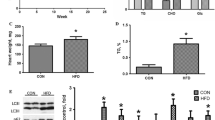
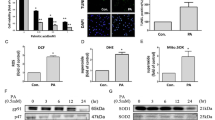
Abstract
Background and aims
The cardiovascular health benefits of eicosapentaenoic acid (EPA) have been demonstrated previously; however, the exact mechanism underlying them remains unclear. Our previous study found that lipotoxicity induced cardiomyocyte apoptosis via the inhibition of autophagy. Accordingly, in this study, we investigated whether EPA attenuated lipotoxicity-induced cardiomyocyte apoptosis through autophagy regulation. The role of EPA in mitochondrial dynamics was analyzed as well.
Methods
To explore how EPA protected against lipotoxicity-induced myocardial injury, cardiomyoblast (H9C2) cells were left untreated or were treated with 400 μM palmitic acid (PAM) and/or 80 μM EPA for 24 h.
Results
Excessive PAM treatment induced apoptosis. EPA reduced this PAM-induced apoptosis; however, EPA was unable to ameliorate the effects of PAM when autophagy was blocked by 3-methyladenine and bafilomycin A1. PAM blocked the autophagic flux, thus causing the accumulation of autophagosomes and acid vacuoles, whereas EPA restored the autophagic flux. PAM caused a decrease in polyunsaturated fatty acid (PUFA) content and an increase in saturated fatty acid content in the mitochondrial membrane, while EPA was incorporated in the mitochondrial membrane and caused a significant increase in the PUFA content. PAM also decreased the mitochondrial membrane potential, whereas EPA enhanced it. Finally, PAM elevated the expressions of autophagy-related proteins (LC3I, LC3II, p62) and mitochondrial fission protein (Drp1), whereas EPA inhibited their elevation under PAM treatment.
Conclusions
EPA reduces lipotoxicity-induced cardiomyoblast apoptosis through its effects on autophagy.





Similar content being viewed by others
Abbreviations
- Baf A1:
-
Bafilomycin A1
- EPA:
-
Eicosapentaenoic acid
- HFD:
-
High-fat diet
- 3-MA:
-
Methyladenine
- ∆ψ:
-
Mitochondrial membrane potential
- PAM:
-
Palmitic acid
- PUFA:
-
Polyunsaturated fatty acid
- SFA:
-
Saturated fatty acid
References
Akhmedov AT, Rybin V, Marin-Garcia J. Mitochondrial oxidative metabolism and uncoupling proteins in the failing heart. Heart Fail Rev. 2015;20:227–49. doi:10.1007/s10741-014-9457-4.
American Heart Association Nutrition C, Lichtenstein AH, Appel LJ, et al. Diet and lifestyle recommendations revision 2006: a scientific statement from the American Heart Association nutrition committee. Circulation. 2006;114:82–96. doi:10.1161/CIRCULATIONAHA.106.176158.
Chan YK, Sung HK, Jahng JW, et al. Lipocalin-2 inhibits autophagy and induces insulin resistance in H9c2 cells. Mol Cell Endocrinol. 2016;430:68–76. doi:10.1016/j.mce.2016.04.006.
Del Gobbo LC, Imamura F, Aslibekyan S, et al. Omega-3 polyunsaturated fatty acid biomarkers and coronary heart disease pooling project of 19 cohort studies. JAMA Intern Med. 2016;176:1155–66. doi:10.1001/jamainternmed.2016.2925.
Eng KE, Panas MD, Hedestam GBK, et al. A novel quantitative flow cytometry-based assay for autophagy. Autophagy. 2010;6:634–41.
Gottlieb E, Armour SM, Harris MH, et al. Mitochondrial membrane potential regulates matrix configuration and cytochrome c release during apoptosis. Cell Death Differ. 2003;10:709–17. doi:10.1038/sj.cdd.4401231.
Hom J, Sheu SS. Morphological dynamics of mitochondria—a special emphasis on cardiac muscle cells. J Mol Cell Cardiol. 2009;46:811–20. doi:10.1016/j.yjmcc.2009.02.023.
Hsu HC, Chen CY, Chen MF. N-3 polyunsaturated fatty acids decrease levels of doxorubicin-induced reactive oxygen species in cardiomyocytes—involvement of uncoupling protein UCP2. J Biomed Sci. 2014a;21:101. doi:10.1186/s12929-014-0101-3.
Hsu HC, Chen CY, Chiang CH, et al. Eicosapentaenoic acid attenuated oxidative stress-induced cardiomyoblast apoptosis by activating adaptive autophagy. Eur J Nutr. 2014b;53:541–7. doi:10.1007/s00394-013-0562-2.
Hsu HC, Chen CY, Lee BC, et al. High-fat diet induces cardiomyocyte apoptosis via the inhibition of autophagy. Eur J Nutr. 2016;55:2245–54. doi:10.1007/s00394-015-1034-7.
Ikeda Y, Shirakabe A, Brady C, et al. Molecular mechanisms mediating mitochondrial dynamics and mitophagy and their functional roles in the cardiovascular system. J Mol Cell Cardiol. 2015;78:116–22. doi:10.1016/j.yjmcc.2014.09.019.
