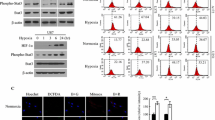
Abstract
Tumor hypoxia has been reported to be a negative prognostic factor in a number of tumor sites, which suggests a positive correlation between tumor hypoxia and increased metastatic efficiency. Evidence shows that vascular endothelial growth factor (VEGF) stimulates angiogenesis in tumor growth and mediates neuroprotection to prevent an apoptotic cell death. Human neuroblastoma cells (CHP126) were exposed to moderate hypoxia for different time spans to explore the molecular stress responses. Apoptotic features as an increase of Bax/Bcl-2 ratio and activation of caspase 3 were observed at early period of exposure time, but these effects were reversed with the extension of hypoxic treatment. Hypoxia also activated MAPKs signaling pathways in a time-relative manner, which were involved in the regulation of hypoxia-related resistance of CHP126 cells. Meanwhile, VEGF and its receptor KDR were found to interact with MAPKs signaling pathways except the effect of hypoxia. Furthermore, rhVEGF165 was utilized to discern that VEGF increased Bcl-2 and procaspase 3 expressions, contributing to a synergistic relationship of an angiogenic response with Bcl-2 in hypoxia via a cross talk, while the activation of ERK MAPK is important for both productions. These altered signals may be critical to predict a poor outcome; therefore, our knowledge provides new insight into apoptosis and angiogenesis control of tumor cells and suggests a strategy based on the blockade of hypoxia-induced VEGF signaling under hypoxia in neuroblastoma.







Similar content being viewed by others
References
Adams JM, Cory S (2007) The Bcl-2 apoptotic switch in cancer development and therapy. Oncogene 26:1324–1337. doi:10.1038/sj.onc.1210220
Alfranca A, Gutiérrez MD, Vara A, Aragonés J, Vidal F, Landázuri MO (2002) c-Jun and hypoxia-inducible factor 1 functionally cooperate in hypoxia-induced gene transcription. Mol Cell Biol 22:12–22. doi:10.1128/MCB.22.1.12-22.2002
Anai S, Goodison S, Shiverick K, Hirao Y, Brown BD, Rosser CJ (2007) Knock-down of Bcl-2 by antisense oligodeoxynucleotides induces radiosensitization and inhibition of angiogenesis in human PC-3 prostate tumor xenografts. Mol Cancer Ther 6:101–111. doi:10.1158/1535-7163.MCT-06-0367
Ballif BA, Blenis J (2001) Molecular mechanisms mediating mammalian mitogen-activated protein kinase (MAPK) kinase (MEK)-MAPK cell survival signals. Cell Growth Differ 12:397–408
Barone FC, Irving EA, Ray AM, Lee JC, Kassis S, Kumar S, Badger AM, Legos JJ, Erhardt JA, Ohlstein EH, Hunter AJ, Harrison DC, Philpott K, Smith BR, Adams JL, Parsons AA (2001) Inhibition of p38 mitogen-activated protein kinase provides neuroprotection in cerebral focal ischemia. Med Res Rev 21:129–145. doi:10.1002/1098-1128(200103)21:2<129::AID-MED1003>3.0.CO;2-H
Benasciutti E, Pagès G, Kenzior O, Folk W, Blasi F, Crippa MP (2004) MAPK and JNK transduction pathways can phosphorylate Sp1 to activate the uPA minimal promoter element and endogenous gene transcription. Blood 104:256–262. doi:10.1182/blood-2003-08-2661
Berra E, Milanini J, Richard DE, Le Gall M, Viñals F, Gothié E, Roux D, Pagès G, Pouysségur J (2000) Signaling angiogenesis via p42/p44 MAP kinase and hypoxia. Biochem Pharmacol 60:1171–1178. doi:10.1016/S0006-2952(00)00423-8
