Abstract
A series of aromatic ring spaced binuclear Pd(II) complexes were synthesized from sterically tuned Schiff base ligands derived from 4,6-diacetylresorcinol and 2,6-dialkyl substituted anilines to provide a general and flexible set of complexes for terminal olefin epoxidation studies using “green” oxidant 30 % hydrogen peroxide as a terminal oxidant. All the compounds were fully characterized by analytical and spectrometric methods. Additionally, the ligands L1 and L2 were characterized by single crystal X-ray diffraction technique. The complexes C1 and C2 displayed high activity and substrate selectivity in the oxidation of 1-octene, whereas mixed products were observed in case of styrene with reasonable yields, arguably assignable to stringent steric and electronic factors of the complexes employed. Up to 98.5 % conversion is observed for epoxidation of 1-octene with high substrate selectivity, whereas up to 95 % conversion is observed for styrene with poor selectivity.
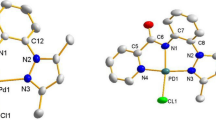
Graphical Abstract
A series of Pd(II) complexes were synthesized from sterically tuned Schiff base ligands derived from 4,6-diacetylresorcinol and 2,6-dialkyl substituted anilines to provide a flexible set of complexes for olefin epoxidation studies using hydrogen peroxide as a terminal oxidant. The complexes C1–C3 showed substrate selectivity in the oxidation of 1-octene, where as mixed products were observed in case of styrene with reasonable yields.










Similar content being viewed by others
References
Leeuwen PWNM (2004) Homogeneous catalysis. Kluwer Academic Publishers, Dordrecht
Weissermel K, Arpe HJ (1978) Industrial organic chemistry: important raw materials and intermediates. Verlag Chemie, Weinheim
Jacobsen EN, Zhang W, Muci AR, Ecker JR, Deng L (1991) J Am Chem Soc 113:7063
Bagherzadeh M, Zare M (2012) J Coord Chem 65:4054
Ishii Y, Yamawaki K, Ura T, Yamada H, Yoshida T, Ogawa M (1988) J Org Chem 53:3587
Herrmann WA, Fischer RW, Marz DW (1991) Angew Chem Int Ed 30:1638
Vos DE, Sels BF, Reynaers M, Subba Rao YV, Jacobs PA (1998) Tetrahedron Lett 39:3221
Copéret C, Adolfsson H, Sharpless KB (1997) Chem Commun 1565
Rudler H, Gregorio JR, Denise B, Brégeault JM, Deloffre A (1998) J Mol Catal A 133:255
van Vliet MCA, Arends IWCE, Sheldon RA (1999) Chem Commun 821
Tsuji Y, Ohta T, Ido T, Minbu H, Watanabe Y (1984) J Organomet Chem 270:333
Yang DH, Gao L, Zhao WJ (2008) Catal Lett 126:84
Zhao J, Han J, Zhang YC (2005) J Mol Catal A 231:129
Barak G, Dakka J, Sasson Y (1988) J Org Chem 53:3553
Marko IE, Gautier A, Chelle-Regnaut I, Giles PR, Tsukazaki M, Urch CJ, Brown SM (1998) J Org Chem 63:7576
Wang RM, Hao CJ, He YF, Wang YP, Xia CG (2002) Polym Adv Technol 13:6
Abdi SHR, Kureshy RI, Khan NH, Jasra RV (2004) Catal Surv Jpn 8:187
Halligudi SB, Devassy BM, Kala Raj NK, Degaonkar MP, Gopinathan S (2000) React Kinet Catal Lett 71:289
Fernández I, Pedro JR, Roselló AL, Ruiz R, Castro I, Ottenwaelder X, Journaux Y (2001) Eur J Org Chem 7:1235
Atlay MT, Preece M, Strukul G, James BR (1982) J Chem Soc Chem Commun 406
Atlay MT, Preece M, Strukul G, James BR (1983) Can J Chem 61:1332
Strukul G, Michelin RA, Orbell JD, Randaccio L (1983) Inorg Chem 22:3706
