Abstract
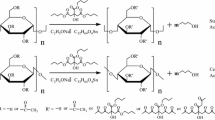
Biodegradable blends were prepared from cellulose and poly (vinyl alcohol) (PVA) using the ionic liquid (IL) solvent, 1-butyl-3-methylimidazolium chloride. The blends were regenerated into films, fibers and rectangular blocks. The films showed optical transparency throughout the entire composition of the blends. The infrared spectroscopic experiments proved the existence of intermolecular hydrogen bonding interactions between the hydroxyl groups of cellulose and PVA. The miscibility between cellulose and PVA lead to increase in glass transition temperature (T g) and of decrease in crystallinity of the blends. The T g-composition data showed a negative deviation from Fox predictions, however fit well with BCKV model. The addition of PVA improved the tensile strength and elongation at break, considerably plasticizing cellulose. The blends can be degraded completely in soil. Moreover, the IL was completely recycled with high yield after the processing.









Similar content being viewed by others
References
Anastas PT, Warner JC (1998) Green chemistry: theory and practice. Oxford University Press, NY
Braun G, Kovacs AJ (1965) In: Prins JA (ed) Proceedings of the international conference, Delft, July, 1964. North-Holland Pub. Co, Amsterdam
Brekner M-J, Schneider HA, Cantow H-J (1988) Approach to the composition dependence of the glass transition temperature of compatible polymer blends. 2 The effect of local chain orientation. Die Makromolekulare Chemie 189:2085
Coleman MM, Painter PC (1995) Hydrogen bonded polymer blends. Prog Polym Sci 20:1
Couchman PR, Karasz FE (1978) A classical thermodynamic discussion of the effect of composition on glass-transition temperatures. Macromolecules 11:117
De Kesel C, Lefevre C, Nagy J, David C (1999) Blends of polycaprolactone with polyvinylalcohol: a DSC, optical microscopy and solid state NMR study. Polymer 40:1969
Dupont J, de Souza RF, Suarez PAZ (2002) Ionic liquid (molten salt) phase organometallic catalysis. Chem Rev 102:3667
Feldstein MM, Roos A, Chevallier C, Creton C, Dormidontova EE (2003) Relation of glass transition temperature to the hydrogen bonding degree and energy in poly(N-vinyl pyrrolidone) blends with hydroxyl-containing plasticizers: 3. Analysis of two glass transition temperatures featured for PVP solutions in liquid poly(ethylene glycol). Polymer 44:1819
Fox TG (1956) Influence of diluent and of copolymer composition on the glass temperature of a polymer system. Bull Am Phys Soc 1:123
Garrett RH, Grisham CM (2002) Biochemistry, 2nd edn. Higher Education Press, Beijing
Gordon M, Taylor JSJ (1952) Ideal copolymers and the second-order transitions of synthetic rubbers. i. non-crystalline copolymers. Appl Chem 2:493
Hameed N, Guo Q (2009) Natural wool/cellulose acetate blends regenerated from the ionic liquid 1-butyl-3-methylimidazolium chloride. Carbohydr Polym 78:999
Hameed N, Guo Q (2010) Blend films of natural wool and cellulose prepared from an ionic liquid. Cellulose 17:803
Hameed N, Guo Q, Tay FH, Kazarian SG (2011) Blends of cellulose and poly (3-hydroxybutyrate-co-3-hydroxyvalerate) prepared from the ionic liquid 1-butyl-3-methylimidazolium chloride. Carbohydr Polym 86:94
Imam SH, Greene RV, Zaidi BR (eds) (1999) Biopolymers: utilizing nature’s advanced materials. ACS symposium series, No. 723. American Chemical Society, Washington, DC
Kaplan DL (1998) Biopolymers from renewable resources. Springer, New York
Konno H, Taylor LSJ (2006) Influence of different polymers on the crystallization tendency of molecularly dispersed amorphous felodipine. Pharm Sci 95:2692
Kubat J, Pattyrante C (1967) Transition in cellulose in the vicinity of −30° C. Nature 215:390
