Abstract
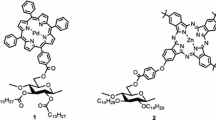
Langmuir-Blodgett (LB) films containing porphyrin molecules were fabricated by use of 6-O-dihydrophytylcellulose (DHPC) toward anodic photocurrent generation systems. To suppress the porphyrin aggregation, two different approaches were applied: (1) mixing a low-molecular-weight porphyrin having a diterpenoid carbon skeleton (DPor) with DHPC as a matrix (matrix fabrication) and (2) bonding porphyrin molecules to the hydroxyl groups of DHPC covalently, converting into 6-O-dihydrophytyl-2,3-di-O-[p-(10,15,20-triphenyl-5-porphyrinyl)-benzoyl]cellulose as a scaffold (scaffold fabrication). The structure and film properties of the monolayers and the LB films were investigated by the surface pressure (π)–area (A) isotherm measurements, atomic force microscopy, UV–Vis spectroscopy, and absorption dichroism measurements. The porphyrin aggregation in the LB film could be well suppressed only by the scaffold fabrication, leading to the improvement of the photocurrent quantum yields. The efficient photocurrent performance can be demonstrated by the isolation and the parallel orientation of porphyrin moieties due to the cellulose rigid scaffold.









Similar content being viewed by others
References
Aoki A, Abe Y, Miyashita T (1999) Effective photoinduced electron transfer in hetero-deposited redox polymer LB films. Langmuir 15:1463–1469
Brabec CJ, Sariciftci NS, Hummelen JC (2001) Plastic solar cells. Adv Funct Mater 11:15–26
Choudhury B, Weedon AC, Bolton JR (1998) Effects of molecular organization on photophysical behavior. 2. Photoelectrochemical and photocurrent quantum yield studies of the Langmuir-Blodgett monolayers of some surfactant porphyrins. Langmuir 14:6199–6206
Elemans JAAW, Hameren R, Nolte RJM, Rowan AE (2006) Molecular materials by self-assembly of porphyrins, phthalocyanines, and perylenes. Adv Mater 18:1251–1266
Fujihira M, Yamada H (1988) Molecular photodiodes consisting of unidirectionally oriented amphathic acceptor-sensitizer-donor triads. Thin Solid Films 160:125–132
Ifuku S, Tsujii Y, Kamitakahara H, Takano T, Nakatsubo F (2005) Preparation and characterization of redox cellulose Langmuir-Blodgett films containing a ferrocene derivative. J Polym Sci A Polym Chem 43:5023–5031
Imahori H, Fukuzumi S (2004) Porphyrin- and fullerene-based molecular photovoltaic devices. Adv Funct Mater 14:525–536
Ishiwatari M, Yamada K, Ishiwatari R (2000) Pyrolytic formation of C19 isoprenoid hydrocarbons from dihydrophytol: in relation to the genesis of pristane in petroleum. Chem Lett 2000:206–207
Itoh T, Yano K, Inada Y, Fukushima Y (2002) Photostabilized chrolophyll a in mesoporous silica: absorption properties and photoreduction activity of chlorophyll a. J Am Chem Soc 124:13437–13441
Kadish KM, Morrison MM (1976) Solvent and substituent effects on the redox reactions of para-substituted tetraphenylporphyrin. J Am Chem Soc 98:3326–3328
Kondo T, Uosaki K (2007) Self-assembled monolayers (SAMs) with photo-functionalities. J Photochem Photobiol C 8:1–17
Konishi T, Ikeda A, Asai M, Hatano T, Shinkai S, Fujitsuka M, Ito O, Tsuchiya Y, Kikuchi J-I (2003) Improvement of quantum yields for photoinduced energy/electron transfer by isolation of self-aggregative zinc tetraphenyl porphyrin-pendant polymer using cyclodextrin inclusion in aqueous solution. J Phys Chem B 107:11261–11266
Konishi T, Ikeda A, Shinkai S (2005) Supramolecular design of photocurrent-generating devices using fullerenes aimed at modelling artificial photosynthesis. Tetrahedron 61:4881–4899
