Abstract
Purpose
To determine whether there is a correlation between the presence of peritumoral edema detected on T2-weighted sequences and pathological prognostic factors and specific biological subtypes of breast cancer.
Methods
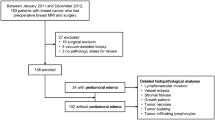
A retrospective study on 120 women with invasive breast cancer, who underwent breast MRI on a 3 T scanner over a period of 12 months, was performed. Peritumoral edema was assessed visually by two breast radiologist in consensus and patients were consequently divided into two groups. For each lesion dimensions, histology and molecular profiling were evaluated. The comparison between the two groups was performed using the χ2 test, subsequently carrying out a univariate and a multivariate logistic regression analysis.
Results
41 (34.2%) invasive breast cancers with perilesional edema and 79 (65.8%) without edema were identified. A significant association between the presence of perilesional edema and a larger lesion size (p = 0.0001), grade 3 (p < 0.001) and a higher Ki-67 index (p < 0.001) was found. The multivariate analysis confirmed that a larger tumor size is independently associated with peritumoral edema. A total of 101 (84.2%) luminal-like, 8 (6.7%) HER2-positive, and 11 (9.2%) triple-negative tumors were included in the study. Peritumoral edema turned out to be less frequently associated with luminal-like lesions (p < 0.001), while the luminal A status was found to have a significant association with the absence of peritumoral edema in the univariate analysis.
Conclusion
The detection of peritumoral edema on T2-weighted sequences should be considered as a valid additional prognostic tool in the evaluation of breast cancer, since it is associated with biologically aggressive non-luminal breast cancers, characterized by large dimension, high tumor grade, and high Ki-67 values.





Similar content being viewed by others
References
Bray F, Ferlay J, Soerjomataram I et al (2018) Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin 66:394–424
Mann RM, Balleyguier C, Baltzer PA et al (2015) Breast MRI: EUSOBI recommendations for women’s information. Eur Radiol 25:3669–3678
Sardanelli F, Boetes C, Borisch B et al (2010) Magnetic resonance imaging of the breast: recommendations from the EUSOMA working group. Eur J Cancer 46:1296–1316
Pinker K, Helbich TH, Morris EA (2017) The potential of multiparametric MRI of the breast. Br J Radiol 90:20160715
Uematsu T, Kasami M, Watanabe J (2014) Is evaluation of the presence of prepectoral edema on T2-weighted with fat-suppression 3 T breast MRI a simple and readily available noninvasive technique for estimation of prognosis in patients with breast cancer? Breast Cancer 21:684–692
ACR (American College of Radiology) (Revised 2018) ACR practice parameter for the performance of contrast-enhanced magnetic resonance imaging (MRI) of the breast. Available at https://www.acr.org/-/media/ACR/Files/Practice-Parameters/mr-contrast-breast.pdf. Accessed 7 August 2019
Arponen O, Masarwah A, Sutela A et al (2016) Incidentally detected enhancing lesions found in breast MRI: analysis of apparent diffusion coefficient and T2 signal intensity significantly improves specificity. Eur Radiol 26:4361–4370
Uematsu T (2015) Focal breast edema associated with malignancy on T2-weighted images of breast MRI: peritumoral edema, prepectoral edema, and subcutaneous edema. Breast Cancer 22:66–70
Baltzer PA, Dietzel M, Kaiser WA (2011) Nonmass lesions in magnetic resonance imaging of the breast: additional T2-weighted images improve diagnostic accuracy. J Comput Assist Tomogr 35:361–366
Kawashima H, Kobayashi-Yoshida M, Matsui O et al (2010) Peripheral hyperintense pattern on T2-weighted magnetic resonance imaging (MRI) in breast carcinoma: correlation with early peripheral enhancement on dynamic MRI and histopathologic findings. J Magn Reson Imaging 32:1117–1123
Baltzer PA, Yang F, Dietzel M et al (2010) Sensitivity and specificity of unilateral edema on T2w-TSE sequences in MR-Mammography considering 974 histologically verified lesions. Breast J 16:233–239
Cx Wu, Lin GS, Lin ZX et al (2015) Peritumoral edema on magnetic resonance imaging predicts a poor clinical outcome in malignant glioma. Oncol Lett 10:2769–2776
