Abstract
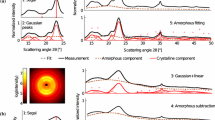
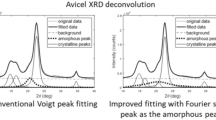
X-ray powder diffraction is one of the most commonly used methods in cellulose science. This technique is used to identify the cellulose allomorphs, their crystallinity, and the size of their crystallites. In this paper, a novel model is introduced that implicitly takes into account the shape and size of cellulose Iβ crystallites in the interpretation of powder diffractograms. Because of the limited amount of data in cellulose powder patterns, this model focuses on a small number of adjustable parameters. The method hypothesizes that cellulose Iβ crystallites are straight crystalline rods with superelliptical cross-sections. This superellipse is a parametric curve that can, for example, describe various crystallite shapes as rectangles or ellipses. Additionally, preferred orientation along the (0 0 1) crystallographic planes can be modelled using the March–Dollase approach. The simulated background has a semi-empirical form. An initial model comprised cellulose Iβ crystallites and the amorphous background. A second model comprised a biphasic distribution of crystallites and the same amorphous background. In this second model, large cellulose Iβ crystallites coexisted with more slender crystallites, usually less than 20 Å in lateral size. Cellulose IVI nanocrystals were selected as a modeling construct to represent these small and distorted forms of native cellulose. Both models produced simulations in excellent agreement with the experimental measurements.










Similar content being viewed by others
References
Ahvenainen P, Kontro I, Svedström K (2016) Comparison of sample crystallinity determination methods by X-ray diffraction for challenging cellulose I materials. Cellulose 23:1073–1086
Bansal P, Hall M, Realff MJ, Lee JH, Bommarius AS (2010) Multivariate statistical analysis of X-ray data from cellulose: a new method to determine degree of crystallinity and predict hydrolysis rates. Bioresour Technol 101:4461–4471
Battista OA, Coppick S, Howsmon JA, Morehead FF, Sisson WA (1956) Level-off degree of polymerization. Ind Eng Chem 48:333–335
Bayer EA, Chanzy H, Lamed R, Shoham Y (1998) Cellulose, cellulases and cellulosomes. Curr Opin Struct Biol 8:548–557
Bu L, Himmel ME, Crowley MF (2015) The molecular origins of twist in cellulose I-beta. Carbohydr Polym 125:146–152
De Figueiredo LP, Ferreira FF (2014) The Rietveld method as a tool to quantify the amorphous amount of microcrystalline cellulose. J Pharm Sci 103:1394–1399
Ding S-Y, Himmel ME (2006) The maize primary cell wall microfibril: a new model derived from direct visualization. J Agric Food Chem 54:597–606
Ding S-Y, Zhao S, Zeng Y (2013) Size, shape, and arrangement of native cellulose fibrils in maize cell walls. Cellulose 21:863–871
Dollase WA (1986) Correction of intensities for preferred orientation in powder diffractometry: application of the March model. J Appl Crystallogr 19:267–272
Dong S, Bortner MJ, Roman M (2016) Analysis of the sulfuric acid hydrolysis of wood pulp for cellulose nanocrystal production: a central composite design study. Ind Crops Prod 93:76–87
Driemeier C (2014) Two-dimensional Rietveld analysis of celluloses from higher plants. Cellulose 21:1065–1073
Driemeier C, Bragatto J (2013) Crystallite width determines monolayer hydration across a wide spectrum of celluloses isolated from plants. J Phys Chem B 117:415–421
Driemeier C, Calligaris GA (2010) Theoretical and experimental developments for accurate determination of crystallinity of cellulose I materials. J Appl Crystallogr 44:184–192
Driemeier C, Santos WD, Buckeridge MS (2012) Cellulose crystals in fibrovascular bundles of sugarcane culms: orientation, size, distortion, and variability. Cellulose 19:1507–1515
Duchemin BJC (2015) Mercerisation of cellulose in aqueous NaOH at low concentrations. Green Chem 17:3941–3947
Duchemin B, Newman R, Staiger M (2007) Phase transformations in microcrystalline cellulose due to partial dissolution. Cellulose 14:311–320
Duchemin B, Thuault A, Vicente A, Rigaud B, Fernandez C, Eve S (2012) Ultrastructure of cellulose crystallites in flax textile fibres. Cellulose 19:1837–1854
Duchemin B, Corre DL, Leray N, Dufresne A, Staiger MP (2015) All-cellulose composites based on microfibrillated cellulose and filter paper via a NaOH-urea solvent system. Cellulose 23:1–17
Fink H-P, Philipp B, Paul D, Serimaa R, Paakkari T (1987) The structure of amorphous cellulose as revealed by wide-angle X-ray scattering. Polymer 28:1265–1270
French AD (2014) Idealized powder diffraction patterns for cellulose polymorphs. Cellulose 21:885–896
