Abstract
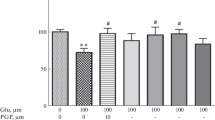
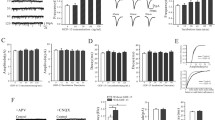
We have previously shown that interleukin-6 (IL-6) has neuroprotective effect against N-methyl-d-aspartate (NMDA)-induced excitotoxicity. The current study aimed to reveal signal transduction pathways involved in the IL-6 neuroprotection. Cerebellar granule neurons (CGNs) from postnatal 8-day infant rats were exposed to IL-6 (120 ng/ml) for 8 days and stimulated with NMDA (100 μM) for 15 or 30 min. Dynamic intracellular Ca2+ fluorescence intensity, cytosolic Ca2+-dependent phospholipase A2 (cPLA2) expression, and apoptosis and necrosis in cultured CGNs were measured by laser scanning confocal microscope, real-time PCR and Western blot, and annexin V-FITC/propidium iodide staining, respectively. NMDA stimulation of neurons evoked an intracellular Ca2+ overload, an upregulated expression of cPLA2, and an increase in cell death. Chronic IL-6 exposure prevented the NMDA-evoked neuronal Ca2+ overload, cPLA2 expression upregulation, and apoptosis and necrosis. Anti-gp130 monoclonal antibody (mAb), a blocker of gp130 that is a 130-kDa signal-transducing β-subunit of IL-6 receptor complex, blocked these effects of IL-6 preventing NMDA neurotoxicity. AG490, PD98059, or LY294002, inhibitors specific for the intracellular signals, JAK, MAPK, and PI3K, respectively, partially blocked these IL-6 neuroprotective effects. Phosphorylation levels of STAT3, ERK1/2, and AKT, the downstream proteins for these enzymes of JAK, MAPK, and PI3K, respectively, were elevated by IL-6 pretreatment. The enhanced activation of STAT3, ERK1/2, and AKT by IL-6 was abolished by AG490, PD98059, and LY294002, respectively. Anti-gp130 mAb attenuated the activation of all the three detected signaling molecules. The present findings suggest that IL-6 neuroprotection is jointly mediated by the cellular signal transduction pathways, gp130-JAK-STAT3, gp130-MAPK-ERK, and gp130-PI3K-AKT.




Similar content being viewed by others
References
Ali C, Nicole O, Docagne F, Lesne S, MacKenzie ET, Nouvelot A, Buisson A, Vivien D (2000) Ischemia-induced interleukin-6 as a potential endogenous neuroprotective cytokine against NMDA receptor-mediated excitotoxicity in the brain. J Cereb Blood Flow Metab 20:956–966
Arundine M, Tymianski M (2003) Molecular mechanisms of calcium-dependent neurodegeneration in excitotoxicity. Cell Calcium 34:325–337
Baker DG, Ekhator NN, Kasckow JW, Hill KK, Zoumakis E, Dashevsky BA, Chrousos GP, Geracioti TD Jr (2001) Plasma and cerebrospinal fluid interleukin-6 concentrations in posttraumatic stress disorder. NeuroImmunoModulation 9:209–217
Baranzini SE, Elfstrom C, Chang SY, Butunoi C, Murray R, Higuchi R, Oksenberg JR (2000) Transcriptional analysis of multiple sclerosis brain lesions reveals a complex pattern of cytokine expression. J Immunol 165:6576–6582
Bauer J, Strauss S, Volk B, Berger M (1991) IL-6-mediated events in Alzheimer’s disease pathology. Immunol Today 12:422
Bonventre JV (1996) Roles of phospholipases A2 in brain cell and tissue injury associated with ischemia and excitotoxicity. J Lipid Mediat Cell Signall 14:15–23
Burke JE, Dennis EA (2009) Phospholipase A2 structure/function, mechanism, and signaling. J Lipid Res 50:S237–S242
Campbell IL, Abraham CR, Masliah E, Kemper P, Inglis JD, Oldstone MB, Mucke L (1993) Neurologic disease induced in transgenic mice by cerebral overexpression of interleukin 6. Proc Natl Acad Sci USA 90:10061–10065
