Abstract
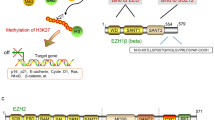
Tumor angiogenesis and metastatic spreading are two highly interconnected phenomena, which contribute to cancer-associated deaths. Thus, the identification of novel strategies to target angiogenesis and metastatic spreading is crucial. Polycomb genes are a set of epigenetic effectors, structured in multimeric repressive complexes. EZH2 is the catalytic subunit of Polycomb repressive complex 2 (PRC2), which methylates histone H3 lysine 27, thereby silencing several tumor-suppressor genes. EZH2 is essential for cancer stem cell self-renewal. Interestingly, cancer stem cells are thought to be the seeds of metastatic spreading and are able to differentiate into tumor-associated endothelial cells. Pre-clinical studies showed that EZH2 is able to silence several anti-metastatic genes ( e.g., E-cadherin and tissue inhibitors of metalloproteinases), thereby favoring cell invasion and anchorage-independent growth. In addition, EZH2 seems to play a crucial role in the regulation of tumor angiogenesis. High EZH2 expression predicts poor prognosis, high grade, and high stage in several cancer types. Recently, a small molecule inhibitor of PRC2 (DZNeP) demonstrated promising anti-tumor activity, both in vitro and in vivo. Interestingly, DZNeP was able to inhibit cancer cell invasion and tumor angiogenesis in prostate and brain cancers, respectively. At tumor-inhibiting doses, DZNeP is not harmful for non-transformed cells. In the present manuscript, we review current evidence supporting a role of EZH2 in metastatic spreading and tumor angiogenesis. Using Oncomine datasets, we show that DZNeP targets are specifically silenced in some metastatic cancers, and some of them may inhibit angiogenesis. Based on this evidence, we propose the development of EZH2 inhibitors as anti-angiogenic and anti-metastatic therapy.


Similar content being viewed by others
References
Wirtz D, Konstantopoulos K, Searson PC (2011) The physics of cancer: the role of physical interactions and mechanical forces in metastasis. National Review Cancer, 11:512-522.
Fornier M (2010) Ixabepilone plus capecitabine for breast cancer patients with an early metastatic relapse after adjuvant chemotherapy: two clinical trials. Clinical Breast Cancer, 10:352-358.
Soria JC, Massard C, Le Chevalier T (2010) Should progression-free survival be the primary measure of efficacy for advanced NSCLC therapy? Annales Oncologica, 21:2324-2332.
Kerbel, R. S. (2008). Tumor angiogenesis. The New England Journal of Medicine, 358, 2039–2049.
Su, J. L., Yang, P. C., Shih, J. Y., Yang, C. Y., Wei, L. H., Hsieh, C. Y., et al. (2006). The VEGF-C/Flt-4 axis promotes invasion and metastasis of cancer cells. Cancer Cell, 9, 209–223.
Hurwitz, H. I., Fehrenbacher, L., Hainsworth, J. D., Heim, W., Berlin, J., Holmgren, E., et al. (2005). Bevacizumab in combination with fluorouracil and leucovorin: an active regimen for first-line metastatic colorectal cancer. Journal of Clinical Oncology, 23, 3502–3508.
Lee, F. Y., Covello, K. L., Castaneda, S., Hawken, D. R., Kan, D., Lewin, A., et al. (2008). Synergistic antitumor activity of ixabepilone (BMS-247550) plus bevacizumab in multiple in vivo tumor models. Clinical Cancer Research, 14, 8123–8131.
Rousseau B, Chibaudel B, Bachet JB, Larsen AK, Tournigand C, Louvet C, Andre T, de Gramont A (2010) Stage II and stage III colon cancer: treatment advances and future directions. Cancer Journal, 16:202-209.
Valachis A, Polyzos NP, Patsopoulos NA, Georgoulias V, Mavroudis D, Mauri D (2010) Bevacizumab in metastatic breast cancer: a meta-analysis of randomized controlled trials. Breast Cancer Research Treatment, 122:1-7.
