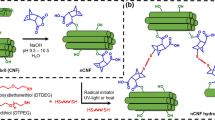
Abstract
Chemically crosslinked cellulose hydrogels have wide applications in agriculture and biomedicine, but most crosslinking methods involve potentially toxic crosslinking chemistries or lack significant control over the final modulus of the material. To overcome these challenges, carboxymethyl cellulose (CMC) hydrogels were synthesized utilizing thiol-ene click chemistry. CMC was functionalized with norbornene groups through a base catalyzed, water-borne functionalization reaction with carbic anhydride, yielding a reactive norbornene group. Both reaction pH and anhydride concentration could be used to control the degree of norbornene functionalization of CMC up to 45% norbornene functionalization per CMC repeat unit. This new norbornene functionalized CMC (cCMC) was crosslinked though a UV-light initiated thiol-ene reaction with a 2,2′-(ethylenedioxy)diethanethiol (DEG) crosslinker. Both the ratio of thiols to norbornenes and the irradiation time could be varied at a constant polymer concentration to control the modulus over an order of magnitude. Interestingly, thiol to norbornene ratios of 1:2 and 1:1 yielded the same modulus values, which was attributed to the crosslinking limiting chain mobility early in the reaction and preventing increased crosslink density as the reaction progressed. Hydrolytic degradation of the hydrogels yielded two degradation regimes of initial burst release and continuous daily release. Burst release behavior was tied to the thiol to norbornene ratio used to fabricate the hydrogel, while the daily degradation rate could be correlated to the crosslinking density. Due to its straightforward synthesis and significant control over modulus and degradation rates, cCMC offers high utility for future applications where cellulose derived hydrogels are needed.
Graphical abstract






Similar content being viewed by others
References
Ahmed EM (2015) Hydrogel: preparation, characterization, and applications: a review. J Adv Res 6:105–121. https://doi.org/10.1016/j.jare.2013.07.006
Anseth KS, Bowman CN, Brannon-Peppas L (1996) Mechanical properties of hydrogels and their experimental determination. Biomaterials 17:1647–1657. https://doi.org/10.1016/0142-9612(96)87644-7
Bakarich SE, Pidcock GC, Balding P et al (2012) Recovery from applied strain in interpenetrating polymer network hydrogels with ionic and covalent cross-links. Soft Matter 8:9985–9988. https://doi.org/10.1039/c2sm26745d
Barbucci R, Magnani A, Consumi M (2000) Swelling behavior of carboxymethylcellulose hydrogels in relation to cross-linking, pH, and charge density. Macromolecules 33:7475–7480. https://doi.org/10.1021/ma0007029
Browning MB, Cereceres SN, Luong PT, Cosgriff-Hernandez EM (2014) Determination of the in vivo degradation mechanism of PEGDA hydrogels. J Biomed Mater Res A 102:4244–4251. https://doi.org/10.1002/jbm.a.35096
Caló E, Khutoryanskiy VV (2015) Biomedical applications of hydrogels: a review of patents and commercial products. Eur Polym J 65:252–267. https://doi.org/10.1016/j.eurpolymj.2014.11.024
Camacho FG, Grima EM, Mirón AS et al (2001) Carboxymethyl cellulose protects algal cells against hydrodynamic stress. Enzyme Microb Technol 29:602–610. https://doi.org/10.1016/S0141-0229(01)00442-2
