Abstract
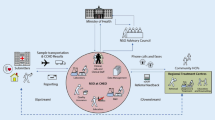
In a survey conducted in 2010/2011 data from the 28 EU member states, four EU candidate states (Croatia, FYROM, Iceland, Turkey), three potential EU candidate states (Bosnia Herzegovina, Montenegro, Serbia), and two EFTA states (Norway and Switzerland) were collected. The status and function of newborn screening (NBS) programmes were investigated from the information to prospective parents and the public via confirmation of a positive screening result up to decisions on treatment. This article summarises the results from screening laboratory findings to start of treatment. In addition we asked about the existence of feedback loops reporting the conclusions of confirmation of screening results to the screening laboratory and communication of long-term outcome to diagnostic units and possibly existing central registries. Parallel to the description of actual practices of where, how and by whom the different steps of the programmes are executed, we also asked for the existence of guidelines or directives regulating the screening programmes, material to support information of parents about diagnoses and treatment and training facilities for professionals involved in the programmes. This survey gives a first comprehensive overview of the steps following a positive screening result in European NBS programmes. The 37 data sets reveal substantial variation of national screening panels, but also a lot of similarities. Analysis across all countries revealed that actual practice is often organised but not regulated by guidelines. Material to inform patients is available more often for explaining treatment (69 %) than explaining the necessity of confirmatory diagnostics (41 %). Training of professionals is rarely regulated by a guideline (2 %), but is offered for paediatricians (40 %) and dieticians (29 %) and only rarely for other professions (e.g. geneticists, clinical nurse specialists, psychologists). Registry-based evaluation of long-term outcome is as yet almost nonexistent (3 %).





Similar content being viewed by others
References
Auchus RJ, Witchel SF, Leight KR, Aisenberg J, Azziz R, Bachega TA, Baker LA, Baratz AB, Baskin LS, Berenbaum SA, Breault DT, Cerame BI, Conway GS, Eugster EA, Fracassa S, Gearhart JP, Geffner ME, Harris KB, Hurwitz RS, Katz AL, Kalro BN, Lee PA, Alger Lin G, Loechner KJ, Marshall I, Merke DP, Migeon CJ, Miller WL, Nenadovich TL, Oberfield SE, Pass KA, Poppas DP, Lloyd-Puryear MA, Quigley CA, Riepe FG, Rink RC, Rivkees SA, Sandberg DE, Schaeffer TL, Schlussel RN, Schneck FX, Seely EW, Snyder D, Speiser PW, Therrell BL, Vanryzin C, Vogiatzi MG, Wajnrajch MP, White PC, Zuckerman AE (2010) Guidelines for the Development of Comprehensive Care Centers for Congenital Adrenal Hyperplasia: Guidance from the CARES Foundation Initiative. Int J Pediatr Endocrinol 2010:275213
Blumberg MS (1957) Evaluating health screening procedures. Operations Research 5:351–360
Castellani C, Southern K, Brownlee K, Dankert Roelse J, Duff A, Farrell M, Mehta A, Munck A, Pollitt R, Sermet-Gaudelus I, Wilcken B, Ballmann M, Corbetta C, de Monestrol I, Farrell P, Feilcke M, Férec C, Gartner S, K G, Hammermann J, Kashirskaya N, Loeber G, Macek M, G M, Reiman A, Rizzotti P, Sammon A, Dands D, Smyth A, Sommerburg O, Torresani T, Travert G, Vernooji A & Elborn S (2009) European best practice guidelines for cystic fibrosis neonatal screening. Journal of Cystic Fibrosis 8:153–73
Cornel M, Rigter T, Weinreich S, Burgard P, Hoffmann GF, Linder M, Loeber JG, Rupp K, Taruscio D, Vitozzi L (2011) Newborn Screening in Europe; Expert opinion document.http://www.iss.it/cnmr/prog/cont.php?id=1621&lang=1&tipo=64; retrieved 29.02.2012
Drummond MF, Sculpher M.J, Torrance GW, O'Brien BJ, Stoddart GL (2005) Methods for the Economic Evaluation of Health Care Programmes, 3rd edition. Oxford University Press
European Commission (2009) Complete Council Recommendation of 8 June 2009 on an action in the field of rare diseases (2009/C 151/02). Official Journal of the European Communities C151:7–10
Fant KE, Clark SJ, Kemper AR (2005) Completeness and complexity of information available to parents from newborn-screening programmes. Pediatrics 115(5):1268–1272
Heringer J, Boy SP, Ensenauer R, Assmann B, Zschocke J, Harting I, Lücke T, Maier EM, Mühlhausen C, Haege G, Hoffmann GF, Burgard P, Kölker S (2010) Use of guidelines improves the neurological outcome in glutaric aciduria type I. Ann Neurol 68(5):743–752
Hinton C, Feuchtbaum L, Kus C, Kemper A, Berry SA, Levy-Fisch J, Luedtke J, Kays C, Boyle C (2011) What questions should newborn screening long-term follow-up be able to answer? A statement of the US Secretary for Health and Human Services´ Advisory Committee on Heritable Disorders in Newborns and Children. Genetics in Medicine 13(10):861–865
