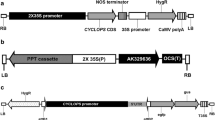
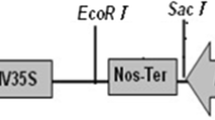
Abstract
Two selection systems for Agrobacterium tumefaciens mediated transformation of tomato and potato were compared. In the tomato (Lycopersicon esculentum cv. Moneymaker), the highest transformation rate, 4.2 %, of cotyledon explants on mannose-selection medium was obtained when mannose/sucrose concentration in the regeneration medium was 5/15 g dm−3. The best transformation efficacy with the commonly used concentration of 100 mg dm−3 kanamycin as a selection agent was 9 %. In the potato (Solanum tuberosum cv. Bintje), the highest transformation frequency was 53.3 % when mannose concentration in the regeneration medium was 5 g dm−3 during the first 3 weeks after transformation and 10 g dm−3 afterwards. The optimum concentration of sucrose was 20 g dm−3. The transformation efficiency using kanamycin as a selection agent at a concentration 100 mg dm−3 was 33.3 % with potato. Our results demonstrate that the transformation efficiency using mannose selection is 1.6-fold higher for potato and about 2 times lower for tomato comparing with the ordinary protocol using kanamycin.
Similar content being viewed by others
Abbreviations
- MS:
-
Murashige and Skoog (1962) medium
- NPT II:
-
neomycin phosphotransferase II
- PMI:
-
phosphomannose isomerase
- RM:
-
regeneration medium
- SE:
-
selection efficiency
- TE:
-
transformation efficiency
References
An, G., Watson, B.D., Chiang, C.C.: Transformation of tobacco, tomato, potato, and Arabidopsis thaliana using a binary Ti vector system.-Plant Physiol. 81: 301–305, 1986.
Aswath, C.R., Mo, S.Y., Kim, D.H., Park, S.W.: Agrobacterium and biolistic transformation of onion using non-antibiotic selection marker phosphomannose isomerase.-Plant Cell Rep. 25: 92–99, 2006.
Boscariol, R.L., Almeida, W.A.B., Derbyshire, M.T.V.C., Mourao, F.A.A., Mendes, B.M.J.: The use of the PMI/mannose selection system to recover transgenic sweet orange plants (Citrus sinensis L. Osbeck).-Plant Cell Rep. 22: 122–128, 2003.
Bříza, J., Pavingerová, D., Vlasák, J., Ludvíková, V., Niedermeierová, H.: Production of human papillomavirus type16 E7 oncoprotein fused with β-glucuronidase in transgenic tomato and potato.-Biol. Plant. 51: 268–276, 2007.
Carroll, B.J., Klimyuk, V.I., Thomas, C.M., Bishop, G.J., Harrison, K., Scofield, S.R., Jones, J.D.G.: Germinal transpositions of the maize element Dissociation from T-DNA loci in tomato.-Genetics 139: 407–420, 1995.
Church, G.M., Gilbert, W.: Genomic sequencing.-Proc. nat. Acad. Sci. USA 81: 1991–1995, 1984.
Ebmeier, A., Allison, L., Cerutti, H., Clemente, T.: Evaluation of the Escherichia coli threonine deaminase gene as a selectable marker for plant transformation.-Planta 218: 751–758, 2004.
Erikson, O., Hertzberg, M., Nasholm, T.: A conditional marker gene allowing both positive and negative selection in plants.-Nat. Biotechnol. 22: 455–458, 2004.
Erikson, O., Hertzberg, M., Nasholm, T.: The dsdA gene from Escherichia coli provides a novel selectable marker for plant transformation.-Plant mol. Biol. 57: 425–433, 2005.
Feeney, M., Punja, Z.K.: Tissue culture and Agrobacterium-mediated transformation of hemp (Cannabis sativa L.).-In Vitro cell. dev. Biol. Plant 39: 578–585, 2003.
Fillatti, J.J., Kiser, J., Rose, R., Comai, L.: Efficient transfer of glyphosate tolerance gene into tomato using a binary Agrobacterium vector.-Bio/Technology 5: 726–730, 1987.