Bickler PE, Donohoe PH, Buck LT (2000) Hypoxia-induced silencing of NMDA receptors in turtle neurons. J Neurosci 20:3522–3528
Boldt S, Weidle UH, Kolch W (2002) The role of MAPK pathways in the action of chemotherapeutic drugs. Carcinogenesis 23:1831–1838. doi:10.1093/carcin/23.11.1831
Brunet A, Roux D, Lenormand P, Dowd S, Keyse S, Pouysségur J (1999) Nuclear translocation of p42/p44 mitogen-activated protein kinase is required for growth factor-induced gene expression and cell cycle entry. EMBO J 18:664–674. doi:10.1093/emboj/18.3.664
Byzova TV, Goldman CK, Pampori N, Thomas KA, Bett A, Shattil SJ, Plow EF (2000) A mechanism for modulation of cellular responses to VEGF: activation of the integrins. Mol Cell 6:851–860
Choi WS, Eom DS, Han BS, Kim WK, Han BH, Choi EJ, Oh TH, Markelonis GJ, Cho JW, Oh YJ (2004) Phosphorylation of p38 MAPK induced by oxidative stress is linked to activation of both caspase-8- and -9-mediated apoptotic pathways in dopaminergic neurons. J Biol Chem 279:20451–20460. doi:10.1074/jbc.M311164200
Conrad PW, Rust RT, Han J, Millhorn DE, Beitner-Johnson D (1999) Selective activation of p38alpha and p38gamma by hypoxia. Role in regulation of cyclin D1 by hypoxia in PC12 cells. J Biol Chem 274:23570–23576. doi:10.1074/jbc.274.33.23570
Dewil M, dela Cruz VF, Van Den Bosch L, Robberecht W (2007) Inhibition of p38 mitogen activated protein kinase activation and mutant SOD1(G93A)-induced motor neuron death. Neurobiol Dis 26(2):332–341. doi:10.1016/j.nbd.2006.12.023
Dias S, Shmelkov SV, Lam G, Rafii S (2002) VEGF(165) promotes survival of leukemic cells by Hsp90-mediated induction of Bcl-2 expression and apoptosis inhibition. Blood 99:2532–2540. doi:10.1182/blood.V99.7.2532
Feng H, Xiang H, Mao YW, Wang J, Liu JP, Huang XQ, Liu Y, Liu SJ, Luo C, Zhang XJ, Liu Y, Li DW (2004) Human Bcl-2 activates ERK signaling pathway to regulate activating protein-1, lens epithelium-derived growth factor and downstream genes. Oncogene 23:7310–7321. doi:10.1038/sj.onc.1208041
Fernandez A, Udagawa T, Schwesinger C, Beecken W, Achilles-Gerte E, McDonnell T, D’Amato R (2001) Angiogenic potential of prostate carcinoma cells overexpressing bcl-2. J Natl Cancer Inst 93:208–213. doi:10.1093/jnci/93.3.208
Ferrara N, Gerber HP, LeCouter J (2003) The biology of VEGF and its receptors. Nat Med 9:669–676. doi:10.1038/nm0603-669
Folkman J (1990) What is the evidence that tumors are angiogenesis dependent? J Natl Cancer Inst 82:4–6. doi:10.1093/jnci/82.1.4
Helmlinger G, Yuan F, Dellian M, Jain RK (1997) Interstitial pH and pO2 gradients in solid tumors in vivo: high-resolution measurements reveal a lack of correlation. Nat Med 3:177–182. doi:10.1038/nm0297-177
Hou ST, Xie X, Baggley A, Park DS, Chen G, Walker T (2002) Activation of the Rb/E2F1 pathway by the nonproliferative p38 MAPK during Fas (APO1/CD95)-mediated neuronal apoptosis. J Biol Chem 277:48764–48770. doi:10.1074/jbc.M206336200
Jin K, Mao XO, Zhu Y, Greenberg DA (2002) MEK and ERK protect hypoxic cortical neurons via phosphorylation of Bad. J Neurochem 80:119–125. doi:10.1046/j.0022-3042.2001.00678.x
