Abstract
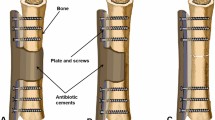
Infection of an orthopedic implant is considered a devastating complication, necessitating its complete removal and thorough debridement of the site. Osseous defects are common in such conditions and need to be addressed before a new implant may be inserted. So far bone grafting has been contraindicated in bacterially contaminated areas and could only be performed as soon as all signs of infection have ceased. Usually long term antibiotic treatment and a multitude of surgical interventions within a period of several months is required until a definitive supply can be achieved. Allograft bone may be impregnated with high loads of antibiotics using special incubation techniques. Based on this technology 48 exchange procedures of infected orthopaedic implants were performed in a single stage, all of them without the use of bone cement. There were 37 infected hips, 8 knees and 3 infected osteosyntheses. Two hips required re-revision because of persisting infection, the remaining 46 patients stayed infect free for a period between 1 and 7 years after surgery. No adverse side effects could be found. Incorporation appeared as after grafting with unimpregnated bone grafts. Antibiotic loaded allograft bone is a powerful tool in septic revision surgery, enabling restoration of bone stock, insertion of a new implant and control of infection in a single operation.
Similar content being viewed by others
References
Buttaro MA, Pusso R, Piccaluga F (2005) Vancomycin-supplemented impacted bone allografts in infected hip arthroplasty. Two-stage revision results. J Bone Joint Surg Br 87(3):314–319
Buttaro MA, Gimenez MI, Greco G, Barcan L, Piccaluga F (2005) High active local levels of vancomycin without nephrotoxicity released from impacted bone allografts in 20 revision hip arthroplasties. Acta Orthop 76(3):336–340
Costerton JW (2005) Biofilm theory can guide the treatment of device-related orthopaedic infections. Clin Orthop Relat Res 437:7–11
Edin ML, Miclau T, Lester GE, Lindsey RW, Dahners LE (1996) Effect of cefazolin and vancomycin on osteoblasts in vitro. Clin Orthop 333:245–251
Gristina AG (1987) Biomaterial-centered infection: microbial adhesion versus tissue integration. Science 237–4822:1588–1595
Miclau T, Edin ML, Lester GE, Lindsey RW, Dahners LE (1995) Bone toxicity of locally applied aminoglycosides. J Orthop Trauma 9(5):401–406
Miclau T, Edin ML, Lester GE, Lindsey RW, Dahners LE (1998) Effect of ciprofloxacin on the proliferation of osteoblast-like MG-63 human osteosarcoma cells in vitro. J Orthop Res 16–4:509–512
Nelson CL, McLaren AC, McLaren SG, Johnson JW, Smeltzer MS (2005) Is aseptic loosening truly aseptic? Clin Orthop Relat Res 437:25–30
Neut D, van de Belt H, Stokroos I, van Horn JR, van der Mei HC, Busscher HJ (2001) Biomaterial-associated infection of gentamicin-loaded PMMA beads in orthopaedic revision surgery. J Antimicrob Chemother 47(6):885–891
Neut D, van Horn JR, van Kooten TG, van der Mei HC, Busscher HJ (2003) Detection of biomaterial-associated infections in orthopaedic joint implants. Clin Orthop Relat Res 413:261–268
Winkler H, Janata O, Berger C, Wein W, Georgopoulos A (2000) In vitro release of vancomycin and tobramycin from impregnated human and bovine bone grafts. J Antimicrob Chemother 46(3):423–428
Witso E, Persen L, Loseth K, Bergh K (1999) Adsorption and release of antibiotics from morselized cancellous bone. In vitro studies of 8 antibiotics. Acta Orthop Scand 70(3):298–304
Witso E, Persen L, Loseth K, Benum P, Bergh K (2000) Cancellous bone as an antibiotic carrier. Acta Orthop Scand 71(1):80–84
Witso E, Persen L, Benum P, Aamodt A, Husby OS, Bergh K (2004) High local concentrations without systemic adverse effects after impaction of netilmicin-impregnated bone. Acta Orthop Scand 75(3):339–346
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Winkler, H., Kaudela, K., Stoiber, A. et al. Bone grafts impregnated with antibiotics as a tool for treating infected implants in orthopedic surgery – one stage revision results. Cell Tissue Banking 7, 319–323 (2006). https://doi.org/10.1007/s10561-006-9010-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10561-006-9010-3