Kubo S, Kadla JF (2003) The formation of strong intermolecular interactions in immiscible blends of poly (vinyl alcohol) (PVA) and lignin. Biomacromolecules 4:561
Kwei TKJ (1984) The effect of hydrogen bonding on the glass transition temperatures of polymer mixtures. Polym Lett Ed 22:307
Mayer JM, Kaplan DL, Stote RE, Dixon KL, Shupe AE, Allen AL, McCassie JE (1996) ACS symposium series, chap 13, vol 627, p 159
Mikhailov GP, Artyukhov AI, Shevele VA (1976) Molecular mobility in cellulose and its derivatives studied by dielectric and nuclear magnetic resonance methods. Vysokomol Soyed A11:553
Nishi T, Wang TT (1975) Melting point depression and kinetic effects of cooling on crystallization in poly (vinylidene fluoride)–poly (methyl methacrylate) mixtures. Macromolecules 8:909
Nishio Y, Manley RSJ (1988) Cellulose-poly (vinyl alcohol) blends prepared from solutions in N,N-dimethylacetamide-lithium chloride. Macromolecules 21:1270
Nishio Y, Haratani T, Takahashi T, Manley RS (1989) Miscibility and orientation behavior of poly (vinyl alcohol)/poly (vinyl pyrrolidone) blends. Macromolecules 22:2547
Paranhos CM, Soares BG, Oliveira RN, Pessan LA (2007) Poly (vinyl alcohol)/clay‐based nanocomposite hydrogels: swelling behavior and characterization. Macromol Mater Eng 292:620
Parvin F, Khan MA, Saadat AHM, Khan MAH, Islam JMM, Ahmed M, Gafur MAJ (2011) Preparation and characterization of gamma irradiated sugar containing starch/poly (vinyl alcohol)-based blend films. Polym Environ 19:1013
Ray D, Roy P, Sengupta S, Sengupta S, Mohanty A, Misra MJ (2009) A study of physicomechanical and morphological properties of starch/poly (vinylalcohol) based films. Polym Environ 17:56
Rim PB, Runt JP (1984) Melting point depression in crystalline/compatible polymer blends. Macromolecules 17:1520
Sathasivam K, Mas Haris MRH, Noorsal K (2010) The preparation and characterization of esterified banana trunk fibers/poly (vinyl alcohol) blend film. Polym Plast Tech Eng 49:1378
Sathigari SK (2011) Single step preparation and deagglomeration of itraconazole microflakes by supercritical antisolvent method for dissolution enhancement. Auburn University, Auburn
Swamy YB, Venkata Prasad C, Reedy C, Mallikarjuna B, Rao K, Subha M (2011) Interpenetrating polymer network microspheres of hydroxy propyl methyl cellulose/poly (vinyl alcohol) for control release of ciprofloxacin hydrochloride. Cellulose 18:349
Takahashi Y, Matsunaga H (1991) Crystal structure of native cellulose. Macromolecules 24:3968
Turner MB, Spear SK, Holbrey JD, Rogers RD (2004) Production of bioactive cellulose films reconstituted from ionic liquids. Biomacromolecules 5:1379
Witt MA, Barra GMO, Bertolino JR, Pires ATNJ (2010) Crosslinked chitosan/poly (vinyl alcohol) blends with proton conductivity characteristic. Braz Chem Soc 21:1692
Xiong R, Hameed N, Guo Q (2012) Cellulose/polycaprolactone blends regenerated from ionic liquid 1-butyl-3-methylimidazolium chloride. Carbohydr Polym 90:575
Zhang H, Wu J, Zhang J, He J (2005) 1-Allyl-3-methylimidazolium chloride room temperature ionic liquid: a new and powerful nonderivatizing solvent for cellulose. Macromolecules 38:8272
Zhou WY, Guo B, Liu M, Liao R, Rabie ABM, Jia DJ (2010) Poly(vinyl alcohol)/halloysite nanotubes bionanocomposite films: properties and in vitro osteoblasts and fibroblasts response. Biomed Mater Res Part A 93A:1574
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Hameed, N., Xiong, R., Salim, N.V. et al. Fabrication and characterization of transparent and biodegradable cellulose/poly (vinyl alcohol) blend films using an ionic liquid. Cellulose 20, 2517–2527 (2013). https://doi.org/10.1007/s10570-013-0017-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10570-013-0017-1