Chang JY, Tsai PF (2009) IL-6 release from mouse glia caused by MeHg requires cytosolic phospholipase A2 activation. Neurosci Lett 461:85–89
Cucchiaroni ML, Viscomi MT, Bernardi G, Molinari M, Guatteo E, Mercuri NB (2010) Metabotropic glutamate receptor 1 mediates the electrophysiological and toxic actions of the cycad derivative beta-N-methylamino-l-alanine on substantia nigra pars compacta DAergic neurons. J Neurosci 30:5176–5188
D’ Arcangelo G, Tancredi V, Onofri F, D’ Antuono M, Giovedi S, Benfenati F (2000) Interleukin-6 inhibits neurotransmitter release and the spread of excitation in the rat cerebral cortex. Eur J Neurosci 12:1241–1252
Farooqui AQA, Ong WY, Horrocks LA (2006) Inhibitors of brain phospholipase A2 activity: their neuropharmacological effects and therapeutic importance for the treatment of neurologic disorders. Pharmacol Rev 58:591–620
Fernández-Sánchez MT, Novelli A (1993) Basic fibroblast growth factor protects cerebellar neurons in primary culture from NMDA and non-NMDA receptor mediated neurotoxicity. FEBS Lett 235:124–131
Frei K, Fredrikson S, Fontana A, Link H (1991) Interleukin-6 is elevated in plasma in multiple sclerosis. J Neuroimmunol 31:147–153
Fukada T, Hibi M, Yamanaka Y, Takahashi-Tezuka M, Fujitani Y, Yamaguchi T, Nakajima K, Hirano T (1996) Two signals are necessary for cell proliferation induced by a cytokine receptor gp130: involvement of STAT3 in anti-apoptosis. Immunity 5:449–460
Gadient RA, Otten U (1993) Differential expression of interleukin-6 (IL-6) and interleukin-6 receptor (IL-6R) mRNAs in rat hypothalamus. Neurosci Lett 153:13–16
Gadient RA, Otten U (1994) Identification of interleukin-6 (IL-6)-expressing neurons in the cerebellum and hippocampus of normal adult rats. Neurosci Lett 182:243–246
Gentile MT, Reccia MG, Sorrentino PP, Vitale E, Sorrentino G, Puca AA, Colucci-D’Amato L (2012) Role of cytosolic calcium-dependent phospholipase A2 in Alzheimer’s disease pathogenesis. Mol Neurobiol 45:596–604
Gruol DL, Nelson TE (2005) Purkinje neuron physiology is altered by the inflammatory factor interleukin-6. Cerebellum 4:198–205
Ha BK, King JS (2000) Localization of gp130 in the developing and adult mouse cerebellum. J Chem Neuroanat 19:129–141
Hibi M, Hirano T (2000) Gab-family adapter molecules in signal transduction of cytokine and growth factor receptors, and T and B cell antigen receptors. Leuk Lymphoma 37:299–307
Inomata Y, Hirata A, Yonemura N, Koga T, Kido N, Tanihara H (2003) Neuroprotective effects of interleukin-6 on NMDA-induced rat retinal damage. Biochem Biophys Res Commun 302:226–232
Kamimura D, Ishihara K, Hirano T (2003) IL-6 signal transduction and its physiological roles: the signal orchestration model. Rev Physiol Biochem Pharmacol 149:1–38
Kriem B, Sponne I, Fifre A, Malaplate-Armand C, Lozac’h-Pillot K, Koziel V, Yen-Potin FT, Bihain B, Oster T, Jean-Luc Olivier, Pillot T (2005) Cytosolic phospholipase A2 mediates neuronal apoptosis induced by soluble oligomers of the amyloid-β peptide. FASEB J 19:85–87
Kushima Y, Hama T, Hatanaka H (1992) Interleukin-6 as a neurotrophic factor for promoting the survival of cultured catecholaminergic neurons in a chemically defined medium from fetal and postnatal rat midbrains. Neurosci Res 13:267–280
Liu Z, Qiu YH, Li B, Ma SH, Peng YP (2011) Neuroprotection of interleukin-6 against NMDA-induced apoptosis and its signal-transduction mechanisms. Neurotox Res 19:484–495
Livak KJ, Schmittgen TD (2001) Analysis of relative gene expression data using Real-Time quantitative PCR and the 2–44Ct method. Methods 25:402–408