Bear HD, Tang G, Rastogi P, Geyer CE, Jr., Robidoux A, Atkins JN, Baez-Diaz L, Brufsky AM, Mehta RS, Fehrenbacher L, et al (2012) Bevacizumab added to neoadjuvant chemotherapy for breast cancer. The New England Journal of Medicine, 366:310-320.
von Minckwitz G, Eidtmann H, Rezai M, Fasching PA, Tesch H, Eggemann H, Schrader I, Kittel K, Hanusch C, Kreienberg R, et al. (2012) Neoadjuvant chemotherapy and bevacizumab for HER2-negative breast cancer. New England Journal of Medicine, 366:299-309.
Mulder K, Koski S, Scarfe A, Chu Q, King K, Spratlin J (2010) Antiangiogenic agents in advanced gastrointestinal malignancies: past, present and a novel future. Oncotarget, 1:515-529.
Lu X, Kang Y (2010) Hypoxia and hypoxia-inducible factors: master regulators of metastasis. Clinical Cancer Research, 16:5928-5935.
Mathews, L. A., Crea, F., & Farrar, W. L. (2009). Epigenetic gene regulation in stem cells and correlation to cancer. Differentiation, 78, 1–17.
Tan, J., Yang, X., Zhuang, L., Jiang, X., Chen, W., Lee, P. L., et al. (2007). Pharmacologic disruption of Polycomb -repressive complex 2-mediated gene repression selectively induces apoptosis in cancer cells. Genes & Development, 21, 1050–1063.
Weber, G. F. (2008). Molecular mechanisms of metastasis. Cancer Letters, 270, 181–190.
Thiery, J. P. (2002). Epithelial-mesenchymal transitions in tumour progression. Nature Reviews. Cancer, 2, 442–454.
Lombaerts, M., van Wezel, T., Philippo, K., Dierssen, J. W., Zimmerman, R. M., Oosting, J., et al. (2006). E-cadherin transcriptional downregulation by promoter methylation but not mutation is related to epithelial-to-mesenchymal transition in breast cancer cell lines. British Journal of Cancer, 94, 661–671.
Iochmann, S., Blechet, C., Chabot, V., Saulnier, A., Amini, A., Gaud, G., et al. (2009). Transient RNA silencing of tissue factor pathway inhibitor-2 modulates lung cancer cell invasion. Clinical & Experimental Metastasis, 26, 457–467.
Kraljevic Pavelic S, Sedic M, Bosnjak H, Spaventi S, Pavelic K (2011) Metastasis: new perspectives on an old problem. Molecular Cancer, 10:22.
Mani, S. A., Guo, W., Liao, M. J., Eaton, E. N., Ayyanan, A., Zhou, A. Y., et al. (2008). The epithelial-mesenchymal transition generates cells with properties of stem cells. Cell, 133, 704–715.
Hurt, E. M., & Farrar, W. L. (2008). Cancer stem cells: the seeds of metastasis? Molecular Interventions, 8, 140–142.
Hanahan, D., & Weinberg, R. A. (2000). The hallmarks of cancer. Cell, 100, 57–70.
Thullberg, M., & Stromblad, S. (2008). Anchorage-independent cytokinesis as part of oncogenic transformation? Cell Cycle, 7, 984–988.
Berezovska, O. P., Glinskii, A. B., Yang, Z., Li, X. M., Hoffman, R. M., & Glinsky, G. V. (2006). Essential role for activation of the Polycomb group (PcG) protein chromatin silencing pathway in metastatic prostate cancer. Cell Cycle, 5, 1886–1901.
Weis SM, Cheresh DA (2011) Tumor angiogenesis: molecular pathways and therapeutic targets. National Medicine, 17:1359-1370.
Jubb AM, Harris AL (2010) Biomarkers to predict the clinical efficacy of bevacizumab in cancer. Lancet Oncol, 11:1172-1183.
Ellis, L. M., & Hicklin, D. J. (2008). Pathways mediating resistance to vascular endothelial growth factor-targeted therapy. Clinical Cancer Research, 14, 6371–6375.
Bergers, G., & Hanahan, D. (2008). Modes of resistance to anti-angiogenic therapy. Nature Reviews. Cancer, 8, 592–603.