Kaur G, Begg DP, Barr D, et al. Short-term docosapentaenoic acid (22:5 n-3) supplementation increases tissue docosapentaenoic acid, DHA and EPA concentrations in rats. Br J Nutr. 2010;103:32–7. doi:10.1017/S0007114509991334.
Li YY, Xiang Y, Zhang S, et al. Thioredoxin-2 protects against oxygen-glucose deprivation/reperfusion injury by inhibiting autophagy and apoptosis in H9c2 cardiomyocytes. Am J Transl Res. 2017;9:1471–82.
Liang Q, Kobayashi S. Mitochondrial quality control in the diabetic heart. J Mol Cell Cardiol. 2016;95:57–69. doi:10.1016/j.yjmcc.2015.12.025.
Lionetti L, Mollica MP, Donizzetti I, et al. High-lard and high-fish-oil diets differ in their effects on function and dynamic behaviour of rat hepatic mitochondria. PLoS One. 2014;9:e92753. doi:10.1371/journal.pone.0092753.
Liu J, Wang P, Zou L, et al. High-fat, low-carbohydrate diet promotes arrhythmic death and increases myocardial ischemia-reperfusion injury in rats. Am J Physiol Heart Circ Physiol. 2014;307:H598–608. doi:10.1152/ajpheart.00058.2014.
Monteiro JP, Oliveira PJ, Jurado AS. Mitochondrial membrane lipid remodeling in pathophysiology: a new target for diet and therapeutic interventions. Prog Lipid Res. 2013;52:513–28. doi:10.1016/j.plipres.2013.06.002.
Mozaffarian D, Wu JH. (n-3) fatty acids and cardiovascular health: are effects of EPA and DHA shared or complementary? J Nutr. 2012;142:614S–25S. doi:10.3945/jn.111.149633.
Poudyal H, Panchal SK, Ward LC, et al. Effects of ALA, EPA and DHA in high-carbohydrate, high-fat diet-induced metabolic syndrome in rats. J Nutr Biochem. 2013;24:1041–52. doi:10.1016/j.jnutbio.2012.07.014.
Schiattarella GG, Hill JA. Therapeutic targeting of autophagy in cardiovascular disease. J Mol Cell Cardiol. 2016;95:86–93. doi:10.1016/j.yjmcc.2015.11.019.
Sciarretta S, Volpe M, Sadoshima J. Mammalian target of rapamycin signaling in cardiac physiology and disease. Circ Res. 2014;114:549–64. doi:10.1161/CIRCRESAHA.114.302022.
Sekikawa A, Curb JD, Ueshima H, et al. Marine-derived n-3 fatty acids and atherosclerosis in Japanese, Japanese-American, and white men—a cross-sectional study. J Am Coll Cardiol. 2008;52:417–24. doi:10.1016/j.jacc.2008.03.047.
Sishi BJ, Loos B, van Rooyen J, et al. Autophagy upregulation promotes survival and attenuates doxorubicin-induced cardiotoxicity. Biochem Pharmacol. 2013;85:124–34. doi:10.1016/j.bcp.2012.10.005.
Stanley WC, Khairallah RJ, Dabkowski ER. Update on lipids and mitochondrial function: impact of dietary n-3 polyunsaturated fatty acids. Curr Opin Clin Nutr. 2012;15:122–6. doi:10.1097/Mco.0b013e32834fdaf7.
Sun R, Wang X, Liu Y, et al. Dietary supplementation with fish oil alters the expression levels of proteins governing mitochondrial dynamics and prevents high-fat diet-induced endothelial dysfunction. Br J Nutr. 2014;112:145–53. doi:10.1017/S0007114514000701.
Ting HC, Chao YJ, Hsu YHH. Polyunsaturated fatty acids incorporation into cardiolipin in H9c2 cardiac myoblast. J Nutr Biochem. 2015;26:769–75. doi:10.1016/j.jnutbio.2015.02.005.
Wai T, Garcia-Prieto J, Baker MJ, et al. Imbalanced OPA1 processing and mitochondrial fragmentation cause heart failure in mice. Science. 2015;350:aad0116. doi:10.1126/science.aad0116.
Weylandt KH. Docosapentaenoic acid derived metabolites and mediators - the new world of lipid mediator medicine in a nutshell. Eur J Pharmacol. 2016;785:108–15. doi:10.1016/j.ejphar.2015.11.002.
Acknowledgements
This work was supported by the research grants MOST 104-2313-B-002-038-MY3 (from the Ministry of Science and Technology) and NTU-CESRP-104R7615-3 (from National Taiwan University). The authors are grateful to Miss Mai-Jun Lai and Miss Cian-Jyun Kao for their technical assistance.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflicts of interest.
Rights and permissions
About this article
Cite this article
Hsu, HC., Li, SJ., Chen, CY. et al. Eicosapentaenoic acid protects cardiomyoblasts from lipotoxicity in an autophagy-dependent manner. Cell Biol Toxicol 34, 177–189 (2018). https://doi.org/10.1007/s10565-017-9406-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10565-017-9406-9