Fraley, R.T., Rogers, S.G., Horsch, R.B., Sanders, P.R., Flick, J.S., Adams, S.P., Bittner, M.L., Brand, L.A., Fink, C.L., Fry, J.S., Galluppi, G.R., Goldberg, S.B., Hoffmann, N.L., Woo, S.C.: Expression of bacterial genes in plant cells.-Proc nat. Acad. Sci. USA 80: 4803–4807, 1983.
Fu, D.L., Xiao, Y.M., Muthukrishnan, S., Liang, G.H.: In vivo performance of a dual genetic marker, manA-gfp, in transgenic bentgrass.-Genome 48: 722–730, 2005.
Gadaleta, A., Giancaspro, A., Blechl, A., Blanco, A.: Phosphomannose isomerase, pmi, as a selectable marker gene for durum wheat transformation.-J. Cereal Sci. 43: 31–37, 2006.
Gao, Z.S., Xie, X.J., Ling, Y., Muthukrishnan, S., Liang, G.H.: Agrobacterium tumefaciens-mediated sorghum transformation using a mannose selection system.-Plant Biotechnol. J. 3: 591–599, 2005.
Haldrup, A., Petersen, S.G., Okkels, F.T.: The xylose isomerase gene from Thermoanaerobacterium thermosulfurogenes allows effective selection of transgenic plant cells using D-xylose as the selection agent.-Plant mol. Biol. 37: 287–296, 1998.
Hansen, G., Wright, M.S.: Recent advances in the transformation of plants.-Trends Plant Sci. 4: 226–231, 1999.
He, Z.Q., Duan, Z.Z., Liang, W., Chen, F.J., Yao, W., Liang, H.W., Yue, C.Y., Sun, Z.X., Chen, F., Dai, J.W.: Mannose selection system used for cucumber transformation.-Plant Cell Rep. 25: 953–958, 2006.
Holsters, M., De Waele, D., Depicker, A., Messens, E., Van Montagu, M., Schell, J.: Transfection and transformation of Agrobacterium tumefaciens.-Mol. gen. Genet. 163: 181–187, 1978.
Joersbo, M., Donaldson, I., Kreiberg, J., Petersen, S.G., Brunstedt, J., Okkels, F.T.: Analysis of mannose selection used for transformation of sugar beet.-Mol. Breeding 4: 111–117, 1998.
Joersbo, M., Okkels, F.T.: A novel principle for selection of transgenic plant cells: positive selection.-Plant Cell Rep. 16: 219–221, 1996.
Joersbo, M., Petersen, S.G., Okkels, F.T.: Parameters interacting with mannose selection employed for the production of transgenic sugar beet.-Physiol. Plant. 105: 109–115, 1999.
Klimyuk, V.I., Carroll, B.J., Thomas, C.M., Jones, J.D.G.: Alkali treatment for rapid preparation of plant tissue for reliable PCR analysis.-Plant J. 3: 493–494, 1993.
Ku, J.J., Park, Y.W., Park, Y.D.: A non-antibiotic selection system uses the phosphomannose-isomerase (PMI) gene for Agrobacterium-mediated transformation of Chinese cabbage.-J. Plant Biol. 49: 115–122, 2006.
Kunze, I., Ebneth, M., Heim, U., Geiger, M., Sonnewald, U., Herbers, K.: 2-Deoxyglucose resistance: a novel selection marker for plant transformation.-Mol. Breed. 7: 221–227, 2001.
Langley, R.A., Kado, C.I.: Conditions for mutagenesis by N-methyl-N′-nitro-N-nitrosoguanidine and relationships of A. tumefaciens mutants to crown-gall tumor induction.-Mutat. Res. 14: 277–286, 1972.
Lee, B.T., Matheson, N.K.: Phosphomannoisomerase and phosphoglucoisomerase in seeds of Cassia coluteoides and some other legumes that synthesize galactomannan.-Phytochemistry 23: 983–987, 1984.
Lucca, P., Ye, X.D., Potrykus, I.: Effective selection and regeneration of transgenic rice plants with mannose as selective agent.-Mol. Breed. 7: 43–49, 2001.
Miles, J.S., Guest, J.R.: Nucleotide sequence and transcriptional start point of the phosphomannose isomerase gene (manA) of Escherichia coli.-Gene 32: 41–48, 1984.