Vermeulen L, de Sousa e Melo F, Richel DJ, Medema JP (2012) The developing cancer stem-cell model: clinical challenges and opportunities. Lancet Oncol, 13:e83-89.
Biddle A, Mackenzie IC (2012) Cancer stem cells and EMT in carcinoma. Cancer Metastasis Review (in press)
Siddique HR, Saleem M (2012) Role of BMI1, a stem cell factor, in cancer recurrence and chemoresistance: preclinical and clinical evidences. Stem Cells, 30:372-378.
Ribatti D (2012) Cancer stem cells and tumor angiogenesis. Cancer Letters (in press)
Folkins, C., Man, S., Xu, P., Shaked, Y., Hicklin, D. J., & Kerbel, R. S. (2007). Anticancer therapies combining antiangiogenic and tumor cell cytotoxic effects reduce the tumor stem-like cell fraction in glioma xenograft tumors. Cancer Research, 67, 3560–3564.
Daenen, L. G., Shaked, Y., Man, S., Xu, P., Voest, E. E., Hoffman, R. M., et al. (2009). Low-dose metronomic cyclophosphamide combined with vascular disrupting therapy induces potent antitumor activity in preclinical human tumor xenograft models. Molecular Cancer Therapeutics, 8, 2872–2881.
Hugo, H., Ackland, M. L., Blick, T., Lawrence, M. G., Clements, J. A., Williams, E. D., et al. (2007). Epithelial–mesenchymal and mesenchymal–epithelial transitions in carcinoma progression. Journal of Cellular Physiology, 213, 374–383.
Ebos, J. M., Lee, C. R., Cruz-Munoz, W., Bjarnason, G. A., Christensen, J. G., & Kerbel, R. S. (2009). Accelerated metastasis after short-term treatment with a potent inhibitor of tumor angiogenesis. Cancer Cell, 15, 232–239.
Karanikolas BD, Figueiredo ML, Wu L( 2010) Comprehensive evaluation of the role of EZH2 in the growth, invasion, and aggression of a panel of prostate cancer cell lines. Prostate, 70:675-688.
Du J, Li L, Ou Z, Kong C, Zhang Y, Dong Z, Zhu S, Jiang H, Shao Z, Huang B, Lu J (2012) FOXC1, a target of Polycomb, inhibits metastasis of breast cancer cells. Breast Cancer Research Treatment, 131:65-73.
Zheng F, Liao YJ, Cai MY, Liu YH, Liu TH, Chen SP, Bian XW, Guan XY, Lin MC, Zeng YX, et al (2011) The putative tumour suppressor microRNA-124 modulates hepatocellular carcinoma cell aggressiveness by repressing ROCK2 and EZH2. Gut, 61:278-289.
Smits M, Nilsson J, Mir SE, van der Stoop PM, Hulleman E, Niers JM, de Witt Hamer PC, Marquez VE, Cloos J, Krichevsky AM, et al. (2010) miR-101 is down-regulated in glioblastoma resulting in EZH2-induced proliferation, migration, and angiogenesis. Oncotarget, 1:710-720.
Tong ZT, Cai MY, Wang XG, Kong LL, Mai SJ, Liu YH, Zhang HB, Liao YJ, Zheng F, Zhu W, et al. (2011) EZH2 supports nasopharyngeal carcinoma cell aggressiveness by forming a co-repressor complex with HDAC1/HDAC2 and Snail to inhibit E-cadherin. Oncogene, 31:583-594.
Min J, Zaslavsky A, Fedele G, McLaughlin SK, Reczek EE, De Raedt T, Guney I, Strochlic DE, Macconaill LE, Beroukhim R, et al. (2010) An oncogene-tumor suppressor cascade drives metastatic prostate cancer by coordinately activating Ras and nuclear factor-kappaB. National Medicine, 16:286-294.
Leung-Kuen Au S, Chak-Lui Wong C, Man-Fong Lee J, Ngo-Yin Fan D, Hoching Tsang F, Oi-Lin Ng I, Wong CM (2012) Enhancer of zeste homolog 2 (EZH2) epigenetically silences multiple tumor suppressor miRNAs to promote liver cancer metastasis. Hepatology.
