Abstract
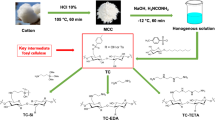
A series of dialdehyde microcrystalline cellulose (DAMC) were prepared by NaIO4 oxidation of microcrystalline cellulose (MCC), and their physico-chemical properties, antioxidant activity, and antibacterial activity were further investigated. The results of scanning electron microscopy indicated that the particle size of DAMC became shorter than that of unoxidized MCC, and the surface erosion of particles was observed. The degree of crystallinity and thermal stability of DAMCs decreased as their aldehyde contents increased. The formation of aldehyde and hemiacetal groups of the DAMC was confirmed by Fourier transform infrared spectroscopy spectra. The antioxidant activity assays demonstrated that the DAMC with 6.59 mmol/g of aldehyde content showed the highest scavenging effect on DPPH, ABTS, and hydroxyl radicals with half-inhibitory concentration (IC50) values of 5.9, 5.6 and 8.1 mg/mL; its reducing power was also the best among the three samples. The antimicrobial activity test results showed that DAMCs with high aldehyde contents (more than 5.14 mmol/g) exhibited the strongest antibacterial activity against S. aureus, B. subtilis, E. coli and S. typhimurium, and their MIC values were 15, 15, 15, and 30 mg/mL, respectively. Our results proved that the physico-chemical properties of DAMC may have great influence on its antioxidant and antibacterial capacities.





Similar content being viewed by others
References
Andresen M, Johansson L-S, Tanem BS, Stenius P (2006) Properties and characterization of hydrophobized microfibrillated cellulose. Cellulose 13:665–677
Bansal M, Chauhan GS, Kaushik A, Sharmaca A (2016) Extraction and functionalization of bagasse cellulose nanofibres to Schiff-base based antimicrobial membranes. Int J Biol Macromol 91:887–894
Berlioz S, Molina-Boisseau S, Nishiyama Y, Heux L (2009) Gas-phase surface esterification of cellulose microfibrils and whiskers. Biomacromolecules 10:2144–2151
Dinand E, Chanzy H, Vignon MR (1999) Suspensions of cellulose microfibrils from sugar beet pulp. Food Hydrocoll 13:275–283
El Meligy MG, El Rafie S, Abu-Zied KM (2005) Preparation of dialdehyde cellulose hydrazone derivatives and evaluating their efficiency for sewage wastewater treatment. Desalination 173:33–44
Giese EC, Gascon J, Anzelmo G, Barbosa AM, Cunha MAA, Dekker RF (2015) Free-radical scavenging properties and antioxidant activities of botryosphaeran and some other β-d-glucans. Int J Biol Macromol 72:125–130
Hofreiter BT, Alexander BH, Wolff IA (1955) Rapid estimation of dialdehyde content of periodate oxystarch through quantitative alkali consumption. Anal Chem 27:1930–1931
Hou QX, Liu W, Liu ZH, Duan B, Bai LL (2008) Characteristics of antimicrobial fibers prepared with wood periodate oxycellulose. Carbohydr Polym 74:235–240
Hubbell CA, Ragauskas AJ (2010) Effect of acid-chlorite delignification on cellulose degree of polymerization. Bioresour Technol 101:7410–7415
Jin LQ, Sun QC, Xu QH, Xu YJ (2015) Adsorptive removal of anionic dyes from aqueous solutions using microgel based on nanocellulose and polyvinylamine. Bioresour Technol 197:348–355
Kanth SV, Ramaraj A, Rao RJ, Nair BU (2009) Stabilization of type I collagen using dialdehyde cellulose. Process Biochem 44:869–874
Keshk MASS, Ramadan AM, Bondock S (2015) Physicochemical characterization of novel Schiff bases derived from developed bacterial cellulose 2,3-dialdehyde. Carbohydr Polym 127:246–251
Kim U-J, Kuga S (2001) Thermal decomposition of dialdehyde cellulose and its nitrogen-containing derivatives. Thermochim Acta 369:79–85
Kim U-J, Kuga S, Wada M, Okano T, Kondo T (2000) Periodate oxidation of crystalline cellulose. Biomacromolecules 1:488–492
Kim U-J, Wada M, Kuga S (2004) Solubilization of dialdehyde cellulose by hot water. Carbohydr Polym 56:7–10
Klemm D, Schumann D, Kramer F, Heßler N, Hornung M, Schmauder HP (2006) Nanocelluloses as innovative polymers in research and application. Adv Polym Sci 5:49–96
Kumari S, Mankotia D, Chauhan GS (2016) Crosslinked cellulose dialdehyde for Congo red removal from its aqueous solutions. J Environ Chem Eng 4:1126–1136
Laçin NT (2014) Development of biodegradable antibacterial cellulose based hydrogel membranes for wound healing. Int J Biol Macromol 67:22–27
Li J, Wan YZ, Li LF, Liang H, Wang JH (2009) Preparation and characterization of 2,3-dialdehyde bacterial cellulose for potential biodegradable tissue engineering scaffolds. Mater Sci Eng C 29:1635–1642
Li H, Wu B, Mu C, Lin W (2011) Concomitant degradation in periodate oxidation of carboxymethyl cellulose. Carbohydr Polym 84:881–886
Li XC, Lin J, Gao YX, Han WJ, Chen DF (2012) Antioxidant activity and mechanism of Rhizoma Cimicifugae. Chem Cent J 6(1):1–10
Liu X, Wang L, Song X, Song H, Zhao JR, Wang S (2012) A kinetic model for oxidative degradation of bagasse pulp fiber by sodium periodate. Carbohydr Polym 90:218–223
