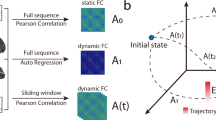
Abstract
Recent studies have shown the importance of graph theory in analyzing characteristic features of functional networks of the human brain. However, many of these explorations have focused on static patterns of a representative graph that describe the relatively long-term brain activity. Therefore, this study established and characterized functional networks based on the synchronization likelihood and graph theory. Quasidynamic graphs were constructed simply by dividing a long-term static graph into a sequence of subgraphs that each had a timescale of 1 s. Irregular changes were then used to investigate differences in human brain networks between resting and math-operation states using magnetoencephalography, which may provide insights into the functional substrates underlying logical reasoning. We found that graph properties could differ from brain frequency rhythms, with a higher frequency indicating a lower small-worldness, while changes in human brain state altered the functional networks into more-centralized and segregated distributions according to the task requirements. Time-varying connectivity maps could provide detailed information about the structure distribution. The frontal theta activity represents the essential foundation and may subsequently interact with high-frequency activity in cognitive processing.





Similar content being viewed by others
References
Achard S, Bullmore E (2007) Efficiency and cost of economical brain functional networks. PLoS Comput Biol 3(2):e17
Barrat A, Barthelemy M, Pastor-Satorras R, Vespignani A (2004) The architecture of complex weighted networks. Proc Natl Acad Sci USA 101(11):3747–3752
Bosboom JL, Stoffers D, Stam CJ, Berendse HW, Wolters ECh (2009a) Cholinergic modulation of MEG resting-state oscillatory activity in Parkinson’s disease related dementia. Clin Neurophysiol 120(5):910–915
Bosboom JL, Stoffers D, Wolters ECh, Stam CJ, Berendse HW (2009b) MEG resting state functional connectivity in Parkinson’s disease related dementia. J Neural Transm 116(2):193–202
Brainard DH (1997) The psychophysics toolbox. Spat Vis 10(4):433–436
Canolty RT, Knight RT (2010) The functional role of cross-frequency coupling. Trends Cognitive Sci 14(4):506–515
Chang C, Glover GH (2010) Time-frequency dynamics of resting-state brain connectivity measured with fMRI. Neuroimage 50(1):81–98
Chen JL, Ros T, Gruzelier JH (2013) Dynamic changes of ICA-derived EEG functional connectivity in the resting state. Hum Brain Mapp 34(4):852–868
de Cheveigné A, Simon JZ (2007) Denoising based on time-shift PCA. J Neurosci Methods 165(2):297–305
De Luca M, Beckmann CF, De Stefano N, Matthews PM, Smith SM (2006) fMRI resting state networks define distinct modes of long-distance interactions in the human brain. Neuroimage 29(4):1359–1367
Douw L, Schoonheim MM, Landi D, van der Meer ML, Geurts JJ, Reijneveld JC et al (2011) Cognition is related to resting-state small-world network topology: an magnetoencephalographic study. Neuroscience 175:169–177
Freeman LC (1979) Centrality in social networks. Conceptual clarification. Soc Netw 1(3):215–239
Friston KJ (2011) Functional and effective connectivity: a review. Brain Connectivity 1(1):13–36
Gong G, He Y, Concha L, Lebel C, Gross DW, Evans AC et al (2009) Mapping anatomical connectivity patterns of human cerebral cortex using in vivo diffusion tensor imaging tractography. Cereb Cortex 19(3):524–536
Grindrod P (2002) Range-dependent random graphs and their application to modeling large small-world Proteome datasets. Phys Rev E 66(6 Pt 2):066702
Hyvärinen A, Ramkumar P, Parkkonen L, Hari R (2010) Independent component analysis of short-time Fourier transforms for spontaneous EEG/MEG analysis. Neuroimage 49(1):257–271
Ioannides AA (2001) Real time human brain function: observations and inferences from single trial analysis of magnetoencephalographic signals. Clin Electroencephalogr 32(3):98–111
Jensen O, Colgin LL (2007) Cross-frequency coupling between neuronal oscillations. Trends Cognitive Sci 11(7):267–269
Lachaux JP, Rodriguez E, Martinerie J, Varela FJ (1999) Measuring phase synchrony in brain signals. Hum Brain Mapp 8(4):194–208
Langer N, von Bastian CC, Wirz H, Oberauer K, Jancke L (2013) The effects of working memory training on functional brain network efficiency. Cortex 49(9):2424–2438
Liu Z, Fukunaga M, de Zwart JA, Duyn JH (2010) Large-scale spontaneous fluctuations and correlations in brain electrical activity observed with magnetoencephalography. Neuroimage 51(1):102–111
Moewes C, Kruse R, Sabel BA (2013) Analysis of Dynamic Brain Networks Using VAR Models. In: Kruse R et al (eds) Synergies of soft computing and statistics for intelligent data analysis advances in intelligent systems and computing. Springer, Berlin, pp 525–532
Montez T, Linkenkaer-Hansen KB, van Dijk W, Stam CJ (2006) Synchronization likelihood with explicit time-frequency priors. Neuroimage 33(4):1117–1125