Crea F, Paolicchi E, Marquez VE, Danesi R (2012) Polycomb genes and cancer: Time for clinical application? Critical Review Oncology Hematology (in press)
Wang CG, Ye YJ, Yuan J, Liu FF, Zhang H, Wang S (2010) EZH2 and STAT6 expression profiles are correlated with colorectal cancer stage and prognosis. World Journal Gastroenterology, 16:2421-2427.
Lv Y, Yuan C, Xiao X, Wang X, Ji X, Yu H, Wu Z, Zhang J (2012) The expression and significance of the enhancer of zeste homolog 2 in lung adenocarcinoma. Oncology Report, 28:147-154.
Crea F, Hurt EM, Farrar WL (2010) Clinical significance of Polycomb gene expression in brain tumors. Molecular Cancer, 9:265.
Reijm EA, Jansen MP, Ruigrok-Ritstier K, van Staveren IL, Look MP, van Gelder ME, Sieuwerts AM, Sleijfer S, Foekens JA, Berns EM (2011) Decreased expression of EZH2 is associated with upregulation of ER and favorable outcome to tamoxifen in advanced breast cancer. Breast Cancer Research Treatment, 125:387-394.
Crea F, Hurt EM, Mathews LA, Cabarcas SM, Sun L, Marquez VE, Danesi R, Farrar WL (2010) Pharmacologic disruption of Polycomb Repressive Complex 2 inhibits tumorigenicity and tumor progression in prostate cancer. Molecular Cancer, 10:40.
Cai MY, Hou JH, Rao HL, Luo RZ, Li M, Pei XQ, Lin MC, Guan XY, Kung HF, Zeng YX, Xie D (2011) High expression of H3K27me3 in human hepatocellular carcinomas correlates closely with vascular invasion and predicts worse prognosis in patients. Molecular Medicine, 17:12-20.
Fujii S, Tokita K, Wada N, Ito K, Yamauchi C, Ito Y, Ochiai A (2011) MEK-ERK pathway regulates EZH2 overexpression in association with aggressive breast cancer subtypes. Oncogene, 30:4118-4128.
Lovat F, Valeri N, Croce CM (2011) MicroRNAs in the pathogenesis of cancer. Seminars Oncology, 38:724-733.
Zhang B, Liu XX, He JR, Zhou CX, Guo M, He M, Li MF, Chen GQ, Zhao Q (2011) Pathologically decreased miR-26a antagonizes apoptosis and facilitates carcinogenesis by targeting MTDH and EZH2 in breast cancer. Carcinogenesis, 32:2-9.
Kong D, Heath E, Chen W, Cher ML, Powell I, Heilbrun L, Li Y, Ali S, Sethi S, Hassan O, et al. (2012) Loss of let-7 up-regulates EZH2 in prostate cancer consistent with the acquisition of cancer stem cell signatures that are attenuated by BR-DIM. PLoS One, 7:e33729.
Varambally, S., Cao, Q., Mani, R. S., Shankar, S., Wang, X., Ateeq, B., et al. (2008). Genomic loss of microRNA-101 leads to overexpression of histone methyltransferase EZH2 in cancer. Science, 322, 1695–1699.
Wang HJ, Ruan HJ, He XJ, Ma YY, Jiang XT, Xia YJ, Ye ZY, Tao HQ (2010) MicroRNA-101 is down-regulated in gastric cancer and involved in cell migration and invasion. European Journal Cancer, 46:2295-2303.
Cao Q, Mani RS, Ateeq B, Dhanasekaran SM, Asangani IA, Prensner JR, Kim JH, Brenner JC, Jing X, Cao X, et al. (2011) Coordinated regulation of polycomb group complexes through microRNAs in cancer. Cancer Cell, 20:187-199.
Lu, C., Bonome, T., Li, Y., Kamat, A. A., Han, L. Y., Schmandt, R., et al. (2007). Gene alterations identified by expression profiling in tumor-associated endothelial cells from invasive ovarian carcinoma. Cancer Research, 67, 1757–1768.
Lu C, Han HD, Mangala LS, Ali-Fehmi R, Newton CS, Ozbun L, Armaiz-Pena GN, Hu W, Stone RL, Munkarah A, et al. (2010) Regulation of tumor angiogenesis by EZH2. Cancer Cell, 18:185-197.