Chang C, Zhang L (2011) Cellulose-based hydrogels: present status and application prospects. Carbohydr Polym 84:40–53. https://doi.org/10.1016/j.carbpol.2010.12.023
Chang C, Lue A, Zhang L (2008) Effects of crosslinking methods on structure and properties of cellulose/PVA hydrogels. Macromol Chem Phys 209:1266–1273. https://doi.org/10.1002/macp.200800161
Dadoo N, Gramlich WM (2016) Spatiotemporal modification of stimuli-responsive hyaluronic acid/poly(N-isopropylacrylamide) hydrogels. ACS Biomater Sci Eng 2:1341–1350. https://doi.org/10.1021/acsbiomaterials.6b00259
Dadoo N, Landry SB, Bomar JD, Gramlich WM (2017) Synthesis and spatiotemporal modification of biocompatible and stimuli-responsive carboxymethyl cellulose hydrogels using thiol-norbornene chemistry. Macromol Biosci. https://doi.org/10.1002/mabi.201700107
Demitri C, Scalera F, Madaghiele M et al (2013) Potential of cellulose-based superabsorbent hydrogels as water reservoir in agriculture. Int J Polym Sci. https://doi.org/10.1155/2013/435073
Denisin AK, Pruitt BL (2016) Tuning the range of polyacrylamide gel stiffness for mechanobiology applications. ACS Appl Mater Interfaces 8:21893–21902. https://doi.org/10.1021/acsami.5b09344
Drury JL, Mooney DJ (2003) Hydrogels for tissue engineering: scaffold design variables and applications. Biomaterials 24:4337–4351. https://doi.org/10.1016/S0142-9612(03)00340-5
Fekete T, Borsa J, Takács E, Wojnárovits L (2017) Synthesis of carboxymethylcellulose/starch superabsorbent hydrogels by gamma-irradiation. Chem Cent J 11:46. https://doi.org/10.1186/s13065-017-0273-5
Fox SC, Li B, Xu D, Edgar KJ (2011) Regioselective esterification and etherification of cellulose: a review. Biomacromol 12:1956–1972. https://doi.org/10.1021/bm200260d
Giz A, Catalgil-Giz H, Alb A et al (2001) Kinetics and mechanisms of acrylamide polymerization from absolute, online monitoring of polymerization reaction. Macromolecules 34:1180–1191. https://doi.org/10.1021/ma000815s
Graham HD (1972) Determination of carboxymethylcellulose with chromotropic acid. J Dairy Sci 55:42–50. https://doi.org/10.3168/jds.S0022-0302(72)85430-4
Gramlich WM, Kim IL, Burdick JA (2013) Synthesis and orthogonal photopatterning of hyaluronic acid hydrogels with thiol-norbornene chemistry. Biomaterials 34:9803–9811. https://doi.org/10.1016/j.biomaterials.2013.08.089
Gupta MS, Nicoll SB (2014) Functional nucleus pulposus-like matrix assembly by human mesenchymal stromal cells is directed by macromer concentration in photocrosslinked carboxymethylcellulose hydrogels. Cell Tissue Res 358:527. https://doi.org/10.1007/s00441-014-1962-1
Hoenich NA (2007) Cellulose for medical applications: past, present, and future. BioResources 1:270–280. https://doi.org/10.15376/biores.1.2.270-280
Hoffman AS (2012) Hydrogels for biomedical applications. Adv Drug Deliv Rev 64(Supplement):18–23. https://doi.org/10.1016/j.addr.2012.09.010
Hyon S-H, Cha W-I, Ikada Y et al (1994) Poly(vinyl alcohol) hydrogels as soft contact lens material. J Biomater Sci Polym Ed 5:397–406. https://doi.org/10.1163/156856294X00103
Jeong B, Kim SW, Bae YH (2002) Thermosensitive sol–gel reversible hydrogels. Adv Drug Deliv Rev 54:37–51. https://doi.org/10.1016/S0169-409X(01)00242-3
Kade MJ, Burke DJ, Hawker CJ (2010) The power of thiol-ene chemistry. J Polym Sci Part Polym Chem 48:743–750. https://doi.org/10.1002/pola.23824