Musso F, Brinkmeyer J, Mobascher A, Warbrick T, Winterer G (2010) Spontaneous brain activity and EEG microstates. A novel EEG/fMRI analysis approach to explore resting-state networks. Neuroimage 52(4):1149–1161
Mutlu AY, Bernat E, Aviyente S (2012) A signal-processing-based approach to time-varying graph analysis for dynamic brain network identification. Comput Math Methods Med. doi:10.1155/2012/451516
Navas A, Papo D, Boccaletti S, del-Pozo F, Bajo R, Maestú F et al (2013). Functional hubs in mild cognitive impairment. Int J Bifurc Chaos, arXiv:1307.0969
Newman ME (2004) Analysis of weighted networks. Phys Rev E 70(5 Pt 2):056131
Olde Dubbelink KT, Felius A, Verbunt JP, van Dijk BW, Berendse HW, Stam CJ et al (2008) Increased resting-state functional connectivity in obese adolescents; a magnetoencephalographic pilot study. PLoS One 3(7):e2827
Opsahl T, Agneessens F, Skvoretz J (2010) Node centrality in weighted networks: generalizing degree and shortest paths. Soc Netw 32(3):245–251
Pelli DG (1997) The VideoToolbox software for visual psychophysics: transforming numbers into movies. Spat Vis 10(4):437–442
Prichard J, Theiler D (1994) Generating surrogate data for time series with several simultaneously measured variables. Phys Rev Lett 73(7):951–954
Rubinov M, Sporns O (2010) Complex network measures of brain connectivity: uses and interpretations. Neuroimage 52(3):1059–1069
Schiff SJ, So P, Chang T, Burke RE, Sauer T (1996) Detecting dynamical interdependence and generalized synchrony through mutual prediction in a neural ensemble. Phys Rev E 54(6):6708–6724
Schoonheim MM, Geurts JJ, Landi D, Douw L, van der Meer ML, Vrenken H et al (2013) Functional connectivity changes in multiple sclerosis patients: a graph analytical study of MEG resting state data. Hum Brain Mapp 34(1):52–61
Smit DJ, Stam CJ, Posthuma D, Boomsma DI, de Geus EJ (2008) Heritability of “small-world” networks in the brain: a graph theoretical analysis of resting-state EEG functional connectivity. Hum Brain Mapp 29(12):1368–1378
Smit DJ, Boersma M, van Beijsterveldt CE, Posthuma D, Boomsma DI, Stam CJ et al (2010) Endophenotypes in a dynamically connected brain. Behav Genet 40(2):167–177
Stam CJ (2004) Functional connectivity patterns of human magnetoencephalographic recordings: a ‘small-world’ network? Neurosci Lett 355(1–2):25–28
Stam CJ, van Dijk BW (2002) Synchronization likelihood: an unbiased measure of generalized synchronization in multivariate data sets. Physica D 163(3):236–251
Stam CJ, Jones BF, Manshanden I, van Cappellen van Walsum AM, Montez T, Verbunt T et al (2006) Magnetoencephalographic evaluation of resting-state functional connectivity in Alzheimer’s disease. Neuroimage 32(3):1335–1344
Stam CJ, de Haan W, Daffertshofer A, Jones BF, Manshanden I, van Walsum AMVC et al (2009) Graph theoretical analysis of magnetoencephalographic functional connectivity in Alzheimer’s disease. Brain 132(Pt 1):213–224
Stoffers D, Bosboom JL, Deijen JB, Wolters ECh, Stam CJ, Berendse HW (2008) Increased cortico-cortical functional connectivity in early-stage Parkinson’s disease: an MEG study. Neuroimage 41(2):212–222
Tan B, Kong X, Yang P, Jin Z, Li L (2013) The difference of brain functional connectivity between eyes-closed and eyes-open using graph theoretical analysis. Comput Math Methods Med. doi:10.1155/2013/976365
Tian L, Wang J, Yan C, He Y (2011) Hemisphere- and gender-related differences in small-world brain networks: a resting-state functional MRI study. Neuroimage 54(1):191–202
van den Heuvel MP, Hulshoff Pol HE (2010) Exploring the brain network: a review on resting-state fMRI functional connectivity. Eur Neuropsychopharmacol 20(8):519–534
van Diessen E, Otte WM, Braun KP, Stam CJ, Jansen FE (2013) Improved diagnosis in children with partial epilepsy using a multivariable prediction model based on EEG network characteristics. PLoS One 8(4):e59764
van Wijk BC, Stam CJ, Daffertshofer A (2010) Daffertshofer, Comparing brain networks of different size and connectivity density using graph theory. PLoS One 5(10):e13701
Wang Z, Liu J, Zhong N, Qin Y, Zhou H, Li K (2012) Changes in the brain intrinsic organization in both on-task state and post-task resting state. Neuroimage 62(1):394–407
Watts DJ, Strogatz SH (1998) Collective dynamics of ‘small-world’ networks. Nature 393(6684):440–442
Wu L, Eichele T, Calhoun VD (2010) Reactivity of hemodynamic responses and functional connectivity to different states of alpha synchrony: a concurrent EEG-fMRI study. Neuroimage 52(4):1252–1260
Zuo XN, Ehmke R, Mennes M, Imperati D, Castellanos FX, Sporns O et al (2012) Network centrality in the human functional connectome. Cereb Cortex 22(8):1862–1875
Acknowledgments
This study was supported in part by research grants from National Science Council (NSC-101-2410-H-130-025-MY2, NSC 100-2628-E-010-002-MY3), Taiwan.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Yang, CY., Lin, CP. Time-Varying Network Measures in Resting and Task States Using Graph Theoretical Analysis. Brain Topogr 28, 529–540 (2015). https://doi.org/10.1007/s10548-015-0432-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10548-015-0432-8