Kottakis F, Polytarchou C, Foltopoulou P, Sanidas I, Kampranis SC, Tsichlis PN (2011) FGF-2 regulates cell proliferation, migration, and angiogenesis through an NDY1/KDM2B-miR-101-EZH2 pathway. Molecular Cell, 43:285-298.
Ricci-Vitiani L, Pallini R, Biffoni M, Todaro M, Invernici G, Cenci T, Maira G, Parati EA, Stassi G, Larocca LM, De Maria R (2010) Tumour vascularization via endothelial differentiation of glioblastoma stem-like cells. Nature, 468:824-828.
Miranda, T. B., Cortez, C. C., Yoo, C. B., Liang, G., Abe, M., Kelly, T. K., et al. (2009). DZNep is a global histone methylation inhibitor that reactivates developmental genes not silenced by DNA methylation. Molecular Cancer Therapeutics, 8, 1579–1588.
Hayden A, Johnson PW, Packham G, Crabb SJ (2011) S-adenosylhomocysteine hydrolase inhibition by 3-deazaneplanocin A analogues induces anti-cancer effects in breast cancer cell lines and synergy with both histone deacetylase and HER2 inhibition. Breast Cancer Research Treatment, 127:109-119.
Kemp CD, Rao M, Xi S, Inchauste S, Mani H, Fetsch P, Filie A, Zhang M, Hong JA, Walker RL, et al. (2012) Polycomb repressor complex-2 is a novel target for mesothelioma therapy. Clinics Cancer Research, 18:77-90.
Suva, M. L., Riggi, N., Janiszewska, M., Radovanovic, I., Provero, P., Stehle, J. C., et al. (2009). EZH2 is essential for glioblastoma cancer stem cell maintenance. Cancer Research, 69, 9211–9218.
Chiba T, Suzuki E, Negishi M, Saraya A, Miyagi S, Konuma T, Tanaka S, Tada M, Kanai F, Imazeki F, et al. (2012) 3-Deazaneplanocin A is a promising therapeutic agent for the eradication of tumor-initiating hepatocellular carcinoma cells. International Journal of Cancer, 130:2557-2567.
Fiskus, W., Wang, Y., Sreekumar, A., Buckley, K. M., Shi, H., Jillella, A., et al. (2009). Combined epigenetic therapy with the histone methyltransferase EZH2 inhibitor 3-deazaneplanocin A and the histone deacetylase inhibitor panobinostat against human AML cells. Blood, 114, 2733–2743.
Smits M, Mir SE, Nilsson RJ, van der Stoop PM, Niers JM, Marquez VE, Cloos J, Breakefield XO, Krichevsky AM, Noske DP, et al. (2011) Down-regulation of miR-101 in endothelial cells promotes blood vessel formation through reduced repression of EZH2. PLoS One, 6:e16282.
Coulombe, R. A., Jr., Sharma, R. P., & Huggins, J. W. (1995). Pharmacokinetics of the antiviral agent 3-deazaneplanocin A. European Journal of Drug Metabolism and Pharmacokinetics, 20, 197–202.
Sun F, Li J, Yu Q, Chan E (2012) Loading 3-deazaneplanocin A into pegylated unilamellar liposomes by forming transient phenylboronic acid-drug complex and its pharmacokinetic features in Sprague-Dawley rats. European Journal of Pharmacology and Biopharmacology, 80:323-331.
Fiskus, W., Buckley, K., Rao, R., Mandawat, A., Yang, Y., Joshi, R., et al. (2009). Panobinostat treatment depletes EZH2 and DNMT1 levels and enhances decitabine mediated de-repression of JunB and loss of survival of human acute leukemia cells. Cancer Biology & Therapy, 8, 939–950.
Choudhury SR, Balasubramanian S, Chew YC, Han B, Marquez VE, Eckert RL (2011) (-)-Epigallocatechin-3-gallate and DZNep reduce polycomb protein level via a proteasome-dependent mechanism in skin cancer cells. Carcinogenesis, 32:1525-1532.