Junn E, Mouradian MM (2001) Apoptotic signaling in dopamine-induced cell death: the role of oxidative stress, p38 mitogen-activated protein kinase, cytochrome c and caspases. J Neurochem 78:374–383. doi:10.1046/j.1471-4159.2001.00425.x
Kilic E, Kilic U, Wang Y, Bassetti CL, Marti HH, Hermann DM (2006) The phosphatidylinositol-3 kinase/Akt pathway mediates VEGF’s neuroprotective activity and induces blood brain barrier permeability after focal cerebral ischemia. FASEB J 20:1185–1187. doi:10.1096/fj.05-4829fje
Kim DS, Kim SY, Lee JE, Kwon SB, Joo YH, Youn SW, Park KC (2003) Sphingosine-1-phosphate-induced ERK activation protects human melanocytes from UVB-induced apoptosis. Arch Pharm Res 26:739–746
Kraemer M, Tournaire R, Dejong V, Montreau N, Briane D, Derbin C, Binétruy B (1999) Rat embryo fibroblasts transformed by c-Jun display highly metastatic and angiogenic activities in vivo and deregulate gene expression of both angiogenic and antiangiogenic factors. Cell Growth Differ 10:193–200
Le-Niculescu H, Bonfoco E, Kasuya Y, Claret F, Green DR, Karin M (1999) Withdrawal of survival factors results in activation of the JNK pathway in neuronal cells leading to Fas ligand induction and cell death. Mol Cell Biol 19:751–763
Mackay K, Mochly-Rosen D (1999) An inhibitor of p38 mitogen-activated protein kinase protects neonatal cardiac myocytes from ischemia. J Biol Chem 274:6272–6279. doi:10.1074/jbc.274.10.6272
Makin G, Dive C (2001) Modulating sensitivity to drug-induced apoptosis: the future for chemotherapy? Breast Cancer Res 3:150–153. doi:10.1186/bcr289
Martin JL, Avkiran M, Quinlan RA, Cohen P, Marber MS (2001) Antiischemic effects of SB203580 are mediated through the inhibition of p38alpha mitogen-activated protein kinase: Evidence from ectopic expression of an inhibition-resistant kinase. Circ Res 89:750–752. doi:10.1161/hh2101.099504
Maulik D, Ashraf QM, Mishra OP, Delivoria-Papadopoulos M (2008) Activation of p38 mitogen-activated protein kinase (p38 MAPK), extracellular signal-regulated kinase (ERK) and c-jun N-terminal kinase (JNK) during hypoxia in cerebral cortical nuclei of guinea pig fetus at term: Role of nitric oxide. Neurosci Lett 439:94–99. doi:10.1016/j.neulet.2008.02.037
Meister B, Grünebach F, Bautz F, Brugger W, Fink FM, Kanz L, Möhle R (1999) Expression of vascular endothelial growth factor (VEGF) and its receptors in human neuroblastoma. Eur J Cancer 35:445–449. doi:10.1016/S0959-8049(98)00387-6
Neufeld G, Cohen T, Gengrinovitch S, Poltorak Z (1999) Vascular endothelial growth factor (VEGF) and its receptors. FASEB J 13:9–22
Poulaki V, Qin W, Joussen AM, Hurlbut P, Wiegand SJ, Rudge J, Yancopoulos GD, Adamis AP (2002) Acute intensive insulin therapy exacerbates diabetic blood-retinal barrier breakdown via hypoxia-inducible factor-1alpha and VEGF. J Clin Invest 109:805–815
Qian D, Lin HY, Wang HM, Zhang X, Liu DL, Li QL, Zhu C (2004) Normoxic induction of the hypoxic-inducible factor-1 alpha by interleukin-1 beta involves the extracellular signal-regulated kinase 1/2 pathway in normal human cytotrophoblast cells. Biol Reprod 70:1822–1827. doi:10.1095/biolreprod.103.025031
