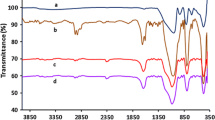
Abstract
Silica supported cinchona alkaloids were prepared by thio-ene coupled reaction so as to develop novel highly efficient heterogeneous organocatalysts for asymmetric Michael reaction. As-prepared supported cinchona alkaloids were used as heterogeneous catalysts to catalyze the asymmetric Michael reaction between 1,3-dicarbonyl compounds and N-benzylmaleimide, and their catalytic performance was evaluated. It was found that, when toluene is employed as the solvent, silica supported cinchona alkaloid catalysts can catalyze the aforementioned Michael reaction with medium enantiomeric excess (ee) values (up to 87 %) and significant diastereo ratio (dr) values (up to 96:4). In the meantime, they can be recovered and reused for at least five cycles while their stereo-selectivity remains almost unchanged. This means that the title catalysts could be highly efficient organocatalysts for the investigated Michael reaction.
Graphical Abstract



Similar content being viewed by others
References
Hashimoto T, Maruoka K (2007) Chem Rev 107:5656
Hélène P (2007) Tetrahedron 63:9267
Vicario JL, Badía D, Carrillo L (2007) Synthesis 2007:2065
Tsogoeva SB (2007) Eur J Org Chem 2007:1701
Mukherjee S, Yang JW, Hoffmann S, List B (2007) Chem Rev 107:5471
Jiang Z, Ye W, Yang Y, Tan CH (2008) Adv Synth Catal 350:2345
Lattanzi A, De Fusco C, Russo A, Poater A, Cavallo L (2012) Chem Commun 48:1650
Bartoli G, Bosco M, Carlone A, Cavalli A, Locatelli M, Mazzanti A, Ricci P, Sambri L, Melchiorre P (2006) Angew Chem Int Ed 45:4966
Luo J, Xu LW, Hay RAS, Lu Y (2008) Org Lett 11:437
Wang YF, Chen RX, Wang K, Zhang BB, Li ZB, Xu DQ (2012) Green Chem 14:893
Yeboah EMO, Yeboah SO, Singh GS (2011) Tetrahedron 67:1725
Czarnecki P, Plutecka A, Gawronski J, Kacprzak K (2011) Green Chem 13:1280
Nakamura S, Hayashi M, Hiramatsu Y, Shibata N, Funahashi Y, Toru T (2009) J Am Chem Soc 131:18240
Itsuno S, Parvez MM, Haraguchi N (2011) Polymer Chemistry 2:1942
Puglisi A, Benaglia M, Chiroli V (2013) Green Chem 15:1790
Trindade AF, Gois PMP, Afonso CAM (2009) Chem Rev 109:418
Qin Y, Yang G, Yang L, Li J, Cui Y (2011) Catal Lett 141:481
Arakawa Y, Haraguchi N, Itsuno S (2008) Angew Chem Int Ed 47:8232
Alvarez R, Hourdin MA, Cavé C, d’Angelo J, Chaminade P (1999) Tetrahedron Lett 40:7091
Thierry B, Plaquevent JC, Cahard D (2001) Tetrahedron Asymmetry 12:983
Danelli T, Annunziata R, Benaglia M, Cinquini M, Cozzi F, Tocco G (2003) Tetrahedron Asymmetry 14:461
Thierry B, Plaquevent JC, Cahard D (2003) Tetrahedron Asymmetry 14:1671
Mandoli A, Pini D, Agostini A, Salvadori P (2000) Tetrahedron Asymmetry 11:4039
Yang L, Zhou D, Qu C, Cui Y (2012) Catal Lett 142:1405
Qin Y, Zhao W, Yang L, Zhang X, Cui Y (2012) Chirality 24:640
Hong J, Lee I, Zaera F (2011) Top Catal 54:1340
Monge-Marcet A, Cattoen X, Alonso DA, Najera C, Man MWC, Pleixats R (2012) Green Chem 14:1601
Lowe AB, Harvison MA (2010) Aust J Chem 63:1251
Bortolini O, Caciolli L, Cavazzini A, Costa V, Greco R, Massi A, Pasti L (2012) Green Chem 14:992
Shi X, He W, Li H, Zhang X, Zhang S (2011) Tetrahedron Lett 52:3204
Li X, Zhang B, Xi ZG, Luo S, Cheng JP (2010) Adv Synth Catal 352:416
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Zhao, W., Zhang, Y., Qu, C. et al. Catalytic Performance of Silica Supported Cinchona Alkaloids as Heterogeneous Catalysts for Asymmetric Michael Reaction. Catal Lett 144, 1681–1688 (2014). https://doi.org/10.1007/s10562-014-1322-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10562-014-1322-5