Abstract
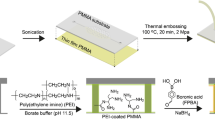
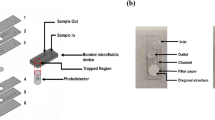
In this study, we integrated sample purification and genetic amplification in a seamless polycarbonate microdevice to facilitate foodborne pathogen detection. The sample purification process was realized based on the increased affinity of the boronic acid-modified surface toward the cis-diol group present on the bacterial outer membrane. The modification procedure was conducted at room temperature using disposable syringe. The visible color and fluorescence signals of alizarin red sodium were used to confirm the success of the surface modification process. Escherichia coli O157:H7 containing green fluorescence protein (GFP) and Staphylococcus aureus were chosen as the microbial models to demonstrate the nonspecific immobilization using the microdevice. Bacterial solutions of various concentrations were injected into the microdevice at three flow rates to optimize the operation conditions. This microdevice successfully amplified the 384-bp fragment of the eaeA gene of the captured E. coli O157:H7 within 1 h. Its detection limit for E. coli O157:H7 was determined to be 1 × 103 colony-forming units per milliliter (CFU mL−1). The proposed microdevice serves as a monolithic platform for facile and on-site identification of major foodborne pathogens.







Similar content being viewed by others
References
A. Adamczyk-Woźniak, R.M. Fratila, I.D. Madura, A. Pawełko, A. Sporzyński, M. Tumanowicz, A.H. Velders, J. Żyła, Tetrahedron Lett. 52, 6639–6642 (2011)
C.D. Ahrberg, A. Manz, B.G. Chung, Lab Chip 16, 3866–3884 (2016)
T. Artham, M. Doble, Macromol. Biosci. 8, 14–24 (2008)
S.A. Barghouthi, Indian J. Microbiol. 51, 430–444 (2011)
R.F. Borch, M.D. Bernstein, H.D. Durst, J. Am. Chem. Soc. 93, 2897–2904 (1971)
J.M. Butler, Advanced topics in forensic DNA typing: methodology chapter 4, 69–97 (2012)
N. Debreczeni, A. Fodor, Z. Hell, Catal. Lett. 144, 1547–1551 (2014)
L. Garibyan, N. Avashia, J. Invest. Dermatol. 133, 1–4 (2013)
T.R. Garrett, M. Bhakoo, Z. Zhang, Prog. Nat. Sci. 18, 1049–1056 (2008)
L.E. Govaert, T.A. Tervoort, J. Polym. Sci. B Polym. Phys. 42, 2041–2049 (2004)
N.S.K. Gunda, M. Singh, L. Norman, K. Kaur, S.K. Mitra, Appl. Surf. Sci. 305, 522–530 (2014)
M.L. Ha, Y. Zhang, N.Y. Lee, Biotechnol. Bioeng. 113, 2614–2623 (2016)
R. Jackeray, C.K.V.Z. Abid, G. Singh, S. Jain, S. Chattopadhyaya, S. Sapra, T.G. Shrivastav, H. Singh, Talanta 84, 952–962 (2011)
T. Kajisa, T. Sakata, Jpn. J. Appl. Phys. 56, 1–5 (2015)
J. Kaur, C. Jiang, G. Liu, Biosens. Bioelectron. 1123, 85–100 (2019)
K.V. Kong, Z. Lam, W.K.O. Lau, W.K. Leong, M. Olivo, J. Am. Chem. Soc. 135, 18028–18031 (2013)
M. Krishnan, V.M. Ugaz, M.A. Burns, Science 298, 793 (2002)