Merrill JE, Chen IS (1991) HIV-1, macrophages, glial ells, and cytokines in AIDS nervous system disease. FASEB J 5:2391–2397
Nelson TE, Olde Engberink A, Hernandez R, Puro A, Huitron-Resendiz S, Hao C, De Graan PN, Gruol DL (2012) Altered synaptic transmission in the hippocampus of transgenic mice with enhanced central nervous systems expression of interleukin-6. Brain Behav Immun 26:959–971
Peng YP, Qiu YH, Lu JH, Wang JJ (2005) Interleukin-6 protects cultured cerebellar granule neurons against glutamate-induced neurotoxicity. Neurosci Lett 374:192–196
Pizzi M, Sarnico I, Boroni F, Benarese M, Dreano M, Garotta G, Valerio A, Spano P (2004) Prevention of neuron and oligodendrocyte degeneration by interleukin-6 (IL-6) and IL-6 receptor/IL-6 fusion protein in organotypic hippocampal slices. Mol Cell Neurosci 25:301–311
Qiu Z, Gruol DL (2003) Interleukin-6, beta-amyloid peptide and NMDA interactions in rat cortical neurons. J Neuroimmunol 139:51–57
Sharp JD, White DL (1993) Cytosolic PLA2: mRNA levels and potential for transcriptional regulation. J Lipid Mediat 8:183–189
Shelat PB, Chalimoniuk M, Wang JH, Strosznajder JB, Lee JC, Sun AY, Simonyi A, Sun GY (2008) Amyloid beta peptide and NMDA induce ROS from NADPH oxidase and AA release from cytosolic phospholipase A2 in cortical neurons. J Neurochem 106:45–55
Spooren A, Kolmus K, Laureys G, Clinckers R, De Keyser J, Haegeman G, Gerlo S (2011) Interleukin-6, a mental cytokine. Brain Res Rev 67:157–183
Stahl N, Farruggella TJ, Boulton TG, Zhong Z, Darnell JE Jr, Yancopoulos GD (1995) Choice of STATs and other substrates specified by modular tyrosine-based motifs in cytokine receptors. Science 267:1349–1353
Sun XM, Lu JH, Qiu YH, Liu Z, Wang XQ, Peng YP (2011) Interleukin-6 reduces NMDA-induced Ca2+ overload via prevention of Ca2+ release from intracellular store. Int J Neurosci 121:423–429
Tran HY, Shin EJ, Saito K, Nguyen XK, Chung YH, Jeong JH, Bach JH, Park DH, Yamada K, Nabeshima T, Yoneda Y, Kim HC (2012) Protective potential of IL-6 against trimethyltin-induced neurotoxicity in vivo. Free Radic Biol Med 52:1159–1174
Van Wagoner NJ, Benveniste EN (1999) Interleukin-6 expression and regulation in astrocytes. J Neuroimmunol 100:124–139
Wang XQ, Peng YP, Lu JH, Cao BB, Qiu YH (2009) Neuroprotection of interleukin-6 against NMDA attack and its signal transduction by JAK and MAPK. Neurosci Lett 450:122–126
Acknowledgments
We wish to thank Dr. M. Kerry O’Banion, University of Rochester, for editing this manuscript. This work was supported by Grants BK2010278 and BK2011386 from the Natural Science Foundation of Jiangsu Province of China; K2010047, BK2012014, BK2012015, and CP12012003 from the Nantong Applied Research Program of China; and a Project funded by the Priority Academic Program Development of Jiangsu Higher Education Institutions.
Conflict of interest
The authors declare that they have no conflict of interest.
Author information
Authors and Affiliations
Corresponding authors
Additional information
Xiao-Xia Fang and Xiao-Lin Jiang contributed equally to this study.
Rights and permissions
About this article
Cite this article
Fang, XX., Jiang, XL., Han, XH. et al. Neuroprotection of Interleukin-6 Against NMDA-induced Neurotoxicity is Mediated by JAK/STAT3, MAPK/ERK, and PI3K/AKT Signaling Pathways. Cell Mol Neurobiol 33, 241–251 (2013). https://doi.org/10.1007/s10571-012-9891-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10571-012-9891-6