Rosser CJ, Reyes AO, Vakar-Lopez F, Levy LB, Kuban DA, Hoover DC, Lee AK, Pisters LL (2003) Bcl-2 is significantly overexpressed in localized radio-recurrent prostate carcinoma, compared with localized radio-naive prostate carcinoma. Int J Radiat Oncol Biol Phys 56:1–6. doi:10.1016/S0360-3016(02)04468-1
Schlesinger HR, Gerson JM, Moorhead PS, Maguire H, Hummeler K (1976) Establishment and characterization of human neuroblastoma cell lines. Cancer Res 36:3000–3094
Semenza GL (2001) HIF-1 and mechanisms of hypoxia sensing. Curr Opin Cell Biol 13:167–171. doi:10.1016/S0955-0674(00)00194-0
Shakibaei M, Schulze-Tanzil G, de Souza P, John T, Rahmanzadeh M, Rahmanzadeh R, Merker HJ (2001) Inhibition of mitogen-activated protein kinase kinase induces apoptosis of human chondrocytes. J Biol Chem 276:13289–13294. doi:10.1074/jbc.M010859200
Takahashi H, Shibuya M (2005) The vascular endothelial growth factor (VEGF)/VEGF receptor system and its role under physiological and pathological conditions. Clin Sci (Lond) 109:227–241. doi:10.1042/CS20040370
Tokuda H, Hirade K, Wang X, Oiso Y, Kozawa O (2003) Involvement of SAPK/JNK in basic fibroblast growth factor-induced vascular endothelial growth factor release in osteoblasts. J Endocrinol 177:101–107. doi:10.1677/joe.0.1770101
Trisciuoglio D, Iervolino A, Candiloro A, Fibbi G, Fanciulli M, Zangemeister-Wittke U, Zupi G, Del Bufalo D (2004) bcl-2 induction of urokinase plasminogen activator receptor expression in human cancer cells through Sp1 activation: involvement of ERK1/ERK2 activity. J Biol Chem 279:6737–6745. doi:10.1074/jbc.M308938200
Trisciuoglio D, Iervolino A, Zupi G, Del Bufalo D (2005) Involvement of PI3 K and MAPK signaling in bcl-2-induced vascular endothelial growth factor expression in melanoma cells. Mol Biol Cell 16:4153–4162. doi:10.1091/mbc.E04-12-1087
Yokoi K, Fidler IJ (2004) Hypoxia increases resistance of human pancreatic cancer cells to apoptosis induced by gemcitabine. Clin Cancer Res 10:2299–2306. doi:10.1158/1078-0432.CCR-03-0488
Zachary I (2003) VEGF signalling: integration and multi-tasking in endothelial cell biology. Biochem Soc Trans 31(Pt 6):1171–1177
Zeitlin BD, Joo E, Dong Z, Warner K, Wang G, Nikolovska-Coleska Z, Wang S, Nör JE (2006) Antiangiogenic effect of TW37, a small-molecule inhibitor of Bcl-2. Cancer Res 66:8698–8706. doi:10.1158/0008-5472.CAN-05-3691
Zhang Y, Furumura M, Morita E (2008) Distinct signaling pathways confer different vascular responses to VEGF 121 and VEGF 165. Growth Factors 26:125–131. doi:10.1080/08977190802105909
Zhu X, Mei M, Lee HG, Wang Y, Han J, Perry G, Smith MA (2005) P38 activation mediates amyloid-beta cytotoxicity. Neurochem Res 30:791–796. doi:10.1007/s11064-005-6872-x
Acknowledgments
The authors thank the grant sponsors. Grant information: This study received financial support from the National Natural Science Foundation (30672484), the Natural Science Foundation of Zhejiang Province (R20512), and Zhejiang Provincial Program for the cultivation of High-level Innovative Health talents.
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Wang, D., Weng, Q., Zhang, L. et al. VEGF and Bcl-2 Interact Via MAPKs Signaling Pathway in the Response to Hypoxia in Neuroblastoma. Cell Mol Neurobiol 29, 391–401 (2009). https://doi.org/10.1007/s10571-008-9331-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10571-008-9331-9