Luo CC, Wang H, Chen Y (2015) Progress in modification of cellulose and application. Chem Ind Eng Prog 34(3):767–773
National Committee for Clinical Laboratory Standards (2006) Performance standards for antimicrobial disk susceptibility tests: approved standards. 11th edn. National Committee for Clinical Laboratory Standards
Peng F, Ren JL, Xu F, Bian J, Peng P, Sun RC (2009) Comparative study of hemicelluloses obtained by graded ethanol precipitation from sugarcane bagasse. J Agric Food Chem 57(14):6305–6317
Pietrucha K, Safandowska M (2015) Dialdehyde cellulose-crosslinked collagen and its physicochemical properties. Process Biochem 50:2105–2111
Rangel-Vázquez NA, Guilbert-García E, Salgado-Delgado R, Rubio-Rosas E, Hernandez EG, Vargas-Galarza Z, Crispín-Espino I (2010) Synthesis and characterization of chitosan coated dialdehyde cellulose with potential antimicrobial behavior. J Mater Sci Eng 4(12):62–67
Sarac N, Sen B (2014) Antioxidant, mutagenic, antimutagenic activities, and phenolic compounds of Liquidambar orientalis Mill. var. orientalis. Ind Crop Prod 53:60–64
Segal L, Creely JJ, Martin AE, Conrad CM (1959) An empirical method for estimating the degree of crystallinity of native cellulose using the X-ray diffractometer. Text Res J 29:786–794
Shen GY, Zhang XY, Shen YM, Zhang SB, Fang L (2015) One-step immobilization of antibodies for α-1-fetoprotein immunosensor based on dialdehyde cellulose/ionic liquid composite. Anal Biochem 471:38–43
Sirviö J, Hyväkkö U, Liimatainen H, Niinimäki J, Hormi O (2011) Periodate oxidation of cellulose at elevated temperatures using metal salts as cellulose activators. Carbohydr Polym 83:1293–1297
Song L, Sang YJ, Cai LM, Shi YC, Farrah SR, Baney RH (2010) The effect of cooking on the antibacterial activity of the dialdehyde starch suepnsions. Starch/Stärke 62:458–466
Song L, Farrah SR, Baney RH (2011) Bacterial inactivation kinetics of dialdehyde starch aqueous suspension. Polymers 3:1902–1910
Sun B, Hou QX, Liu ZH, Ni YH (2015) Sodium periodate oxidation of cellulose nanocrystal and its application as a paper wet strength additive. Cellulose 22:1135–1146
Tang A, Zhang H, Chen G, Xie G, Liang W (2005) Influence of ultrasound treatment on accessibility and regioselective oxidation reactivity of cellulose. Ultrason Sonochem 12:467–472
Turbak AF, Snyder FW, Sandberg KRJ (1983) Microfibrillated cellulose, a new cellulose product: properties, uses and commercial potential. J Appl Polym Sci 7:815–827
Varma AJ, Chavan VB (1995) A study of crystallinity changes in oxidised celluloses. Polym Degrad Stab 49:245–250
Varma AJ, Kulkarni MP (2002) Oxidation of cellulose under controlled conditions. Polym Degrad Stab 77:25–27
Verma V, Verma P, Ray P, Ray AR (2008) 2,3-Dihydrazone cellulose: prospective material for tissue engineering scaffolds. Mater Sci Eng C 28:1441–1447
Vicini S, Princi E, Luciano G, Franceschi E, Pedemonte E, Oldak D, Kaczmarek H, Sionkowska A (2004) Thermal analysis and characterization of cellulose oxidised with sodium methaperiodate. Thermochim Acta 418:123–130
Yildirim A, Mavi A, Kara AA (2001) Determination of antioxidant and antimicrobial activities of Rumex crispus L. extracts. J Agric Food Chem 49:4083–4089
Yu JG, Chang PR, Ma XF (2010) The preparation and properties of dialdehyde starch and thermoplastic dialdehyde starch. Carbohydr Polym 79:296–300
Yuen SN, Choi SM, Phillips DL, Ma CY (2009) Raman and FTIR spectroscopic study of carboxymethylated nonstarch polysaccharides. Food Chem 114:1091–1098
Zaman M, Xiao H, Chibante F, Ni Y (2012) Synthesis and characterization of cationically modified nanocrystalline cellulose. Carbohydr Polym 89:163–170
Zhang Y, Lu X, Fu Z, Wang Z, Zhang J (2011) Sulphated modification of a polysaccharide obtained from fresh persimmon (Diospyros kaki L.) fruit and antioxidant activities of the sulphated derivatives. Food Chem 127:1084–1090
Zhang LM, Zhang S, Dong F, Cai WT, Shan J, Zhang XB, Man SL (2014a) Antioxidant activity and in vitro digestibility of dialdehyde starches as influenced by their physical and structural properties. Food Chem 149:296–301
Zhang XY, Shen GY, Sun SY, Shen YM, Zhang CX, Xiao AG (2014b) Direct immobilization of antibodies on dialdehyde cellulose film for convenient construction of an electrochemical immunosensor. Sens Actuators B 200:304–309
Acknowledgments
This study was supported by the National Natural Science Foundation of China (Project No. 31271809). The authors thank Prof. Haiyan Du (School of Material Science and Engineering, Tianjin University, China) for her helpful assistance in the experiment.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Zhang, L., Ge, H., Xu, M. et al. Physicochemical properties, antioxidant and antibacterial activities of dialdehyde microcrystalline cellulose. Cellulose 24, 2287–2298 (2017). https://doi.org/10.1007/s10570-017-1255-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10570-017-1255-4