Abstract
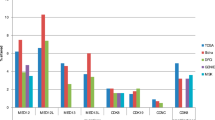
Small Ubiquitin-like Modifier proteins (or SUMO) modify the function of protein substrates involved in various cellular processes including DNA damage response (DDR). It is becoming apparent that dysregulated SUMO contribute to carcinogenesis by affecting post-transcriptional modification of key proteins. It is hypothesised that SUMO contributes to the aggressive nature of breast cancer particularly those associated with features similar to breast carcinoma arising in patients with BRCA1 germline mutations. This study aims to assess the clinical and biological significance of three members of SUMO in a well-characterised annotated series of BC with emphasis on DDR. The study cohort comprised primary operable invasive BC including tumours from patients with known BRCA1 germline mutations. SUMO proteins PIAS1, PIAS4 and UBC9 were assessed using immunohistochemistry utilising tissue microarray technology. Additionally, their expression was assessed using reverse phase protein microarray utilising different cell lines. PIAS1 and UBC9 showed cytoplasmic and/or nuclear expression while PIAS4 was detected only in the nuclei. There was a correlation between subcellular localisation and expression of the nuclear transport protein KPNA2. Tumours showing positive nuclear/negative cytoplasmic expression of SUMO featured good prognostic characteristics including lower histologic grade and had a good outcome. Strong correlation with DDR-related proteins including BRCA1, Rad51, ATM, CHK1, DNA-PK and KU70/KU80 was observed. Correlation with ER and BRCA1 was confirmed using RPPA on cell lines. SUMO proteins seem to play important role in BC. Not only expression but also subcellular location is associated with BC phenotype.





Similar content being viewed by others
References
Pinder JB, Attwood KM, Dellaire G (2013) Reading, writing, and repair: the role of ubiquitin and the ubiquitin-like proteins in DNA damage signaling and repair. Front Genet 4:45
Bologna S, Ferrari S (2013) It takes two to tango: ubiquitin and SUMO in the DNA damage response. Front Genet 4:106
Al-Hakim A et al (2010) The ubiquitous role of ubiquitin in the DNA damage response. DNA Repair (Amst) 9(12):1229–1240
Matunis MJ, Coutavas E, Blobel G (1996) A novel ubiquitin-like modification modulates the partitioning of the Ran-GTPase-activating protein RanGAP1 between the cytosol and the nuclear pore complex. J Cell Biol 135(6 Pt 1):1457–1470
Mahajan R et al (1997) A small ubiquitin-related polypeptide involved in targeting RanGAP1 to nuclear pore complex protein RanBP2. Cell 88(1):97–107
Morris JR (2010) More modifiers move on DNA damage. Cancer Res 70(10):3861–3863
Mo YY et al (2005) A role for Ubc9 in tumorigenesis. Oncogene 24(16):2677–2683
Moschos SJ, Mo YY (2006) Role of SUMO/Ubc9 in DNA damage repair and tumorigenesis. J Mol Histol 37(5–7):309–319
Moschos SJ et al (2010) Expression analysis of Ubc9, the single small ubiquitin-like modifier (SUMO) E2 conjugating enzyme, in normal and malignant tissues. Hum Pathol 41(9):1286–1298
Hoefer J et al (2012) PIAS1 is increased in human prostate cancer and enhances proliferation through inhibition of p21. Am J Pathol 180(5):2097–2107
Agboola A et al (2013) PIASgamma expression in relation to clinicopathological, tumour factors and survival in indigenous black breast cancer women. J Clin Pathol. doi:10.1136/jclinpath-2013-201658
Alshareeda AT et al (2013) Clinicopathological significance of KU70/KU80, a key DNA damage repair protein in breast cancer. Breast Cancer Res Treat 139(2):301–310
Rakha EA et al (2006) Morphological and immunophenotypic analysis of breast carcinomas with basal and myoepithelial differentiation. J Pathol 208(4):495–506
Rakha EA et al (2009) Triple-negative breast cancer: distinguishing between basal and nonbasal subtypes. Clin Cancer Res 15(7):2302–2310