Schoenegger K, Oberndorfer S, Wuschitz B et al (2009) Peritumoral edema on MRI at initial diagnosis: an independent prognostic factor for glioblastoma? Eur J Neurol 16:874–878
Schmitz AM, Loo CE, Wesseling J et al (2014) Association between rim enhancement of breast cancer on dynamic contrast-enhanced MRI and patient outcome: impact of subtype. Breast Cancer Res Treat 148:541–551
Costantini M, Belli P, Distefano D et al (2012) Magnetic resonance imaging features in triple-negative breast cancer: comparison with luminal and HER2-overexpressing tumors. Clin Breast Cancer 12:331–339
Song SE, Shin SU, Moon HG et al (2017) MR imaging features associated with distant metastasis-free survival of patients with invasive breast cancer: a case–control study. Breast Cancer Res Treat 162:559–569
Goldhirsch A, Winer EP, Coates AS et al (2013) Personalizing the treatment of women with early breast cancer: highlights of the St Gallen International Expert Consensus on the Primary Therapy of Early Breast Cancer 2013. Ann Oncol 24:2206–2223
Morris EA, Comstock CE, Lee CH et al. (2013) ACR BI-RADS Magnetic Resonance Imaging. In: ACR BI-RADS atlas, breast imaging reporting and data system, 5th edn. American College of Radiology, Reston
Fitzgibbons PL, Murphy DA, Hammond ME et al (2010) Recommendations for validating estrogen and progesterone receptor immunohistochemistry assays. Arch Pathol Lab Med 134:930–935
Marino MA, Helbich T, Baltzer P et al (2018) Multiparametric MRI of the breast: a review. J Magn Reson Imaging 47:301–315
Mann RM, Cho N, Moy L (2019) Breast MRI: state of the art. Radiology 292:520–536
Galati F, Luciani ML, Caramanico C et al (2019) Breast magnetic resonance spectroscopy at 3 T in biopsy-proven breast cancers: does Choline peak correlate with prognostic factors? Invest Radiol 54:767–773
Ballesio L, Savelli S, Angeletti M et al (2009) Breast MRI: are T2 IR sequences useful in the evaluation of breast lesions? Eur J Radiol 71:96–101
Westra C, Dialani V, Mehta TS, at al. (2014) Using T2-weighted sequences to more accurately characterize breast masses seen on MRI. AJR Am J Roentgenol 202:W183–190
Gutman DA, Cooper LA, Hwang SN et al (2013) MR imaging predictors of molecular profile and survival: multi-institutional study of the TCGA glioblastoma data set. Radiology 267:560–569
Hou J, Kshettry VR, Selman WR et al (2013) Peritumoral brain edema in intracranial meningiomas: the emergence of vascular endothelial growth factor-directed therapy. Neurosurg Focus 35:E2
Cheon H, Kim HJ, Kim TH et al (2018) Invasive breast cancer: prognostic value of peritumoral edema identified at preoperative MR imaging. Radiology 287:68–75
Bae MS, Shin SU, Ryu HS et al (2016) Pretreatment MR imaging features of triple-negative breast cancer: association with Response to neoadjuvant chemotherapy and recurrence-free survival. Radiology 281:392–400
Dietzel M, Baltzer PA, Vag T et al (2010) Magnetic resonance mammography of invasive lobular versus ductal carcinoma: systematic comparison of 811 patients reveals high diagnostic accuracy irrespective of typing. J Comput Assist Tomogr 34:587–595
Tang P, Tse GM (2016) Immunohistochemical surrogates for molecular classification of breast carcinoma: a 2015 update. Arch Pathol Lab Med 140:806–814
Trimboli RM, Verardi N, Cartia F et al (2014) Breast cancer detection using double reading of unenhanced MRI including T1-weighted, T2-weighted STIR, and diffusion-wighted imaging: a proof of concept study. AJR Am J Roentgenol 203:674–681
Funding
No funding was received.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
All Authors declare that they have no conflict of interest.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Informed consent
The study was approved by the local institutional review board, and the requirement for informed consent was waived because of the retrospective nature of this study.
Research involving animal and animal rights
This article does not contain any studies with animals performed by any of the authors.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Panzironi, G., Moffa, G., Galati, F. et al. Peritumoral edema as a biomarker of the aggressiveness of breast cancer: results of a retrospective study on a 3 T scanner. Breast Cancer Res Treat 181, 53–60 (2020). https://doi.org/10.1007/s10549-020-05592-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10549-020-05592-8