Abstract
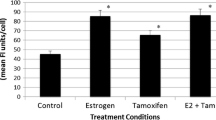
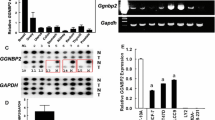
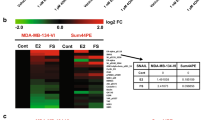
We previously showed that COUP-TFI interacts with the Estrogen Receptor alpha (ERα) to recruit Extracellular signal Regulated Kinases (ERKs) in an Estradiol (E2)-independent manner, resulting in an enhancement of ERα transcriptional activity. However, the involvement of COUP-TFI in physiologically relevant functions of ERα, such as the mitogenic activity that E2 has on breast cancer cells, remains poorly understood. Here, we first showed that the amounts of COUP-TFI protein are higher in dedifferentiated mammary cell lines (MDA-MB-231) and tumor breast cells as compared to the differentiated MCF-7 cell line and normal breast cells. To evaluate the functional relevance of the COUP-TFI/ERα interplay in mammary cells, we generated MCF-7 cells that stably over-express COUP-TFI. We found that the over-expression of COUP-TFI enhances motility and invasiveness of MCF-7 cells. COUP-TFI also promotes the proliferation of MCF-7 cells through ERα-dependent mechanisms that target cell cycle progression and cell survival. To further investigate the mechanisms underlying these effects of COUP-TFI, we evaluated the expression of known E2-target genes in breast cancer, and found that COUP-TFI differentially regulated genes involved in cell proliferation, apoptosis, and migration/invasion. Notably, Cathepsin D (CTSD) transcript and protein levels were significantly higher in presence and absence of E2 in MCF-7 over-expressing COUP-TFI. Chromatin Immunoprecipitation assays showed that ERα, phospho-RNA Polymerase II, as well as p68 RNA Helicase, a phospho-Serine 118 dependent co-activator of ERα, were preferentially recruited onto the CTSD gene proximal promoter in COUP-TFI over-expressing cells. These results suggest that COUP-TFI selectively regulates the expression of endogenous E2-target genes and consequently modifies ERα positive mammary cells response to E2.






Similar content being viewed by others
References
Sommer S, Fuqua SA (2001) Estrogen receptor and breast cancer. Semin Cancer Biol 11(5):339–352
Nilsson S, Makela S, Treuter E et al (2001) Mechanisms of estrogen action. Physiol Rev 81(4):1535–1565
Safe S (2001) Transcriptional activation of genes by 17 beta-estradiol through estrogen receptor-Sp1 interactions. Vitam Horm 62:231–252
Klinge CM (2000) Estrogen receptor interaction with co-activators and co-repressors. Steroids 65(5):227–251
Endoh H, Maruyama K, Masuhiro Y et al (1999) Purification and identification of p68 RNA helicase acting as a transcriptional coactivator specific for the activation function 1 of human estrogen receptor alpha. Mol Cell Biol 19(8):5363–5372
Watanabe M, Yanagisawa J, Kitagawa H et al (2001) A subfamily of RNA-binding DEAD-box proteins acts as an estrogen receptor alpha coactivator through the N-terminal activation domain (AF-1) with an RNA coactivator, SRA. Embo J 20(6):1341–1352
Gadducci A, Biglia N, Sismondi P et al (2005) Breast cancer and sex steroids: critical review of epidemiological, experimental and clinical investigations on etiopathogenesis, chemoprevention and endocrine treatment of breast cancer. Gynecol Endocrinol 20(6):343–360
Foster JS, Henley DC, Ahamed S et al (2001) Estrogens and cell-cycle regulation in breast cancer. Trends Endocrinol Metab 12(7):320–327
Gompel A, Somai S, Chaouat M et al (2000) Hormonal regulation of apoptosis in breast cells and tissues. Steroids 65(10–11):593–598
Osborne CK (1998) Steroid hormone receptors in breast cancer management. Breast Cancer Res Treat 51(3):227–238
Frasor J, Danes JM, Komm B et al (2003) Profiling of estrogen up- and down-regulated gene expression in human breast cancer cells: insights into gene networks and pathways underlying estrogenic control of proliferation and cell phenotype. Endocrinology 144(10):4562–4574
Deblois G, Giguere V (2003) Ligand-independent coactivation of ERalpha AF-1 by steroid receptor RNA activator (SRA) via MAPK activation. J Steroid Biochem Mol Biol 85(2–5):123–131
Métivier R, Gay FA, Hubner MR et al (2002) Formation of an hER alpha-COUP-TFI complex enhances hER alpha AF-1 through Ser118 phosphorylation by MAPK. Embo J 21(13):3443–3453
