from the laboratory of Chromosome Dynamics at the Institute of Molecular and Cellular Biosciences, the University of Tokyo, Drs. Tokoro and Yamagata from Kinki University, and Dr. J Lee from Kobe University for helpful advice and technical assistance, Drs. T. Shinagawa and S. Ishii from the RIKEN Institute for kindly providing the anti-TH2A antibody, and Drs. H. Tanaka and H. Kurumizaka from Waseda University for kindly providing the recombinant TH2A.
Author information
Authors and Affiliations
Contributions
M.H. and Y.O. designed the study, and M.H., J. K., E. I., Y. F., and Y. O. performed experiments. M.H. and Y.O. wrote the paper with helpful discussion from H. T. and Y.W.
Corresponding author
Ethics declarations
Compliance with ethical standards
All experimental procedures involving mice were approved by the Animal Experiment Ethics Committees at the Institute of Molecular and Cellular Biosciences, The University of Tokyo (Experiment# 2611, 2710, 2808), and the experiments were performed precisely in accordance with the manual provided by the Life Science Research Ethics and Safety, the University of Tokyo.
Conflict of interest
The authors declare that they have no conflict of interest.
Ethics approval
All applicable international, national, and/or institutional guidelines for the care and use of animals were followed; all experimental procedures involving mice were approved by the Animal Experiment Ethics Committees at the Institute of Molecular and Cellular Biosciences, The University of Tokyo (Experiment# 2611, 2710, 2808), and the experiments were performed precisely in accordance with the manual provided by the Life Science Research Ethics and Safety, the University of Tokyo.
Research involving human participants and informed consent
This article does not contain any studies with human participants performed by any of the authors.
Funding
This work was supported by a Grant-in-Aid for Scientific Research from the Ministry of Education, Culture, Sports, Science and Technology (MEXT) in Japan (22125007 and 15H05976 to Y.O.). M.H. is a Research Fellow (DC2) for Young Scientists, JSPS (14J10178). This work was also partially supported by the Platform Project for Supporting in Drug Discovery and Life Science Research (Platform for Drug Discovery, Informatics, and Structural Life Science) from Japan Agency for Medical Research and Development (AMED).
Electronic supplementary material
ESM 1
(DOCX 19 kb)
Supplemental Fig. 1
(PDF 293 kb)
Supplemental Fig. 2
(PDF 961 kb)
Supplemental Fig. 3
(PDF 169 kb)
Supplemental Fig. 4
(PDF 167 kb)
Supplemental Fig. 5
(PDF 237 kb)
Supplemental Fig. 6
(PDF 641 kb)
Supplemental Fig. 7
(PDF 208 kb)
Supplemental Fig. 8
(PDF 157 kb)
Supplemental Fig. 9
(PDF 528 kb)
Supplemental Table 1
(PDF 150 kb)
Supplemental Table 2
(PDF 98.5 kb)
Rights and permissions
About this article
Cite this article
Hada, M., Kim, J., Inoue, E. et al. TH2A is phosphorylated at meiotic centromere by Haspin. Chromosoma 126, 769–780 (2017). https://doi.org/10.1007/s00412-017-0638-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00412-017-0638-5