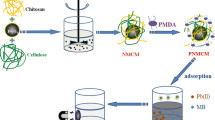
Abstract
Dye contamination of water supplies has a serious threat to human health, prompting the development of highly effective and eco-friendly adsorbents. In this work, polyelectrolyte microspheres derived from positively charged chitosan and negatively charged cellulose were constructed in alkali/urea solvent by a simple water/oil emulsification. The obtained chitosan/cellulose microspheres (CCM) were further used for the removal of reactive black 5. By using alkali/urea solution as the solvent, a homogeneous chitosan/cellulose solution was achieved, which avoided the easy occurrence of agglomeration between oppositely charged polymers. More importantly, CCM showed significantly improved mechanical strength and anti-swelling properties compared with pure chitosan microspheres (CM). Adsorption experiments demonstrated that CCM can effectively remove reactive black 5 with high adsorption capacity of 214.36 mg/g, fast adsorption kinetic that reached 76% of the equilibrium adsorption amount within only 20 min, and good reusability that maintained 75% efficiency even after five times of adsorption/desorption cycle, indicating a great potential for the application of dye removal.








Similar content being viewed by others
References
Ali I (2012) New generation adsorbents for water treatment. Chem Rev 112:5073–5091. https://doi.org/10.1021/cr300133d
Beluci NDL et al (2019) Hybrid treatment of coagulation/flocculation process followed by ultrafiltration in TIO2-modified membranes to improve the removal of reactive black 5 dye. Sci Total Environ 664:222–229. https://doi.org/10.1016/j.scitotenv.2019.01.199
Chacon-Patino ML, Blanco-Tirado C, Hinestroza JP, Combariza MY (2013) Biocomposite of nanostructured MnO2 and fique fibers for efficient dye degradation. Green Chem 15:2920–2928. https://doi.org/10.1039/c3gc40911b
Feng F, Liu Y, Hu K (2004) Influence of alkali-freezing treatment on the solid state structure of chitin. Carbohyd Res 339:2321–2324. https://doi.org/10.1016/j.carres.2004.06.017
Hamzehloo M, Farahani BKA, Rostamian R (2019) Adsorption behaviour of reactive black dye 5 by magnetically separable nanoadsorbent. Phys Chem Res 7:475–490. https://doi.org/10.22036/pcr.2019.186198.1639
Hasani M, Cranston ED, Westman G, Gray DG (2008) Cationic surface functionalization of cellulose nanocrystals. Soft Matter 4:2238–2244. https://doi.org/10.1039/b806789a
He M et al (2018) Construction of novel cellulose/chitosan composite hydrogels and films and their applications. Cellulose 25:1987–1996. https://doi.org/10.1007/s10570-018-1683-9
Heibati B, Rodriguez-Couto S, Amrane A, Rafatullah M, Hawari A, Al-Ghouti MA (2014) Uptake of reactive black 5 by pumice and walnut activated carbon: chemistry and adsorption mechanisms. J Ind Eng Chem 20:2939–2947. https://doi.org/10.1016/j.jiec.2013.10.063
Huang Y et al (2015) Effects of chitin whiskers on physical properties and osteoblast culture of alginate based nanocomposite hydrogels. Biomacromol 16:3499–3507. https://doi.org/10.1021/acs.biomac.5b00928
Huang P, Xia D, Kazlauciunas A, Thornton P, Lin L, Menzel R (2019) Dye-mediated interactions in chitosan-based polyelectrolyte/organoclay hybrids for enhanced adsorption of industrial dyes. Acs Appl Mater Inter 11:11961–11969. https://doi.org/10.1021/acsami.9b01648
Il’Ina AV, Varlamov VP (2005) Chitosan-based polyelectrolyte complexes: a review. Appl Biochem Microbiol 41(1):5–11
Karadag D, Turan M, Akgul E, Tok S, Faki A (2007) Adsorption equilibrium and kinetics of reactive black 5 and reactive red 239 in aqueous solution onto surfactant-modified zeolite. J Chem Eng Data 52:1615–1620. https://doi.org/10.1021/je7000057
Kim TY, Park SS, Cho SY (2012) Adsorption characteristics of reactive black 5 onto chitosan beads cross-linked with epichlorohydrin. J Ind Eng Chem 18:1458–1464. https://doi.org/10.1016/j.jiec.2012.02.006
Kim JK et al (2017) hierarchical chitin fibers with aligned nanofibrillar architectures: a nonwoven-mat separator for lithium metal batteries. ACS Nano 11:6114–6121. https://doi.org/10.1021/acsnano.7b02085
Kumar S, Verma G, Gao WY, Niu Z, Wojtas L, Ma S (2016) Anionic metal–organic framework for selective dye removal and CO2 fixation. Eur J Inorg Chem 2016(27):4373–4377
Lai KC, Lee LY, Hiew BYZ, Yang TCK, Pan GT, Thangalazhy-Gopakumar S, Gan SY (2020) Utilisation of eco-friendly and low cost 3D graphene-based composite for treatment of aqueous Reactive Black 5 dye: Characterisation, adsorption mechanism and recyclability studies. J Taiwan Inst Chem E 114:57–66. https://doi.org/10.1016/j.jtice.2020.09.024
Li J, Revol JF, Marchessault RH (1997) Effect of degree of deacetylation of chitin on the properties of chitin crystallites. J Appl Polym Sci 65:373–380
Li ZC et al (2019) Interpretation of the adsorption mechanism of Reactive Black 5 and Ponceau 4R dyes on chitosan/polyamide nanofibers via advanced statistical physics model. J Mol Liq 285:165–170. https://doi.org/10.1016/j.molliq.2019.04.091