Burbach JP, Lopes da Silva S, Cox JJ et al (1994) Repression of estrogen-dependent stimulation of the oxytocin gene by chicken ovalbumin upstream promoter transcription factor I. J Biol Chem 269(21):15046–15053
Liu Y, Yang N, Teng CT (1993) COUP-TF acts as a competitive repressor for estrogen receptor-mediated activation of the mouse lactoferrin gene. Mol Cell Biol 13(3):1836–1846
Tsai SY, Tsai MJ (1997) Chick ovalbumin upstream promoter–transcription factors (COUP-TFs): coming of age. Endocr Rev 18(2):229–240
Pereira FA, Qiu Y, Zhou G et al (1999) The orphan nuclear receptor COUP-TFII is required for angiogenesis and heart development. Genes Dev 13(8):1037–1049
Pereira FA, Tsai MJ, Tsai SY (2000) COUP-TF orphan nuclear receptors in development and differentiation. Cell Mol Life Sci 57(10):1388–1398
Qiu Y, Pereira FA, DeMayo FJ et al (1997) Null mutation of mCOUP-TFI results in defects in morphogenesis of the glossopharyngeal ganglion, axonal projection, and arborization. Genes Dev 11(15):1925–1937
Kieback DG, Levi T, Kohlberger P et al (1996) Chicken ovalbumin upstream promoter-transcription factor (COUP-TF) expression in human endometrial cancer cell lines. Anticancer Res 16(6B):3371–3376
Kieback DG, Runnebaum IB, Moebus VJ et al (1993) Chicken ovalbumin upstream promoter transcription factor (COUP-TF): an orphan steroid receptor with a specific pattern of differential expression in human ovarian cancer cell lines. Gynecol Oncol 51(2):167–170
Navab R, Gonzalez-Santos JM, Johnston MR et al (2004) Expression of chicken ovalbumin upstream promoter-transcription factor II enhances invasiveness of human lung carcinoma cells. Cancer Res 64(15):5097–5105
Merot Y, Metivier R, Penot G et al (2004) The relative contribution exerted by AF-1 and AF-2 transactivation functions in estrogen receptor alpha transcriptional activity depends upon the differentiation stage of the cell. J Biol Chem 279(25):26184–26191
Metivier R, Penot G, Hubner MR et al (2003) Estrogen receptor-alpha directs ordered, cyclical, and combinatorial recruitment of cofactors on a natural target promoter. Cell 115(6):751–763
Keepers YP, Pizao PE, Peters GJ et al (1991) Comparison of the sulforhodamine B protein and tetrazolium (MTT) assays for in vitro chemosensitivity testing. Eur J Cancer 27(7):897–900
Lacroix M, Leclercq G (2004) Relevance of breast cancer cell lines as models for breast tumours: an update. Breast Cancer Res Treat 83(3):249–289
Klinge CM, Silver BF, Driscoll MD et al (1997) Chicken ovalbumin upstream promoter-transcription factor interacts with estrogen receptor, binds to estrogen response elements and half-sites, and inhibits estrogen-induced gene expression. J Biol Chem 272(50):31465–31474
Perry RR, Kang Y, Greaves B (1995) Effects of tamoxifen on growth and apoptosis of estrogen-dependent and -independent human breast cancer cells. Ann Surg Oncol 2(3):238–245
Platet N, Cathiard AM, Gleizes M et al (2004) Estrogens and their receptors in breast cancer progression: a dual role in cancer proliferation and invasion. Crit Rev Oncol Hematol 51(1):55–67
Platet N, Cunat S, Chalbos D et al (2000) Unliganded and liganded estrogen receptors protect against cancer invasion via different mechanisms. Mol Endocrinol 14(7):999–1009
Vincent-Salomon A, Thiery JP (2003) Host microenvironment in breast cancer development: epithelial-mesenchymal transition in breast cancer development. Breast Cancer Res 5(2):101–106
Adam F, Sourisseau T, Metivier R et al (2000) COUP-TFI (chicken ovalbumin upstream promoter-transcription factor I) regulates cell migration and axogenesis in differentiating P19 embryonal carcinoma cells. Mol Endocrinol 14(12):1918–1933
Garbett EA, Reed MW, Stephenson TJ et al (2000) Proteolysis in human breast cancer. Mol Pathol 53(2):99–106
Marshman E, Green KA, Flint DJ et al (2003) Insulin-like growth factor binding protein 5 and apoptosis in mammary epithelial cells. J Cell Sci 116(Pt 4):675–682
Williams TM, Medina F, Badano I et al (2004) Caveolin-1 gene disruption promotes mammary tumorigenesis and dramatically enhances lung metastasis in vivo. Role of Cav-1 in cell invasiveness and matrix metalloproteinase (MMP-2/9) secretion. J Biol Chem 279(49):51630–51646
Sparatore B, Patrone M, Passalacqua M et al (2005) Activation of A431 human carcinoma cell motility by extracellular high-mobility group box 1 protein and epidermal growth factor stimuli. Biochem J 389(Pt 1):215–221
