Abstract
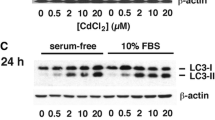
Cadmium represents a major environmental pollutant that may induce severe damage, especially in the kidney where cadmium accumulates. While cadmium is known to severely impair renal tubular functions, glomerular structures are also potential targets. Owing to their contractile properties, glomerular mesangial cells play a major role in the control of glomerular hemodynamics and influence the ultrafiltration coefficient. Cell cultures provide alternative and fruitful models for study of in vitro toxicology. However, the use of primary human mesangial cell cultures is hampered by their limited survival span and their rapid dedifferentiation during passages. This study presents a human stable immortalized mesangial cell line, designated IP15. Cell characteristics were investigated by the detection of known mesangial markers, as well as their ability to contract in response to angiotensin II. IP15 cells were used to investigate cadmium uptake and morphological changes such as cell contraction and cytoskeleton protein expression. The IC50 cytotoxicity index was obtained with 3.55 μmol/L using neutral red assay for 24 h. After cadmium exposure (1 μmol/L, determined as nonlethal concentration), 0.38 μg Cd/mg protein was internalized by the cells as evaluated by inductively coupled plasma optical emission spectrometry (ICP/OES). Cadmium induced a significant cell surface reduction that correlated with smooth-muscle α-actin disorganization. Thus, the IP15 cell line is a suitable model for study of in vitro cadmium cytotoxicity in mesangial cells and allows sufficient material to be obtained for future studies of the intracellular effects of cadmium exposure.
Similar content being viewed by others
Abbreviations
- PAGE:
-
polyacrylamide gel electrophoresis
- PCS:
-
planar cell surface
- RT-PCR:
-
reverse-transcription polymerase chain reaction
- SMA:
-
smooth-muscle α-actin
References
AFNOR XP T90–210. Qualité de l’eau. Protocole d’évaluation d’une méthode alternative d’analyse physico-chimique quantitative par rapport à une méthode de référence. Paris: Association Française de Normalisation; 1999.
Ardaillou N, Blaise V, Costenbader K, Vassitch Y, Ardaillou R. Characterization of a b2-bradykinin receptor in human glomerular podocytes. Am J Physiol. 1996a;27:F754–61.
Ardaillou R, Ronco P, Rondeau E. Biology of the renal cells in culture. In: Brenner BM, ed. The kidney. Brenner and Rector’s, 5th edn., Vol. 1. Philadelphia, PA: W.B. Saunders Company; 1996b:99–192.
Babich H, Borenfreund E. Application of the neutral red cytotoxicity assay to in vitro toxicology. ATLA Altern Lab Anim. 1990;18:129–44.
Banas B, Luckow B, Moller M, et al. Chemokine and chemokine receptor expression in a novel human mesangial cell line. J Am Soc Nephrol. 1999;10:2314–22.
Barbier O, Jacquillet G, Tauc M, Poujeol P, Cougnon M. Acute study of interaction among cadmium, calcium and zinc transport along the rat nephron in vivo. Am J Physiol Renal Physiol. 2004;287:1067–75.
Barrouillet MP, Potier M, Cambar J. Cadmium nephrotoxicity assessed in isolated rat glomeruli and cultured mesangial cells: evidence for contraction of glomerular cells. Exp Nephrol. 1999;7:251–8.
Bernard A, Lauwerys R. Effect of cadmium exposure in man. In: Foulkes EC, ed. Cadmium toxicology. Handbook of exprimental pharmacology. Vol. 80. Berlin: Springer; 1986:135–77.
Bradford MM. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein–dye binding. Anal Biochem. 1976;72:248–54.
Buchet J, Lauwerys R, Roels H, et al. Renal effect of cadmium on body burden of general population. Lancet. 1990;336:699–702.
Cherian G. Rat kidney epithelial cell culture for metal toxicity studies. InVitro Cell Dev Biol Anim. 1985;21:505–8.
Chin TA, Templeton DM. Effect of CdCl2 and Cd-metallothionein on cultured mesangial cells. Toxicol Appl Pharmacol. 1992;116:133–41.
Chuang SM, Wang IC, Yang JL. Roles of JNK, p38 and ERK mitogen-activated protein kinases in the growth inhibition and apoptosis induced by cadmium. Carcinogenesis. 2000;21:1423–32.
Cronin RE, Heinrich WL. Toxic nephropathy. In: Brenner BM, ed. The kidney. Brenner and Rector’s, 5th edn., Vol. 2. Philadelphia, PA: W.B. Saunders Company; 1996:1680–1711.
