Summary
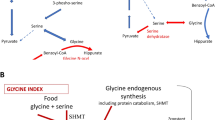
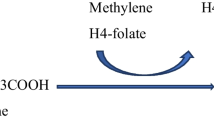
High-dose benzoate treatment aimed at reducing plasma glycine levels to normal reduces seizures and increases wakefulness in patients with nonketotic hyperglycinaemia (NKH). Since benzoate metabolism is dependent on the available glycine pool, and since the glycine pool is variably affected by the deficiency in the glycine cleavage enzyme system, we examined the importance of interpatient variability in benzoate requirement. To correct for the dietary glycine contribution, the glycine index was introduced as the molar requirement of benzoate dose necessary to normalize plasma glycine levels and subtracting from that the dietary glycine intake, both corrected for weight. The glycine index varied between 3.62 and 4.87 mmol/kg per day in five patients with a poor neurodevelopmental outcome and between 0.92 and 1.90 mmol/kg per day in four patients with a better neurodevelopmental outcome, and was 2.54 mmol/kg per day in a single patient with an intermediate outcome. The glycine index was stable over time within each patient. Exceeding the balance by either increasing food glycine intake or decreasing the benzoate dose resulted in increased glycine levels. Exceeding the glycine tolerance by increasing benzoate resulted in elevated and toxic levels of benzoate. The glycine index is a stable, individually specific parameter in patients with NKH. It has clinical consequences for the dose of benzoate required and the role of dietary management. Through its correlation with neurodevelopmental outcome, the glycine index points to potential genetic factors that could contribute to the psychomotor retardation in NKH.
Similar content being viewed by others
References
Aliefendioğlu D, Aslan AT, Coşkun T, Dursun A, Cakmak FN, Kesimer M (2003) Transient nonketotic hyperglycinemia: two case reports and literature review. Pediatr Neurol 28: 151–155.
Barshop BA, Breuer J, Holm J, Leslie J, Nyhan WL (1989) Excretion of hippuric acid during sodium benzoate therapy in patients with hyperglycinaemia or hyperammonaemia. J Inherit Metab Dis 12: 72–79.
Batshaw MI, Brusilow S, Waber L, et al (1982) Treatment of inborn errors of urea synthesis: activation of alternative pathways of waste nitrogen synthesis and excretion. N Engl J Med 306: 1387–1392.
Bridges JW, French MR, Smith RL, Williams RT (1970) The fate of benzoic acid in various species. Biochem J 118: 47–51.
Cole DEC, Meek DC (1985) Juvenile non-ketotic hyerglycinaemia in three siblings. J Inherit Metab Dis 8(supplement 2): 123–124.
Deutsch SI, Rosse RB, Mastropaolo J (1998) Current status of NMDA antagonist interventions in the treatment of nonketotic hyperglycinemia. Clin Neuropharmacol 21: 71–79.
Dobyns WB (1989) Agenesis of the corpus callosum and gyral malformations are frequent manifestations of nonketotic hyperglycinemia. Neurology 39: 817–820.
Gregus Z, Fekete T, Varga F, Klaassen CD (1992) Availability of glycine and coenzyme A limits glycine conjugation in vivo. Drug Metab Dispos 20: 234–240.
Gregus Z, Fekete T, Varga F, Klaassen CD (1993) Dependence of glycine conjugation on availability of glycine: role of the glycine cleavage system. Xenobiotica 23: 141–153.
Gitzelmann R, Steinmann B (1982) NKH Workshop. Clincial and therapeutic aspects of non-ketotic hyperglycinemia. J Inherit Metab Dis 5(supplement 2): 113–116.
Hamosh A, Johnston MV (2001) Nonketotic hyperglycinemia. In: Scriver CR, Beaudet AL, Sly WS, Valle D, eds; Childs B, Kinzler KW, Vogelstein B, assoc. eds. The Metabolic and Molecular Bases of Inherited Disease, 8th edn. New York: McGraw-Hill, 2065–2078.
Hamosh A, Maher JF, Bellus GA, Rasmussen SA, Johnston MV (1998) Long-term use of high-dose benzoate and dextromethorphan for the treatment of nonketotic hyperglycinemia. J Pediatr 132: 709–713.
Heil SG, Van der Put NMJ, Waas ET, den Heijer M, Trijbels FJM, Blom H (2001) Is mutated serine hydroxymethyltransferase (SHMT) involved in the etiology of neural tube defects? Mol Genet Metab 73: 164–172.