More E, Fellner T, Doppelmayr H et al (2003) Activation of the MAP kinase pathway induces chicken ovalbumin upstream promoter-transcription factor II (COUP-TFII) expression in human breast cancer cell lines. J Endocrinol 176(1):83–94
Nakshatri H, Mendonca MS, Bhat-Nakshatri P et al (2000) The orphan receptor COUP-TFII regulates G2/M progression of breast cancer cells by modulating the expression/activity of p21(WAF1/CIP1), cyclin D1, and cdk2. Biochem Biophys Res Commun 270(3):1144–1153
Lin B, Chen GQ, Xiao D et al (2000) Orphan receptor COUP-TF is required for induction of retinoic acid receptor beta, growth inhibition, and apoptosis by retinoic acid in cancer cells. Mol Cell Biol 20(3):957–970
Lannigan DA (2003) Estrogen receptor phosphorylation. Steroids 68(1):1–9
Rochette-Egly C (2003) Nuclear receptors: integration of multiple signalling pathways through phosphorylation. Cell Signal 15(4):355–366
Britton DJ, Hutcheson IR, Knowlden JM et al (2006) Bidirectional cross talk between ERalpha and EGFR signalling pathways regulates tamoxifen-resistant growth. Breast Cancer Res Treat 96(2):131–146
Bunone G, Briand PA, Miksicek RJ et al (1996) Activation of the unliganded estrogen receptor by EGF involves the MAP kinase pathway and direct phosphorylation. Embo J 15(9):2174–2183
Fujita T, Kobayashi Y, Wada O et al (2003) Full activation of estrogen receptor alpha activation function-1 induces proliferation of breast cancer cells. J Biol Chem 278(29):26704–26714
Bailey P, Sartorelli V, Hamamori Y et al (1998) The orphan nuclear receptor, COUP-TF II, inhibits myogenesis by post-transcriptional regulation of MyoD function: COUP-TF II directly interacts with p300 and myoD. Nucleic Acids Res 26(23):5501–5510
Eubank DW, Duplus E, Williams SC et al (2001) Peroxisome proliferator-activated receptor gamma and chicken ovalbumin upstream promoter transcription factor II negatively regulate the phosphoenolpyruvate carboxykinase promoter via a common element. J Biol Chem 276(32):30561–30569
Rochefort H, Garcia M, Glondu M et al (2000) Cathepsin D in breast cancer: mechanisms and clinical applications, a 1999 overview. Clin Chim Acta 291(2):157–170
Hall JM, Korach KS (2003) Stromal cell-derived factor 1, a novel target of estrogen receptor action, mediates the mitogenic effects of estradiol in ovarian and breast cancer cells. Mol Endocrinol 17(5):792–803
Damiens E, El Yazidi I, Mazurier J et al (1999) Lactoferrin inhibits G1 cyclin-dependent kinases during growth arrest of human breast carcinoma cells. J Cell Biochem 74(3):486–498
Cassoni P, Sapino A, Marrocco T et al (2004) Oxytocin and oxytocin receptors in cancer cells and proliferation. J Neuroendocrinol 16(4):362–364
Le Goff P, Montano MM, Schodin DJ et al (1994) Phosphorylation of the human estrogen receptor. Identification of hormone-regulated sites and examination of their influence on transcriptional activity. J Biol Chem 269:4458–4466
Giamarchi C, Solanas M, Chailleux C et al (1999) Chromatin structure of the regulatory regions of pS2 and cathepsin D genes in hormone-dependent and -independent breast cancer cell lines. Oncogene 18(2):533–541
Chalbos D, Philips A, Galtier F et al (1993) Synthetic antiestrogens modulate induction of pS2 and cathepsin-D messenger ribonucleic acid by growth factors and adenosine 3′,5′-monophosphate in MCF7 cells. Endocrinology 133(2):571–576
Wang F, Duan R, Chirgwin J et al (2000) Transcriptional activation of cathepsin D gene expression by growth factors. J Mol Endocrinol 24(2):193–202
Acknowledgments
We are grateful to Pr. Jean Leveque for the initial discussions concerning immunohistochemistry analysis. This work was supported by a fellowship from the Ministère de l’Enseignement et de la Recherche (to F.L.D.) and funds from the Association pour la Recherche sur le Cancer (ARC; contract no. 4487), the CNRS and the University of Rennes I.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Le Dily, F., Métivier, R., Guéguen, MM. et al. COUP-TFI modulates estrogen signaling and influences proliferation, survival and migration of breast cancer cells. Breast Cancer Res Treat 110, 69–83 (2008). https://doi.org/10.1007/s10549-007-9693-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10549-007-9693-6