Kölker S, Christensen E, Leonard JV, Greenberg CR, Boneh A, Burlina AB, Burlina AP, Dixon M, Duran M, Garcia Cazorla A, Goodman SI, Koeller DM, Kyllerman M, Mühlhausen C, Müller E, Okun JG, Wilcken B, Hoffmann GF, Burgard P (2011) Diagnosis and management of glutaric aciduria type I–revised recommendations. J Inherit Metab Dis 34(3):677–694
Kölker S, Garbade SF, Greenberg CR, Leonard JV, Saudubray JM, Ribes A, Kalkanoglu HS, Lund AM, Merinero B, Wajner M, Troncoso M, Williams M, Walter JH, Campistol J, Marti-Herrero M, Caswill M, Burlina AB, Lagler F, Maier EM, Schwahn B, Tokatli A, Dursun A, Coskun T, Chalmers RA, Koeller DM, Zschocke J, Christensen E, Burgard P, Hoffmann GF (2006) Natural history, outcome, and treatment efficacy in children and adults with glutaryl-CoA dehydrogenase deficiency. Pediatr Res 59(6):840–847
Lindner M. (2010) Konfirmationsdiagnostik bei Verdacht auf angeborene Stoffwechselkrankheiten aus dem Neugeborenenscreening. Leitlinien der Gesellschaft für Kinderheilkunde und Jugendmedizin (DGKJ) und der Arbeitsgemeinschaft für Pädiatrische Stoffwechselstörungen (APS). Leitline Nr. 027/021. Arbeitsgemeinschaft der Wissenschaftlichen Medizinischen Fachgesellschaften e.V. (AWMF) (http://www.awmf.org/leitlinien/leitlinien-suche.html#result-list, retrieved 2011.12.01)
Lindner M, Gramer G, Haege G, Fang-Hoffmann J, Schwab KO, Tacke U, Trefz FK, Mengel E, Wendel U, Leichsenring M, Burgard P, Hoffmann GF (2011) Efficacy and outcome of expanded newborn screening for metabolic diseases - report of 10 years from South-West Germany. Orphanet J Rare Dis 6(44)
Loeber JG, Burgard P, Cornel MC, Rigter T, Weinreich SS, Rupp, K, Hoffmann GF, Vittozzi L (2012) Newborn screening programmes in Europe; arguments and efforts regarding harmonization. Part 1 - From blood spot to screening result. J Inherit Metab Dis, this issue
Pandor A, Eastham J, Beverley C, Chilcott J, Paisley S (2004) Clinical effectiveness and cost-effectiveness of neonatal screening for inborn errors of metabolism using tandem mass spectrometry: a systematic review. Health Technol Assess 8(12)
Petros M (2011) Revisiting the Wilson-Jungner criteria: How can supplemental criteria guide public health in the era of genetic screening? Genetics in Medicine 14:129–134
Pollitt RJ (2007) Introducing new screens: why are we all doing different things? J Inherit Metab Dis 30(4):423–429
Raffle A, Gray M (2007) Screening – Evidence and practice. Oxford University Press, Oxford
Rembold CM (1998) Number needed to screen: development of a statistic for disease screening. BMJ 317:307–312
van Dyck PC, Edwards ES (2006) A look at newborn screening: today and tomorrow. Pediatrics 117:193
Wilcken B (2011) Newborn screening: how are we travelling, and where should we be going. J Inherit Metab Dis 34(3):569–574
Wilcken B, Haas M, Joy P, Wiley V, Bowling F, Carpenter K, Christodoulou J, Cowley D, Ellaway C, Fletcher J, Kirk E, Lewis B, McGill J, Peters H, Pitt J, Ranieri E, Yaplito-Lee J, Boneh A (2009) Expanded newborn screening: outcome in screened and unscreened patients at age 6 years. Pediatrics 124(2):e241–e248
Wilcken B, Rinaldo P, Matern D (2012) Newborn screening for inborn errors of metabolism. In: Saudubry JM, van den Berghe G, Walter JH Inborn (eds) Metabolic diseases - diagnosis and treatment, 5th edn. Springer, Heidelberg, pp 76–86
Wilson JMG, Jungner G (1968) The principles and practice of screening for disease. Public Health Papers n. 34. Geneva: World Health Organization. (p 11) (retrieved from http://whqlibdoc.who.int/php/WHO_PHP_34.pdf; 15 Nov 2010)
Parents and patient organisations
European Society for Phenylketonuria: http://www.espku.org/, retrieved 22 Nov 2011
European Organization for Rare Diseases: http://www.eurordis.org/, retrieved 22 Nov 2011
Cystic Fibrosis Europe; http://www.cfww.org/cfe/, retrieved 22 Nov 2011
Acknowledgement
We thank all respondents for contributing their data to the survey
This work was funded by the European Union (Contract number 2009 62 06 of the Executive Agency for Health and Consumers) and by the Dietmar Hopp Foundation, St. Leon -Rot.
Conflict of interest
None.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by: Rodney Pollitt
Electronic supplementary material
Below is the link to the electronic supplementary material.
ESM 1
(DOC 279 kb)
Rights and permissions
About this article
Cite this article
Burgard, P., Rupp, K., Lindner, M. et al. Newborn screening programmes in Europe; arguments and efforts regarding harmonization. Part 2 – From screening laboratory results to treatment, follow-up and quality assurance. J Inherit Metab Dis 35, 613–625 (2012). https://doi.org/10.1007/s10545-012-9484-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10545-012-9484-z