Strukul G, Michelin RA (1984) J Chem Soc Chem Commun 1538
Strukul G, Michelin RA (1985) J Am Chem Soc 107:7563
Zanardo A, Pinna F, Michelin RA, Strukul G (1988) Inorg Chem 27:1966
Colladon M, Scarso A, Sgarbossa P, Michelin RA, Strukul G (2007) J Am Chem Soc 129:7685
Colladon M, Scarso A, Sgarbossa P, Michelin RA, Strukul G (2006) J Am Chem Soc 128:14007
Strukul G, Michelin RA (1984) JCS Chem Commun 1538
Colladon M, Scarso A, Strukul G (2007) Adv Synth Catal 349:797
Trost BM (1995) Angew Chem Int Ed 34:259
Strukul G (ed) (1992) Catalytic oxidations with hydrogen peroxide as oxidant. Kluwer Academic Publishers, Dordrecht
Grigoropoulou G, Clark JH, Elings JA (2003) Green Chem 5:1
Lane BS, Burgess K (2003) Chem Rev 103:2457
Noyori R, Aoki M, Sato K (2003) Chem Commun 1977
Takeuchi D (2010) Dalton Trans 39:311
Mimoun H, Charpentier R, Mitschler A, Fischer J, Weiss R (1980) J Am Chem Soc 102:1047
Igersheim F, Mimoun H (1980) Nouv J Chim 4:711
Bregeault JM, Mimoun H (1981) Nouv J Chim 5:287
Roussel M, Mimoun H (1980) J Org Chem 45:5381
Cooper RI, Thompson AL, Watkin DJ (2010) J Appl Crystallogr 43:1100
Betteridge PW, Carruthers JR, Cooper RI, Prout K, Watkin DJ (2003) J Appl Crystallogr 36:1487
Altomare A, Cascarano G, Giacovazzo C, Guagliardi A (1993) J Appl Crystallogr 26:343
Sheldrick GM (2008) Acta Crystallogr A 64:112
Farrugia LJ (1999) J Appl Crystallogr 32:837
Bruno IJ, Cole JC, Edgington PR, Kessler M, Macrae CF, McCabe P, Pearson J, Taylor R (2002) Acta Crystallogr B 58:389
Watkin DJ, Prout CK, Pearce LJ (1996) CAMERON. Chemical Crystallography Laboratory, Oxford
Rao PS, Reddy KV, Reddy KV (1997) Int J Rapid Commun Synth Org Chem 27:19
Biyala MK, Sharma K, Swami M, Fahmi N, Singh RV (2008) Transition Met Chem 33:377
Lever ABP (1968) Inorganic electronic spectroscopy. Elsevier, Amsterdam
Bahuleyan BK, Lee UK, Ha CS, Kim I (2008) Appl Catal A 351:36
Jie S, Zhang D, Zhang T, Sun WH, Chen J, Ren Q, Liu D, Zheng G, Chen W (2005) J Organomet Chem 691:1739
Kovach J, Peralta M, Brennessel WW, Jones WD (2011) J Mol Struct 992:33
Prasetyoko D, Fansuri H, Ramli Z, Endud S, Nur H (2009) Catal Lett 128:177
Chin TK, Endud S, Jamil S, Budagumpi S, Lintang HO (2013) Catal Lett 143:282
Ghorbanloo M, Monfared HH, Janiak C (2011) J Mol Catal A Chem 345:12
Altmann P, Cokoja M, Kühn FE (2012) J Organomet Chem 701:51
Yadav GD, Pujari AA (2000) Org Proc Res Dev 4:88
Lu XH, Lei J, Zhou D, Fang SY, Dong YL, Xia QH (2010) Indian J Chem 49:1586
Acknowledgments
The authors are thankful to USIC, Karnatak University, Dharwad, for providing spectral facilities. Recording of NMR and IR spectra from IISc-Bangalore and IIT-Bombay is gratefully acknowledged. One of the authors (Sandeep P. Netalkar) is thankful to Department of Science & Technology for providing financial assistance under INSPIRE fellowship program.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Netalkar, S.P., Nevrekar, A.A. & Revankar, V.K. Design, Synthesis and Characterization of Bimetallic Palladium Complexes for Terminal Olefin Epoxidation. Catal Lett 144, 1573–1583 (2014). https://doi.org/10.1007/s10562-014-1315-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10562-014-1315-4