French AD, Santiago Cintrón M (2012) Cellulose polymorphy, crystallite size, and the Segal Crystallinity Index. Cellulose 20:583–588
Gardiner ES, Sarko A (1985) Packing analysis of carbohydrates and polysaccharides. 16. The crystal structures of celluloses IVI and IVII. Can J Chem 63:173–180
Gjønnes J, Norman N (1958) The use of half width and position of the lines in the X-ray diffractograms of native cellulose to characterize the structural properties of the samples. Acta Chem Scand 12:2028–2033
Guinier A (1963) X-ray diffraction in crystals, imperfect crystals, and amorphous bodies. W. H. Freeman and Company, San Francisco
Hadden JA, French AD, Woods RJ (2013) Effect of microfibril twisting on theoretical powder diffraction patterns of cellulose Iβ. Cellulose 21:879–884
Hearle JW (1963) The development of ideas of fine structure. The Textile Institute, London
Hermans PH, Weidinger A (1948) Quantitative X-ray investigations on the crystallinity of cellulose fibers. A background analysis. J Appl Phys 19:491–506
Hosemann R, Hentschel MP, Baltacalleja FJ, Cabarcos EL, Hindeleh AM (1985) The alpha-star-constant, equilibrium state and bearing net planes in polymers, bio-polymers and catalysts. J Phys C-Solid State Phys 18:961–971
Isogai A (1989) Solid-state CP/MAS 13C NMR study of cellulose polymorphs. Macromolecules 22:3168–3172
Ju X, Bowden M, Brown EE, Zhang X (2015) An improved X-ray diffraction method for cellulose crystallinity measurement. Carbohydr Polym 123:476–481
Kouris M, Ruck H, Mason SG (1958) The effect of water removal on the crystallinity of cellulose. Can J Chem 36:931–948
Kulshreshtha AK (1979) A review of the literature on the formation of cellulose IV, its structure, and its significance in the technology of rayon manufacture. J Text Inst 70:13–18
Langan P, Sukumar N, Nishiyama Y, Chanzy H (2005) Synchrotron X-ray structures of cellulose Iβ and regenerated cellulose II at ambient temperature and 100 K. Cellulose 12:551–562
Larsson PT, Wickholm K, Iversen T (1997) A CP/MAS13C NMR investigation of molecular ordering in celluloses. Carbohydr Res 302:19–25
Lehtiö J, Sugiyama J, Gustavsson M, Fransson L, Linder M, Teeri TT (2003) The binding specificity and affinity determinants of family 1 and family 3 cellulose binding modules. Proc Natl Acad Sci USA 100:484–489
Leppänen K, Andersson S, Torkkeli M, Knaapila M, Kotelnikova N, Serimaa R (2009) Structure of cellulose and microcrystalline cellulose from various wood species, cotton and flax studied by X-ray scattering. Cellulose 16:999–1015
Li Q, Renneckar S (2011) Supramolecular structure characterization of molecularly thin cellulose I nanoparticles. Biomacromol 12:650–659
Macrae CF, Bruno IJ, Chisholm JA, Edgington PR, McCabe P, Pidcock E, Rodriguez-Monge L, Taylor R, Streek JV, Wood PA (2008) Mercury CSD 2.0–new features for the visualization and investigation of crystal structures. J Appl Crystallogr 41:466–470
Maier G, Zipper P, Stubicar M, Schurz J (2005) Amorphization of different cellulose samples by ball milling. Cell Chem Tech 39:167–177
Mann J (1962) Modern methods of determining crystallinity in cellulose. Pure Appl Chem 5:91–105
Mitra GB, Mukherjee PS (1980) X-ray diffraction study of fibrous polymers. I. Degree of paracrystallinity—a new parameter for characterizing fibrous polymers. Polymer 21:1403–1409
Mitra GB, Mukherjee PS (1981) Application of the method of moments to X-ray diffraction line profiles from paracrystals: native cellulose fibres. J Appl Crystallogr 14:421–431
Mukherjee PS, Mitra GB (1983) Anisotropy of X-ray structural parameters of the three phase model in cellulose I. Polymer 24:525–528
Nam S, French AD, Condon BD, Concha M (2016) Segal crystallinity index revisited by the simulation of X-ray diffraction patterns of cotton cellulose Iβ and cellulose II. Carbohydr Polym 135:1–9
Newman RH (1999) Estimation of the lateral dimensions of cellulose crystallites using 13C NMR signal strengths. Solid State Nucl Magn Reson 15:21–29
Newman RH (2008) Simulation of X-ray diffractograms relevant to the purported polymorphs cellulose IVI and IVII. Cellulose 15:769–778
Newman RH, Hill SJ, Harris PJ (2013) Wide-angle X-Ray scattering and solid-state nuclear magnetic resonance data combined to test models for cellulose microfibrils in mung bean cell walls. Plant Physiol 163:1558–1567
Niimura H, Yokoyama T, Kimura S, Matsumoto Y, Kuga S (2010) AFM observation of ultrathin microfibrils in fruit tissues. Cellulose 17:13–18
Nishiyama Y, Langan P, Chanzy H (2002) Crystal structure and hydrogen-bonding system in cellulose Ibeta from synchrotron X-ray and neutron fibre diffraction. J Am Chem Soc 124:9074–9082