Y. Li, C. Zhang, D. Xing, Microfluid. Nanofluid. 10, 367–380 (2011)
Y.C. Liu, C.J. Chen, Mass Spectrometry 6, S0063 (2017)
J. Lyklema, W. Norde, M.C.M. van Loosdrecht, A.J.B. Zehnder, Colloids Surf. 39, 175–187 (1989)
W.M.J. Ma, M.M.P. Morais, F. D’Hooge, J.M.H. van den Elsen, J.P.L. Cox, T.D. James, J.S. Fossey, Chem. Commun. 13, 532–534 (2009)
T.P.O. Nguyen, B.M. Tran, N.Y. Lee, Lab Chip 16, 3251–3259 (2016)
D. Ogończyk, J. Węgrzyn, P. Jankowski, B. Dąbrowski, P. Garstecki, Lab Chip 10, 1324–1327 (2010)
S.Q.F. Parizzi, N.J. de Andrade, C.A.d.S. Silva, N.d.F.F. Soares, E.A.M. da Silva, Braz. Arch. Biol. Technol. 47, 77–83 (2004)
B. Pearson, P. Wang, A. Mills, S. Pang, L. McLandsboroug, L. He, Anal. Methods 9, 4732–4739 (2017)
X.H. Pham, S. Shim, T.H. Kim, E. Hahm, H.M. Kim, W.Y. Rho, D.H. Jeong, Y.S. Lee, B.H. Jun, BioChip J. 11, 46–56 (2017)
Q.N. Pham, K.T.L. Trinh, S.W. Jung, N.Y. Lee, Biotechnol. Bioeng. 115, 2194–2204 (2018)
B. Priyanka, R.K. Patil, S. Dwarakanath, Indian J. Med. Res. 144, 327–338 (2016)
D.S. Raghuvanshi, A.K. Gupta, K.N. Singh, Org. Lett. 14, 4326–4329 (2012)
C. Schrader, A. Schielke, L. Ellerbroek, R. Johne, L. App. Microbio. 113, 1014–1026 (2012)
B. Shu, C. Zhang, D. Xing, Lab Chip 15, 2597–2605 (2015)
B. Shu, C. Zhang, D. Xing, Biosens. Bioelectron. 97, 360–368 (2017)
C. Sousa, P. Teixeira, S. Bordeira, J. Fonseca, R. Oliveira, J. Adhes. Sci. Technol. 22, 675–686 (2008)
G. Springsteen, B. Wang, Tetrahedron 58, 5291–5300 (2002)
K.T.L. Trinh, H. Zhang, D.J. Kang, S.H. Kahng, B.D. Tall, N.Y. Lee, Int. Neurourol J. 20, 38–48 (2016)
K.T.L. Trinh, Y. Zhang, N.Y. Lee, Anal. Chim. Acta 1040, 63–73 (2018)
M. Varea, A. Clavel, O. Doiz, F.J. Castillo, M.C. Rubio, R. Gómez-Lus, Int. J. Parasitol. 28, 1881–1883 (1998)
J. Wang, J. Gao, D. Liu, D. Han, Z. Wang, Nanoscale 4, 451–454 (2012)
S. Wang, G.E. LeCroy, F. Yang, X. Dong, Y.P. Sun, L. Yang, RSC Adv. 5, 91246–91253 (2015a)
H. Wang, Y. Zhou, X. Jiang, B. Sun, Y. Zhu, H. Wang, Y. Su, Y. He, Angew. Chem. Int. Ed. 54, 5132–5136 (2015b)
G.F. Whyte, R. Vilar, R. Woscholski, Chem. Biol. 6, 161–174 (2013)
W. Zhai, X. Sun, T.D. James, J.S. Fossey, Chem. Asian J. 10, 1836–1848 (2015)
Acknowledgements
This work was supported by the National Research Foundation of Korea (NRF) grant funded by the Korea government (MSIP) (No. NRF-2017R1A2B4008179).
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
ESM 1
(DOCX 1.66 MB)
Rights and permissions
About this article
Cite this article
La, H.C., Lee, N.Y. Fabrication of a polycarbonate microdevice and boronic acid-mediated surface modification for on-chip sample purification and amplification of foodborne pathogens. Biomed Microdevices 21, 72 (2019). https://doi.org/10.1007/s10544-019-0420-y
Published:
DOI: https://doi.org/10.1007/s10544-019-0420-y