Karadağ E, Saraydin D, Çaldiran Y, Güven O (2000) Swelling studies of copolymeric acrylamide/crotonic acid hydrogels as carriers for agricultural uses. Polym Adv Technol 11:59–68. https://doi.org/10.1002/(SICI)1099-1581(200002)11:2%3c59:AID-PAT937%3e3.0.CO;2-Z
Kolb HC, Finn MG, Sharpless KB (2001) Click chemistry: diverse chemical function from a few good reactions. Angew Chem Int Ed 40:2004–2021. https://doi.org/10.1002/1521-3773(20010601)40:11%3c2004:AID-ANIE2004%3e3.0.CO;2-5
Kono H (2014) Characterization and properties of carboxymethyl cellulose hydrogels crosslinked by polyethylene glycol. Carbohydr Polym 106:84–93. https://doi.org/10.1016/j.carbpol.2014.02.020
Kulik E, Ikada Y (1996) In vitro platelet adhesion to nonionic and ionic hydrogels with different water contents. J Biomed Mater Res 30:295–304. https://doi.org/10.1002/(SICI)1097-4636(199603)30:3%3c295:AID-JBM4%3e3.0.CO;2-L
Lee S, Park YH, Ki CS (2016) Fabrication of PEG-carboxymethylcellulose hydrogel by thiol-norbornene photo-click chemistry. Int J Biol Macromol 83:1–8. https://doi.org/10.1016/j.ijbiomac.2015.11.050
Lentz RD (2015) Polyacrylamide and biopolymer effects on flocculation, aggregate stability, and water seepage in a silt loam. Geoderma 241–242:289–294. https://doi.org/10.1016/j.geoderma.2014.11.019
Lin C-C, Anseth KS (2009) PEG hydrogels for the controlled release of biomolecules in regenerative medicine. Pharm Res 26:631–643. https://doi.org/10.1007/s11095-008-9801-2
Lin HA, Gupta MS, Varma MS, Gilchrist ML, Nicoll SB (2016) Lower crosslinking density enhances functional nucleus pulposus-like matrix elaboration by human mesenchymal stem cells in carboxymethylcellulose hydrogels. J Biomed Mater Res 104:165–177. https://doi.org/10.1002/jbm.a.35552
Märtson M, Viljanto J, Hurme T et al (1999) Is cellulose sponge degradable or stable as implantation material? An in vivo subcutaneous study in the rat. Biomaterials 20:1989–1995. https://doi.org/10.1016/S0142-9612(99)00094-0
Meng X, Edgar KJ (2016) “Click” reactions in polysaccharide modification. Prog Polym Sci 53:52–85. https://doi.org/10.1016/j.progpolymsci.2015.07.006
Mũnoz Z, Shih H, Lin CC (2014) Gelatin hydrogels formed by orthogonal thiol-norbornene photochemistry for cell encapsulation. Biomater Sci 2:1063–1072
Nakayama A, Kakugo A, Gong JP et al (2004) High mechanical strength double-network hydrogel with bacterial cellulose. Adv Funct Mater 14:1124–1128. https://doi.org/10.1002/adfm.200305197
Nie H, Liu M, Zhan F, Guo M (2004) Factors on the preparation of carboxymethylcellulose hydrogel and its degradation behavior in soil. Carbohydr Polym 58:185–189. https://doi.org/10.1016/j.carbpol.2004.06.035
Oyen ML (2014) Mechanical characterisation of hydrogel materials. Int Mater Rev 59:44–59. https://doi.org/10.1179/1743280413Y.0000000022
Peng BL, Dhar N, Liu HL, Tam KC (2011) Chemistry and applications of nanocrystalline cellulose and its derivatives: a nanotechnology perspective. Can J Chem Eng 89:1191–1206. https://doi.org/10.1002/cjce.20554
Peppas NA, Bures P, Leobandung W, Ichikawa H (2000) Hydrogels in pharmaceutical formulations. Eur J Pharm Biopharm 50:27–46. https://doi.org/10.1016/S0939-6411(00)00090-4
Pourjavadi A, Barzegar S, Mahdavinia GR (2006) MBA-crosslinked Na-Alg/CMC as a smart full-polysaccharide superabsorbent hydrogels. Carbohydr Polym 66:386–395. https://doi.org/10.1016/j.carbpol.2006.03.013