DalleDonne I, Milzani A, Colombo R. Actin assembly by cadmium ions. Biochim Biophys Acta. 1997;1357:5–17.
DelRaso NJ, Foy BD, Gearhart JM, Fraazier JM. Cadmium uptake kinetics in rat hepatocytes: correction for albumin binding. Toxicol Sci. 2003;72:19–30.
Dubus I, Vendrely B, Christophe I, et al. Mycophenolic acid antagonizes the activation of cultured human mesangial cells. Kidney Int. 2002;62:857–67.
Dubus I, L’Azou B, Gordien M, et al. Cytoskeletal reorganization by mycophenolic acid alters mesangial migration and contractility. Hypertension. 2003;42:956–61.
Dubus I, Sena S, Labouyrie JP, Bonnet J, Combe C. In vitro prevention of cyclosporin-induced cell contraction by mycophenolic acid. Life Sci. 2005;77:3366–74.
Elger M, Drenckhahn D, Nobiling R, Mundel P, Kritz W. Cultured rat mesangial cells contain smooth-muscle alpha-actin not found in vivo. Am J Pathol. 1993;142:497–509.
Ellis KJ. Dose–response analysis of heavy metal toxicants in man: direct in vivo assessment of body burden. Trace Subst Environ Health. 1985;19:149–59.
Foulkes EC. Transport of toxic heavy metals across cell membranes. Proc Soc Biol Med. 2000;223:234–40.
Goering PL, Waalkes MP, Klaassen CD. Toxicology of cadmium; Handbook of experimental pharmacology. In: Goyer RA, Cherian MC, eds. Toxicology of metals. Vol. 115. Berlin: Springer-Verlag; 1996:189–214.
Hirano S, Sun X, DeGuzman CA, et al. p38 MAPK/HSP25 signaling mediates cadmium-induced contraction of mesangial cells and renal glomeruli. Am J Physiol Renal Physiol. 2005;288:F1133–43.
Hruby Z, Beck KF. Cytotoxic effect of autocrine and macrophage-derived nitric oxide on cultured rat mesangial cells. Clin Exp Immunol. 1997;107:76–82.
Hu J, Mao Y, White K. Renal cell carcinoma and occupational exposure to chemicals in Canada. Occup Med (Lond). 2002;52:157–64.
IARC. Beryllium, cadmium, mercury and exposures in the glass manufacturing industry. Monographs on the evaluation of the carcinogenic risks to humans. Vol. 58. Lyon: International Agency for Resarch on Cancer, 1993:119–237.
Kawada T, Koyama H, Suzuki S. Cadmium, NAG activity, and β2-microglobulin in the urine of cadmium pigment workers. Br J Ind Med. 1989;46:52–5.
Kim J, Sharma P. Calcium-mediated activation of c-Jun NH2-terminal kinase (JNK) and apoptosis in response to cadmium in murine macrophages. Toxicol Sci. 2004;81:518–27.
Kreisberg JI, Hassid A. Functional properties of glomerular cells in culture. Miner Electrolyte Metab. 1986;12:25.
Kreisberg JI, Venkatachalam IM, Troyer D. Contractile properties of cultured glomerular mesangial cells. Am J Physiol Renal Fluid Electrolyte Physiol. 1985;18:F457–63.
L’Azou B, Cambar J. Les cultures de cellules rénales en pharmacologie. In: Barlovatz-Meinon GAM, ed. Cultures de cellules animales. Vol. 24. Paris: Editions INSERM; 2003:491–548.
L’Azou B, Dubus I, Ohayon-Courtès C, et al. Cadmium induces direct morphological changes in mesangial cell culture. Toxicology. 2002;179:233–45.
Lag M, Refsnes M, Lilleaas EM, Holme JA, Becher R, Scharze PE. Role of mitogen activated protein kinases and protein kinases C in cadmium-induced apoptosis of primary epithelial lung cells. Toxicol. 2005;211:253–64.
Méné P, Simonson MS, Dunn MJ. Physiology of the mesangial cell. Physiol Rev. 1989;69:1347–424.
NF EN ISO 11885. Qualité de l’eau – Dosage de 33 éléments par spectroscopie d’émission atomique avec plasma couplé par induction. Paris: Association Française de Normalisation;1998.
Nguyen G, Bouzhir L, Delarue F, Rondeau E, Sraer JD. Evidence of a renin receptor on human mesangial cells:effects on PAI 1 and CGMP. Nephrologie. 1998;19(7):411–6. [In French].