Li C, Imae T (2003) Protoporphyin IX zinc(II) organization at the air/water interface and its Langmuir-Blodgett films. Langmuir 19:779–784
Matile S, Berova N, Nakanishi K, Fleischhauer J, Woody RW (1996) Structural studies by exciton coupled circular dichroism over a large distance: porphyrin derivatives of steroids, dimeric steroids, and brevetoxin B-. J Am Chem Soc 118:5198–5206
Miyashita T (1993) Recent studies on functional ultrathin polymer films prepared by the Langmuir-Blodgett technique. Prog Polym Sci 18:263–294
Ogi T, Ito S (2006) Migration and transfer of excitation energy in homogeneously dispersed porphyrin monolayers prepared from amphiphilic copolymers. Thin Solid Films 500:289–295
Petty MC (1996) Langmuir-Blodgett films: an introduction. The Press Syndicate of the University of Cambridge, New York
Redl FX, Lutz M, Daub J (2001) Chemistry of porphyrin-appended cellulose strands with a helical structure: spectroscopy, electrochemistry, and in situ circular dichroism. Chem Eur J 7:5350–5358
Sakakibara K, Ifuku S, Tsujii Y, Kamitakahara H, Takano T, Nakatsubo F (2006) Langmuir-Blodgett films of a novel cellulose derivative with dihydrophytyl group: the ability to anchor β-carotene molecules. Biomacromolecules 7:1960–1967
Sakakibara K, Kamitakahara H, Takano T, Nakatsubo F (2007a) Redox-active cellulose Langmuir-Blodgett films containing β-carotene as a molecular wire. Biomacromolecules 8:1657–1664
Sakakibara K, Ogawa Y, Nakatsubo F (2007b) First cellulose Langmuir-Blodgett films towards photocurrent generation systems. Macromol Rapid Commun 28:1270–1275
Sereno L, Silber JJ, Otero L, Bohorquez MDV, Moore AL, Moore TA, Gust D (1996) Photoelectrochemistry of Langmuir-Blodgett films of carotenoid pigments on ITO electrodes. J Phys Chem 100:814–821
Taniguchi T, Fukasawa Y, Miyashita T (1999) Photoelectrochemical response of polymer Langmuir-Blodgett films containing tris(2,2’-bipyridine)ruthenium(II) complex. J Phys Chem B 103:1920–1924
Tomé JPC, Neves MGPMS, Tomé AC, Cavaleiro JAS, Mendonça AF, Pegado IN, Duarte R, Valdeira ML (2005) Synthesis of glycoporphyrin derivatives and their antiviral activity against herpes simplex virus types 1 and 2. Bioorg Med Chem 13:3878–3888
Yoneyama M, Sugi M, Saito M, Ikegame K, Kuroda S-i, Iizima S (1986) Photoelectric properties of copper phthalocyanine Langmuir-Blodgett film. Jpn J Appl Phys 25:961–965
Yu J-H, Li F-Y, Wang X-S, Huang Y, Zhang B-W, Huang C-H, Cao Y (2002) Chain-length dependence of photoelectric conversion from a porphyrin monolayer modified electrode. Opt Mater 21:467–473
Acknowledgments
We are grateful to Prof. Tokuji Miyashita and Dr. Jun Matsui (Tohoku University) for useful suggestions for the photocurrent measurements. This investigation was supported by a Grant-in-Aid for Scientific Research (No. 17380107) from the Ministry of Education, Culture, Sports, Science and Technology, Japan. K.S. acknowledges the Research Fellowships of the Japan Society for the Promotion of Science (JSPS) for Young Scientists.
Author information
Authors and Affiliations
Corresponding author
Additional information
This paper was the subject of the Best Poster Award of the 235th edition of the ACS National Meeting, Cellulose and Renewable Materials.
Rights and permissions
About this article
Cite this article
Sakakibara, K., Nakatsubo, F. Fabrication of anodic photocurrent generation systems by use of 6-O-dihydrophytylcellulose as a matrix or a scaffold of porphyrins. Cellulose 15, 825–835 (2008). https://doi.org/10.1007/s10570-008-9228-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10570-008-9228-2