Nishiyama Y, Johnson GP, French AD, Forsyth VT, Langan P (2008) Neutron crystallography, molecular dynamics, and quantum mechanics studies of the nature of hydrogen bonding in cellulose Iβ. Biomacromol 9:3133–3140
Nishiyama Y, Johnson GP, French AD (2012) Diffraction from nonperiodic models of cellulose crystals. Cellulose 19:319–336
Oehme DP, Doblin MS, Wagner J, Bacic A, Downton MT, Gidley MJ (2015) Gaining insight into cell wall cellulose macrofibril organisation by simulating microfibril adsorption. Cellulose 22:3501–3520
Patterson AL (1939) The Scherrer formula for X-ray particle size determination. Phys Rev 56:978–982
Popa NC, Balzar D (2008) Size-broadening anisotropy in whole powder pattern fitting. Application to zinc oxide and interpretation of the apparent crystallites in terms of physical models. J Appl Crystallogr 41:615–627
Preston RD (1974) The physical biology of plant cell walls. Chapman and Hall, London
Rietveld HM (1969) A profile refinement method for nuclear and magnetic structures. J Appl Crystallogr 2:65–71
Ruland W (1964) The separation of coherent and incoherent Compton X-ray scattering. Br J Appl Phys 15:1301
Segal L, Nelson ML, Conrad CM (1951) Experiments on the reduction of the crystallinity of cotton cellulose. J Phys Colloid Chem 55:325–336
Segal L, Creely JJ, Martin AE, Conrad CM (1959) An empirical method for estimating the degree of crystallinity of native cellulose using the X-ray diffractometer. Text Res J 29:786–794
Sonneveld EJ, Visser JW (1975) Automatic collection of powder data from photographs. J Appl Crystallogr 8:1–7
Spraget HWG (1989) Spreadsheet simulation of X-ray powder diffraction. Comput Educ 13:101–108
Stephens PW (1999) Phenomenological model of anisotropic peak broadening in powder diffraction. J Appl Crystallogr 32:281–289
Takahashi M, Ookubo M (1994) CP/MAS 13C NMR and WAXS studies on the effects of starting cellulose materials on transition between cellulose polymorphs. Kobunshi Ronbunshu 51:107–113
Thomas LH, Forsyth VT, Šturcová A, Kennedy CJ, May RP, Altaner CM, Apperley DC, Wess TJ, Jarvis MC (2013) Structure of cellulose microfibrils in primary cell walls from collenchyma. Plant Physiol 161:465–476
Thygesen A, Oddershede J, Lilholt H, Thomsen AB, Ståhl K (2005) On the determination of crystallinity and cellulose content in plant fibres. Cellulose 12:563–576
Tsuda Y, Mukoyama S (1957) The formation of cellulose IV in the viscose spinning. Bull Chem Soc Jpn 30:718–720
Viëtor RJ, Mazeau K, Lakin M, Pérez S (2000) A priori crystal structure prediction of native celluloses. Biopolymers 54:342–354
Vonk CG (1973) Computerization of Ruland’s X-ray method for determination of the crystallinity in polymers. J Appl Crystallogr 6:148–152
Wada M, Sugiyama J, Okano T (1993) Native celluloses on the basis of two crystalline phase (Iα/Iβ) system. J Appl Polym Sci 49:1491–1496
Wada M, Okano T, Sugiyama J (2001) Allomorphs of native crystalline cellulose I evaluated by two equatoriald-spacings. J. Wood Sci 47:124–128
Wada M, Heux L, Sugiyama J (2004) Polymorphism of cellulose I family: reinvestigation of cellulose IVI. Biomacromolecules 5:1385–1391
Wakelin JH, Virgin HS, Crystal E (1959) Development and comparison of two X-Ray methods for determining the crystallinity of cotton cellulose. J Appl Phys 30:1654–1662
Wang Y, Chan SLI, Amal R, Shen YR, Kiatkittipong K (2011) XRD anisotropic broadening of nano-crystallites. International Centre for Diffraction Data. http://www.icdd.com/resources/axa/VOL54/V54_12.pdf. Accessed 12 May 2017
Wickholm K, Larsson PT, Iversen T (1998) Assignment of non-crystalline forms in cellulose I by CP/MAS 13C NMR spectroscopy. Carbohydr Res 312:123–129
Yoshiharu N, Shigenori K, Masahisa W, Takeshi O (1997) Cellulose microcrystal film of high uniaxial orientation. Macromolecules 30:6395–6397
Yue Y, Han J, Han G, Zhang Q, French AD, Wu Q (2015) Characterization of cellulose I/II hybrid fibers isolated from energycane bagasse during the delignification process: morphology, crystallinity and percentage estimation. Carbohydr Polym 133:438–447
Zugenmaier P (2001) Conformation and packing of various crystalline cellulose fibers. Prog Polym Sci 26:1341–1417
Acknowledgments
The author thanks Editor-in-Chief Alfred French and the anonymous reviewers for their helpful comments on this paper.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Duchemin, B. Size, shape, orientation and crystallinity of cellulose Iβ by X-ray powder diffraction using a free spreadsheet program. Cellulose 24, 2727–2741 (2017). https://doi.org/10.1007/s10570-017-1318-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10570-017-1318-6