Liu J, Wang N, Zhang HL, Baeyens J (2019) Adsorption of Congo red dye on FexCo3-xO4 nanoparticles. J Environ Manage 238:473–483. https://doi.org/10.1016/j.jenvman.2019.03.009
Luo YC, Wang Q (2014) Recent development of chitosan-based polyelectrolyte complexes with natural polysaccharides for drug delivery. Int J Biol Macromol 64:353–367. https://doi.org/10.1016/j.ijbiomac.2013.12.017
Nair AK, Kumar BV, Kalaiarasan G, Babu PEJ (2018) TiO2 nanosheet incorporated polysulfone ultrafiltration membranes for dye removal. Desalin Water Treat 107:324–331. https://doi.org/10.5004/dwt.2018.22157
Qiao LZ, Li SS, Li YL, Liu Y, Du KF (2020a) Fabrication of superporous cellulose beads via enhanced inner cross-linked linkages for high efficient adsorption of heavy metal ions. J Clean Prod. https://doi.org/10.1016/j.jclepro.2020.120017
Qiao LZ, Zhao LS, Du K (2020b) Construction of hierarchically porous chitin microspheres via a novel Dual-template strategy for rapid and High-capacity removal of heavy metal ions. Chem Eng J. https://doi.org/10.1016/j.cej.2020.124818
Ramya M, Karthika M, Selvakumar R, Raj B, Ravi KR (2017) A facile and efficient single step ball milling process for synthesis of partially amorphous Mg-Zn-Ca alloy powders for dye degradation. J Alloy Compd 696:185–192. https://doi.org/10.1016/j.jallcom.2016.11.221
Ranjbari S, Tanhaei B, Ayati A, Sillanpaa M (2019) Novel Aliquat-336 impregnated chitosan beads for the adsorptive removal of anionic azo dyes. Int J Biol Macromol 125:989–998. https://doi.org/10.1016/j.ijbiomac.2018.12.139
Sahel K, Perol N, Chermette H, Bordes C, Derriche Z, Guillard C (2007) Photocatalytic decolorization of Remazol Black 5 (RB5) and Procion Red MX-5B - Isotherm of adsorption, kinetic of decolorization and mineralization. Appl Catal B-Environ 77:100–109. https://doi.org/10.1016/j.apcatb.2007.06.016
Salam A, Lucia LA, Jameel H (2013) A novel cellulose nanocrystals-based approach to improve the mechanical properties of recycled paper. Acs Sustain Chem Eng 1:1584–1592. https://doi.org/10.1021/sc400226m
Smitha B, Sridhar S, Khan AA (2004) Polyelectrolyte complexes of chitosan and poly(acrylic acid) as proton exchange membranes for fuel cells. Macromolecules 37:2233–2239. https://doi.org/10.1021/ma0355913
Wang SJ, Kong FG, Fatehi P, Hou QX (2018) Cationic high molecular weight lignin polymer: a flocculant for the removal of anionic azo-dyes from simulated wastewater. Molecules. https://doi.org/10.3390/molecules23082005
Wei XQ, Duan JJ, Xu XJ, Zhang LN (2017) Highly efficient one-step purification of sulfated polysaccharides via chitosan microspheres adsorbents. Acs Sustain Chem Eng 5:3195–3203. https://doi.org/10.1021/acssuschemeng.6b02975
Xiao H, Peng H, Deng SH, Yang XY, Zhang YZ, Li YW (2012) Preparation of activated carbon from edible fungi residue by microwave assisted K2CO3 activation-Application in reactive black 5 adsorption from aqueous solution. Bioresource Technol 111:127–133. https://doi.org/10.1016/j.biortech.2012.02.054
Yan WL, Bai RB (2005) Adsorption of lead and humic acid on chitosan hydrogel beads. Water Res 39:688–698
Zeng HH, Wang L, Zhang D, Yan P, Nie J, Sharma VK, Wang CY (2019) Highly efficient and selective removal of mercury ions using hyperbranched polyethylenimine functionalized carboxymethyl chitosan composite adsorbent. Chem Eng J 358:253–263. https://doi.org/10.1016/j.cej.2018.10.001
Zhang YQ, Xue CH, Xue Y, Gao RC, Zhang XL (2005) Determination of the degree of deacetylation of chitin and chitosan by X-ray powder diffraction. Carbohyd Res 340:1914–1917. https://doi.org/10.1016/j.carres.2005.05.005
Zhang WJ, Li Q, Mao Q, He GH (2019) Cross-linked chitosan microspheres: an efficient and eco-friendly adsorbent for iodide removal from waste water. Carbohyd Polym 209:215–222. https://doi.org/10.1016/j.carbpol.2019.01.032
Zhang XR, Fan WD, Jiang WF, Li Y, Wang YT, Fu MY, Sun DF (2021) Optimizing Fe-based metal-organic frameworks through ligand conformation regulation for efficient dye adsorption and C2H2/CO2 separation. Chem-Eur J. https://doi.org/10.1002/chem.202101053
Acknowledgments
The work was funded by Natural Science Foundation of China (21676170). We thank Shuping Zheng (Analytical & Testing Center, Sichuan University) for her help in SEM observations. We also thank Yanping Huang from Center of Engineering Experimental Teaching, School of Chemical Engineering, Sichuan University for the help of SEM image.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflict of interests.
Ethical approval
This article does not contain any studies with human participants or animals performed by any of the authors.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Qiao, L., Wang, S., Wang, T. et al. High-strength and low-swelling chitosan/cellulose microspheres as a high-efficiency adsorbent for dye removal. Cellulose 28, 9323–9333 (2021). https://doi.org/10.1007/s10570-021-04111-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10570-021-04111-2