Hennermann JP, Barufe JM, Monch E (2001) Clinical variation in nonketotic hyperglycinemia. J Inherit Metab Dis 24(supplement 1): 35.
Hoover-Fong JE, Shah S, Van Hove JLK, Applegarth D, Toone J, Hamosh A (2004) Natural history of non-ketotic hyperglycinemia in 65 patients. Neurology 63: 1847–1853.
Krieger I, Winbaum ES, Eisenbray AB (1977) Cerebrospinal fluid glycine in nonketotic hyperglycinemia. Effect of treatment with sodium benzoate and a ventricular shunt. Metabolism 26: 517–524.
Kure S, Ichinohe A, Kojima K, et al (2004) Mild variant of nonketotic hyperglycinemia with typical neonatal presentations: mutational and in vitro expression analyses in two patients. J Pediatr 144: 827–829.
MacArthur RB, Altincatal A, Tuchman M (2004) Pharmacokinetics of sodium phenylacetate and sodium benzoate following intravenous administration as both a bolus and continuous infusion to healthy adult volunteers. Mol Genet Metab 81: S67–S73.
Palmer T, Oberholzer VG (1985) Amino acid loading tests in a patient with non-ketotic hyperglycinaemia. J Inherit Metab Dis 8(supplement 2): 125–126.
Qureshi I, Rouleau T, Letarte J, Quellet R (1986) Significance of transported glycine in the conjugation of sodium benzoate in spf mutant mice with omithine transcarbamylase deficiency. Biochem Int 12: 839–846.
Qureshi I A, Clermont P, Letarte J (1989) The importance of glyoxylate and other glycine precursors in the hepatic and renal conjugation of benzoate in normal and hyperammonemic mice. Can J Physiol Pharmacol 67: 1426–1430.
Schoovaerts K (2001) De rol van voeding in de behandeling van niet-ketotische hyperglycinemia [The role of nutrition in the treatment of non-ketotic hyperglycinemia]. BSc thesis, KH Leuven.
Shinka T, Inoue Y, Kuhara T, Matsumoto M, Matsumoto I (1985) Benzoylalanine: detection and identification of an alanine conjugate with benzoic acid in hyperammonemic patients treated with sodium benzoate. Clin Chim Acta 151: 293–300.
Souci SW, Fachmann W, Kraut H (2000) Food Composition and Nutrition Tables. Stuttgart: Medpharm Scientific Publishers; London: CRC Press.
Steiman GS, Yudkoff M, Berman PH, Blazer-Yost B, Segal S (1979) Late-onset nonketotic hyperglycinemia and spinocerebellar degeneration. J Pediatr 94: 907–911.
Steiner RD, Sweetser DA, Rohrbaugh JR, Dowton SB, Toone JR, Applegarth DA (1996) Nonketotic hyperglycinemia: atypical clinical and biochemical manifestations. J Pediatr 128: 243–246.
Trauner DA, Page T, Greco C, Sweetman L, Kulovich S, Nyhan WL (1981) Progressive neurodegenerative disorder in a patient with nonketotic hyperglycinemia. J Pediatr 98: 272–275.
Trijbels JMF, Monnens LAH, van der Zee SPM, Vrenken JA, Sengers RCA, Schretlen EDA (1974) A patient with nonketotic hyperglycinemia: biochemical findings and therapeutic consequences. Pediatr Res 8: 598–605.
Van Hove JLK, Kishnani P, Muenzer J, et al (1995) Benzoate therapy and carnitine deficiency in non-ketotic hyperglycinemia. Am J Med Genet 59: 444–453.
Van Hove JLK, Kishnani PS, Demaerel P, et al (2000) Acute hydrocephalus in nonketotic hyperglycinemia. Neurology 54: 754–756.
Wolff JA, Kulovich S, Yu AL, Qiao C-N, Nyhan WL (1986) The effectiveness of benzoate in the management of seizures in nonketotic hyperglycinemia. Am J Dis Child 140: 596–602.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Van Hove, J.L.K., Kerckhove, K.V., Hennermann, J.B. et al. Benzoate treatment and the glycine index in nonketotic hyperglycinaemia. J Inherit Metab Dis 28, 651–663 (2005). https://doi.org/10.1007/s10545-005-0033-x
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/s10545-005-0033-x