Reza AT, Nicoll SB (2010) Characterization of novel photocrosslinked carboxymethylcellulose hydrogels for encapsulation of nucleus pulposus cells. Acta Biomater 6:179–186. https://doi.org/10.1016/j.actbio.2009.06.004
Sannino A, Demitri C, Madaghiele M (2009) Biodegradable cellulose-based hydrogels: design and applications. Materials 2:353–373. https://doi.org/10.3390/ma2020353
Sehaqui H, Kulasinski K, Pfenninger N et al (2017) Highly carboxylated cellulose nanofibers via succinic anhydride esterification of wheat fibers and facile mechanical disintegration. Biomacromol 18:242–248. https://doi.org/10.1021/acs.biomac.6b01548
Shih H, Lin C-C (2012) Cross-linking and degradation of step-growth hydrogels formed by thiol-ene photoclick chemistry. Biomacromol 13:2003–2012. https://doi.org/10.1021/bm300752j
Shokri J, Adibkia K (2013) Application of cellulose and cellulose derivatives in pharmaceutical industries. In: van de Ven L, Godbout L (eds) Cellulose. IntechOpen. https://doi.org/10.5772/55178
Siirola JJ (2014) The impact of shale gas in the chemical industry. AIChE J 60:810–819. https://doi.org/10.1002/aic.14368
Sjöström E (1993) Wood chemistry: fundamentals and applications. Gulf Professional Publishing, Houston
Stadler RH, Robert F, Riediker S et al (2004) In-depth mechanistic study on the formation of acrylamide and other vinylogous compounds by the Maillard reaction. J Agric Food Chem 52:5550–5558. https://doi.org/10.1021/jf0495486
Tingaut P, Hauert R, Zimmermann T (2011) Highly efficient and straightforward functionalization of cellulose films with thiol-ene click chemistry. J Mater Chem 21:16066–16076. https://doi.org/10.1039/C1JM11620G
Tous E, Weber HM, Lee MH, Koomalsingh KJ, Shuto T, Kondo N, Gorman JH, Lee D, Gorman RC, Burdick JA (2012) Tunable hydrogel-microsphere composites that modulate local inflammation and collagen bulking. Acta Biomater 8:3218–3227
Tyl RW, Friedman MA (2003) Effects of acrylamide on rodent reproductive performance. Reprod Toxicol 17:1–13
Vaca-Garcia C, Thiebaud S, Borredon ME, Gozzelino G (1998) Cellulose esterification with fatty acids and acetic anhydride in lithium chloride/N,N-dimethylacetamide medium. J Am Oil Chem Soc 75:315–319. https://doi.org/10.1007/s11746-998-0047-2
Vaddiraju S, Wang Y, Qiang L et al (2012) Microsphere erosion in outer hydrogel membranes creating macroscopic porosity to counter biofouling-induced sensor degradation. Anal Chem 84:8837–8845. https://doi.org/10.1021/ac3022423
Yoo Y, Youngblood JP (2016) Green one-pot synthesis of surface hydrophobized cellulose nanocrystals in aqueous medium. ACS Sustain Chem Eng 4:3927–3938. https://doi.org/10.1021/acssuschemeng.6b00781
Acknowledgments
The authors would like to thank David Rondeau and Brooke Springer for assistance with data collection and organization. The authors also would like to thank Grape (Hathiathep) Senkum for help with obtaining size exclusion data. This work was supported by National Science Foundation award #11A-1355457 to Maine EPSCoR at the University of Maine.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
McOscar, T.V.C., Gramlich, W.M. Hydrogels from norbornene-functionalized carboxymethyl cellulose using a UV-initiated thiol-ene click reaction. Cellulose 25, 6531–6545 (2018). https://doi.org/10.1007/s10570-018-2015-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10570-018-2015-9