Nitta K, Koriba N, Uchida K, et al. Establishment and characterization of an immortalized bovine glomerular endothelial cell line. Nippon Jinzo Gakkai Shi. 1994;36:883–9.
Panasenko OO, Gusev NB. Mutual effects of alpha-actinin, calponin, and filamin on actin binding. Biochim Biophys Acta. 2001:393–405.
Pesch B, Haerting J, Ranft U, Klimpel A, Oelschlagel B, Schill W. Occupational risk factors for renal cell carcinoma: agent-specific results from a case-control study in Germany. MURC Study Group. Multicenter urothelial and renal cancer study. Int J Epidemiol. 2000;29:1014–24.
Potier M, L’Azou B, Cambar J. Isolated glomeruli and cultured mesangial cells as in vitro models to study immunosuppressive agents. Cell Biol Toxicol. 1996;12:263–70.
Prozialeck WC, Niewenhuis RJ. Cadmium (Cd2+) disrupts intercellular junctions and actin filaments in LLC-PK1 cells. Toxicol Appl Pharmacol. 1991;107:81–97.
Rodriguez-Barbero A, L’Azou B, Cambar J, Lopez-Novoa JM. Potential use of isolated and cultured mesangial cells as in vitro models to assess nephrotoxicity. Cell Biol Toxicol. 2000;16:145–53.
Rops AL, Van der Vlaag J, Jacobs CW, et al. Isolation and characterization of conditionally immortalized mouse glomerular endothelial cells. Kidney Int. 2004;66:2193–201.
Sabolic I, Herak-Krambergera CM, Brown D. Subchronic cadmium treatment affects the abundance and arrangement of cytoskeletal proteins in rat renal proximal tubule cells. Toxicology. 2001;165:205–16.
Sraer JD, Delarue F, Hagege J, et al. Stable cell lines of T-SV40 immortalized human glomerular mesangial cells. Kidney Int. 1996;49:260–70.
Stephenson LA, Haney LB, Hussaini IM, et al. Regulation of smooth-muscle alpha-actin expression and hypertrophy in cultured mesangial cells. Kidney Int. 1998;54:1175–87.
Stockand JD, Sansom SC. Glomerular mesangial cells: electrophysiology and regulation of contraction. Physiol Rev. 1998;78(3):723–44.
Striker LJ, Tannen RL, Lange MA, Striker GE. The contribution of cell culture to the study of renal diseases. Int Rev Exp. 1988;30:105.
Tang JX, Szymanski PT, Janmey PA, Tao T. Electrostatic effects of smooth-muscle calponin on actin assembly. Eur J Biochem. 1997:432–440.
Templeton DM. Cadmium uptake by cells of renal origin. J Biol Chem. 1990;265:21764–70.
Templeton DM, Chaitu N. Effects of divalent metals on the isolated rat glomerulus. Toxicology. 1990;61:119–33.
Templeton DM, Sheepers J. Effects on Ni and Cd on proteoglycan synthesis by isolated glomerulus and glomerular cells in culture. In: Bach PM, Gregg NJ, eds. Nephrotoxicity. Vol 57. New York: Dekker, 1991:377–82.
Uriu K, Kaizu K, Komine N, et al. Renal hemodynamics in rats with cadmium-induced nephropathy. Toxicol Appl Pharmacol. 1998;150:76–85.
Vestergaard P, Shaikh Z. The nephrotoxicity of intravenously administrated cadmium-metallothionein: effect of dose, mode of administration, and preexisting renal cadmium burden. Toxicol Appl Pharmacol. 1994;126:240–47.
Wang Z, Templeton DM. Cellular factors mediate cadmium-dependent actin depolymerization. Toxicol Appl Pharmacol. 1996;139:115–21.
Wang Z, Chin TA, Templeton DM. Calcium-independent effects of cadmium on actin assembly in mesangial and vascular smooth-muscle cells. Cell Motil Cytoskeleton. 1996;33:208–22.
Wilson HM, Stewart KN, MacLeod AM. Glomerular epithelial and mesangial cell culture and characterization. In: Jones GE, ed. Human cell culture protocols. Totowa, NJ: Humana Press; 1996:419–30.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
L’Azou, B., Dubus, I., Ohayon-Courtès, C. et al. Human glomerular mesangial IP15 cell line as a suitable model for in vitro cadmium cytotoxicity studies. Cell Biol Toxicol 23, 267–278 (2007). https://doi.org/10.1007/s10565-006-0888-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10565-006-0888-0