Chang CJ, Yang JY, Xia W, Chen CT, Xie X, Chao CH, Woodward WA, Hsu JM, Hortobagyi GN, Hung MC (2011) EZH2 promotes expansion of breast tumor initiating cells through activation of RAF1-beta-catenin signaling. Cancer Cell, 19:86-100.
Zeng X, Chen S, Huang H (2011) Phosphorylation of EZH2 by CDK1 and CDK2: a possible regulatory mechanism of transmission of the H3K27me3 epigenetic mark through cell divisions. Cell Cycle, 10:579-583.
Rhodes, D. R., Yu, J., Shanker, K., Deshpande, N., Varambally, R., Ghosh, D., et al. (2004). Oncomine: a cancer microarray database and integrated data-mining platform. Neoplasia, 6, 1–6.
Radvanyi, L., Singh-Sandhu, D., Gallichan, S., Lovitt, C., Pedyczak, A., Mallo, G., et al. (2005). The gene associated with trichorhinophalangeal syndrome in humans is overexpressed in breast cancer. Proceedings of the National Academy of Sciences of the United States of America, 102, 11005–11010.
Graudens, E., Boulanger, V., Mollard, C., Mariage-Samson, R., Barlet, X., Gremy, G., et al. (2006). Deciphering cellular states of innate tumor drug responses. Genome Biology, 7, R19.
Chen, X., Leung, S. Y., Yuen, S. T., Chu, K. M., Ji, J., Li, R., et al. (2003). Variation in gene expression patterns in human gastric cancers. Molecular Biology of the Cell, 14, 3208–3215.
Lapointe, J., Li, C., Higgins, J. P., van de Rijn, M., Bair, E., Montgomery, K., et al. (2004). Gene expression profiling identifies clinically relevant subtypes of prostate cancer. Proceedings of the National Academy of Sciences of the United States of America, 101, 811–816.
Tothill, R. W., Tinker, A. V., George, J., Brown, R., Fox, S. B., Lade, S., et al. (2008). Novel molecular subtypes of serous and endometrioid ovarian cancer linked to clinical outcome. Clinical Cancer Research, 14, 5198–5208.
O'Donnell, R. K., Kupferman, M., Wei, S. J., Singhal, S., Weber, R., O'Malley, B., et al. (2005). Gene expression signature predicts lymphatic metastasis in squamous cell carcinoma of the oral cavity. Oncogene, 24, 1244–1251.
Shin YJ, Kim JH (2012) The role of EZH2 in the regulation of the activity of matrix metalloproteinases in prostate cancer cells. PLoS One, 7:e30393.
Qi, J. H., Ebrahem, Q., Moore, N., Murphy, G., Claesson-Welsh, L., Bond, M., et al. (2003). A novel function for tissue inhibitor of metalloproteinases-3 (TIMP3): inhibition of angiogenesis by blockage of VEGF binding to VEGF receptor-2. Nature Medicine, 9, 407–415.
Zucker, S., Hymowitz, M., Conner, C., Zarrabi, H. M., Hurewitz, A. N., Matrisian, L., et al. (1999). Measurement of matrix metalloproteinases and tissue inhibitors of metalloproteinases in blood and tissues. Clinical and experimental applications. Annals of the New York Academy of Sciences, 878, 212–227.
Wu Z, Lee ST, Qiao Y, Li Z, Lee PL, Lee YJ, Jiang X, Tan J, Aau M, Lim CZ, Yu Q: Polycomb protein EZH2 regulates cancer cell fate decision in response to DNA damage. Cell Death and Differentiation, 18:1771-1779.
Acknowledgments
This work has been supported, in part, by AIRC (Associazione Italiana per la Ricerca sul Cancro) to Guido Bocci and by MIUR-PRIN2008 project 20084TASKL_004: “Epigenetic manipulation and reversal of resistance to irinotecan in human colorectal cancer cell lines” to Romano Danesi.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Crea, F., Fornaro, L., Bocci, G. et al. EZH2 inhibition: targeting the crossroad of tumor invasion and angiogenesis. Cancer Metastasis Rev 31, 753–761 (2012). https://doi.org/10.1007/s10555-012-9387-3
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10555-012-9387-3