Alshareeda AT et al (2012) Fatty acid binding protein 7 expression and its sub-cellular localization in breast cancer. Breast Cancer Res Treat 134(2):519–529
Aleskandarany MA et al (2012) Prognostic value of proliferation assay in the luminal, HER2-positive, and triple-negative biologic classes of breast cancer. Breast Cancer Res 14(1):R3
Bergink S, Jentsch S (2009) Principles of ubiquitin and SUMO modifications in DNA repair. Nature 458(7237):461–467
Sachdev S et al (2001) PIASy, a nuclear matrix-associated SUMO E3 ligase, represses LEF1 activity by sequestration into nuclear bodies. Genes Dev 15(23):3088–3103
Sentis S et al (2005) Sumoylation of the estrogen receptor alpha hinge region regulates its transcriptional activity. Mol Endocrinol 19(11):2671–2684
Morris JR et al (2009) The SUMO modification pathway is involved in the BRCA1 response to genotoxic stress. Nature 462(7275):886–890
Qin Y et al (2011) Ubc9 mediates nuclear localization and growth suppression of BRCA1 and BRCA1a proteins. J Cell Physiol 226(12):3355–3367
Chen SF et al (2011) Ubc9 expression predicts chemoresistance in breast cancer. Chin J Cancer 30(9):638–644
Agboola AO et al (2014) Clinicopathological and molecular significance of Sumolyation marker (ubiquitin conjugating enzyme 9 (UBC9)) expression in breast cancer of black women. Pathol Res Pract 210(1):10–17
Clevenger CV (2004) Roles and regulation of stat family transcription factors in human breast cancer. Am J Pathol 165(5):1449–1460
Wei J et al (2011) mRNA expression of BRCA1, PIAS1, and PIAS4 and survival after second-line docetaxel in advanced gastric cancer. J Natl Cancer Inst 103(20):1552–1556
Coppola D et al (2009) Substantially reduced expression of PIAS1 is associated with colon cancer development. J Cancer Res Clin Oncol 135(9):1287–1291
Baek SH (2006) A novel link between SUMO modification and cancer metastasis. Cell Cycle 5(14):1492–1495
Chook Y, Blobel G (2001) Karyopherins and nuclear import. Curr Opin Struct Biol 11(6):703–715
Xu J et al (2009) A novel mechanism whereby BRCA1/1a/1b fine tunes the dynamic complex interplay between SUMO-dependent/independent activities of Ubc9 on E2-induced ERalpha activation/repression and degradation in breast cancer cells. Int J Oncol 34(4):939–949
Martin RW et al (2007) RAD51 up-regulation bypasses BRCA1 function and is a common feature of BRCA1-deficient breast tumors. Cancer Res 67(20):9658–9665
Abdel-Fatah TM et al (2010) The biological, clinical and prognostic implications of p53 transcriptional pathways in breast cancers. J Pathol 220(4):419–434
Aleskandarany MA et al (2011) MIB1/Ki-67 labelling index can classify grade 2 breast cancer into two clinically distinct subgroups. Breast Cancer Res Treat 127(3):591–599
Kahyo T, Nishida T, Yasuda H (2001) Involvement of PIAS1 in the sumoylation of tumor suppressor p53. Mol Cell 8(3):713–718
Park MA et al (2008) SUMO1 negatively regulates BRCA1-mediated transcription, via modulation of promoter occupancy. Nucleic Acids Res 36(1):263–283
Cortez D et al (1999) Requirement of ATM-dependent phosphorylation of brca1 in the DNA damage response to double-strand breaks. Science 286(5442):1162–1166
Mohanty S et al (2014) ROS-PIASgamma cross talk channelizes ATM signaling from resistance to apoptosis during chemosensitization of resistant tumors. Cell Death Dis 5:e1021
Acknowledgments
Alaa Alshareeda is sponsored by the higher education of kingdom of Saudi Arabia.
Conflict of interest
None.
Funding
No financial relationship with the organisation that sponsored the research.
Author information
Authors and Affiliations
Corresponding author
Additional information
Ola H. Negm is first joint author for this study.
Rights and permissions
About this article
Cite this article
Alshareeda, A.T., Negm, O.H., Green, A.R. et al. SUMOylation proteins in breast cancer. Breast Cancer Res Treat 144, 519–530 (2014). https://doi.org/10.1007/s10549-014-2897-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10549-014-2897-7