Murashige, T.F., Skoog, F.: A revised medium for rapid growth and bioassay with tobacco tissue cultures.-Physiol. Plant. 15: 473–497, 1962.
Negrotto, D., Jolley, M., Beer, S., Wenck, A.R., Hansen, G.: The use of phosphomannose isomerase as a selectable marker to recover transgenic plants (Zea mays L.) via Agrobacterium transformation.-Plant Cell Rep. 19: 798–80, 2000.
O’Kennedy, M.M., Burger, J.T., Botha, F.C.: Pearl millet transformation system using the positive selectable marker gene phosphomannose isomerase.-Plant Cell Rep. 22: 684–690, 2004.
Pavingerová, D., Bříza, J., Niedermeierová, H.: Timing of transposition of Ac mobile element in potato.-Biol. Plant. 44: 347–353, 2001.
Ramesh, S.A., Kaiser, B.N., Franks, T., Collins, G., Sedgley, M.: Improved methods in Agrobacterium-mediated transformation of almond using positive (mannose/pmi) or negative (kanamycin resistance) selection-based protocols.-Plant Cell Rep. 25: 821–828, 2006.
Reed, J., Privalle, L., Powell, M.L., Meghji, M., Dawson, J., Dunder, E., Suttie, J., Wenck, A., Launis, K., Kramer, C., Chang, Y.F., Hansen, G., Wright, M.: Phosphomannose isomerase: An efficient selectable marker for plant transformation.-In Vitro cell. dev. Biol. Plant 37: 127–132, 2001.
Sambrook, J., Fritsch, E.F., Maniatis, T.: Molecular Cloning: A Laboratory Manual. 2nd Ed.-Cold Spring Harbor Laboratory Press, Cold Spring Harbor 1989.
Sigareva, M., Spivey, R., Willits, M., Kramer, C., Chang, Y.F.: An efficient mannose selection protocol for tomato that has no adverse effect on the ploidy level of transgenic plants.-Plant Cell Rep. 23: 236–245, 2004.
Stein, J.C., Hansen, G.: Mannose induces an endonuclease responsible for DNA laddering in plant cells.-Plant Physiol. 121: 71–79, 1999.
Tai, T., Tanksley, S.: A rapid and inexpensive method for isolation of total DNA from dehydrated plant tissue.-Plant mol. Biol. Rep. 8: 297–303, 1991.
Todd, R., Tague, B.W.: Phosphomannose isomerase: A versatile selectable marker for Arabidopsis thaliana germ-line transformation.-Plant mol. Biol. Rep. 19: 07–319, 2001.
Wilmink, A., Dons, J.J.M.: Selective agents and marker genes for use in transformation of monocotyledonous plants.-Plant mol. Biol. Rep. 11: 165–185, 1993.
Wright, M., Dawson, J., Dunder, E., Suttie, J., Reed, J., Kramer, C., Chang, Y., Novitzky, R., Wang, H., Artim-Moore, L.: Eficient biolistic transformation of maize (Zea mays L.) and wheat (Triticum aestivum L.) using the phosphomannose isomerase gene, pmi, as the selectable marker.-Plant Cell Rep. 20: 429–436, 2001.
You, S.J., Liau, C.H., Huang, H.E., Feng, T.Y., Prasad, V., Hsiao, H.H., Lu, J.C., Chan, M.T.: Sweet pepper ferredoxin-like protein (pflp) gene as a novel selection marker for orchid transformation.-Planta 217: 60–65, 2003.
Zhang, P., Potrykus, I., Puonti-Caerlas, J.: Efficient production of transgenic cassava using negative and positive selection.-Transgenic Res. 9: 405–415, 2000.
Zhu, Y.J., Agbayani, R., McCafferty, H., Albert, H.H., Moore, P.H.:. Effective selection of transgenic papaya plants with the PMI/Man selection system.-Plant Cell Rep. 24: 426–432, 2005.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Bříza, J., Pavingerová, D., Přikrylová, P. et al. Use of phosphomannose isomerase-based selection system for Agrobacterium-mediated transformation of tomato and potato. Biol Plant 52, 453–461 (2008). https://doi.org/10.1007/s10535-008-0090-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10535-008-0090-8