Abstract
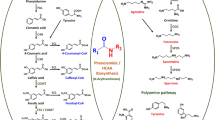
The enzymatic properties of four chalcone synthase homologues CHS_H1, VPS, CHS 2 and CHS 4 from Humulus lupulus L. were investigated after heterologous expression in Escherichia coli. It was found that both VPS and CHS_H1 can utilize isovaleryl-CoA and isobutyryl-CoA as substrates producing compounds with positions in thin layer chromatography characteristic for phloroisovalerophenone and phloroisobutyrophenone. These reactions are accompanied by the formation of associated byproducts. The formation of naringenin chalcone can be catalyzed primarily by CHS_H1. Comparatively the ability of VPS to perform chalcone synthase reaction is very limited. Since only CHS_H1 has true chalcone synthase activity, this enzyme can be considered a key enzyme in prenylflavonoid biosynthesis. Both CHS 2 and CHS 4 utilize isovaleryl-CoA and isobutyryl-CoA as substrates, but the reactions were prematurely terminated. In comparison with VPS and CHS_H1, the optimum pH of CHS 2 was shifted to lower value. High expression of chalcone synthase-like genes were found in maturating hop cones of cultivars with high bitter acid content (Agnus, Magnum, Target) by Northern and Western blotting using probes specific for vps, chs_H1, chs 4 and polyspecific serum risen against recombinant protein CHS4, respectively. It was also found that these cultivars maintained expression of CHS homologues for a longer period of time during cone development in contrast to time-limited expression of CHS homologues in cultivars with low bitter acids content.
Similar content being viewed by others
Abbreviations
- CHS:
-
chalcone synthase
- VPS:
-
valerophenone synthase
References
Ausubel, F.M., Brent, R., Kingston, R.E., Moore, D.D., Seidman, J.G., Smith, J.A., Struhl. K. (ed.): Current Protocols in Molecular Biology.-Publishing Associates and Wiley Interscience, New York 1987.
Ferrer, J., Jez, J.M., Bowman, M.E., Dixon, R.A., Noel, J.P.: Structure of chalcone synthase and the molecular basis of plant polyketide biosynthesis.-Natur. struct. Biol. 6: 775–784, 1999.
Gehrig, H.H., Winter, K., Cushman, J., Borland, A., Taybi, T.: An improved RNA isolation method for succulent plant species rich in polyphenols and polysaccharide.-Plant mol. Biol. Rep. 18: 369–376, 2000.
Harlow, E., Lane, D. (ed): Antibodies: a Laboratory Manual.-Cold Spring Harbor Laboratory, New York 1988.
Hernandez, A., Ruiz, M.T.: An EXCEL template for calculation of enzyme kinetic parameters by non-linear regression.-Bioinformatics 14: 227–228, 1998.
Hornsey, I. (ed.): Brewing.-Royal Society of Chemistry, London 2000.
Ito, M., Ichinose, Y., Kato, H., Shiraishi, T., Yamada, T.: Molecular evolution and functional relevance of the chalcone synthase genes of pea.-Mol. gen. Genet. 255: 28–37, 1997.
Jez, J.M., Austin, M.B., Ferrer, J., Bowman, M.E., Schroder, J., Noel, J.P.: Structural control of polyketide formation in plant-specific polyketide synthases.-Chem. Biol. 7: 919–930, 2000.
Jez, J.M., Bowman, M.E., Noel, J.P.: Structure-guided programming of polyketide chain-length determination in chalcone synthase.-Biochemistry 40: 14829–14839, 2001.
Lanz, T., Tropf, S., Marner, F.J., Schroder, J., Schroder, G.: The role of cysteines in polyketide synthases — site directed mutagenesis of resveratrol and chalcone synthases, 2 key enzymes in different plant-specific pathways.-J. biol. Chem. 266: 9971–9976, 1991.
Matousek, J., Novak, P., Briza, J., Patzak, J., Niedermeierova, H.: Cloning and characterization of chs-specific DNA and cDNA sequences from hop (Humulus lupulus L.).-Plant Sci. 162: 1007–1018, 2002a.
Matousek, J., Novak, P., Patzak, J., Briza, J., Krofta, K.: True chalcone synthase from Humulus lupulus L., biotechnology aspects of “medicinal hops”.-Rost. Vyroba (Praha) 48: 7–14, 2002b.
Miranda, C.L., Stevens, J.F., Helmrich, A., Henderson, M.C., Rodriguez, R.J., Yang, Y.H., Deinzer, M.L., Barnes, D.W., Buhler, D.R.: Antiproliferative and cytotoxic effects of prenylated flavonoids from hops (Humulus lupulus) in human cancer cell lines.-Food Chem. Toxicol. 37: 271–285, 1999.
Novak, P., Matousek, J., Briza, J.: Valerophenone synthase-like chalcone synthase homologues in hop (Humulus lupulus L.).-Biol. Plant. 46: 375–381, 2003.
Okada, Y., Ito K.: Cloning and analysis of valerophenone synthase gene expressed specifically in lupulin gland of hop (Humulus lupulus).-BioSci. Biotechnol. Biochem. 65: 150–155, 2001.
Paniego, N.B., Zuurbier, K.W.M., Fung, S.Y., van der Heijden, R., Scheffer, J.J.C., Verpoorte, R.: Phlorisovalerophenone synthase, a novel polyketide synthase from hop (Humulus lupulus L.) cones.-Eur. J. Biochem. 262: 612–616, 1999.
Raiber, S., Schroder, G., Schroder, J.: Molecular and enzymatic characterization of two stilbene synthases from Easter white pine (Pinus strobus): a single Arg/His difference determines the activity and the pH dependence of the enzymes.-FEBS Lett. 361: 299–302, 1995.
Stevens, J.F., Ivancic, M., Hsu, V.L., Deinzer, M.L.: Prenylflavonoids from Humulus lupulus.-Phytochemistry 44: 1575–1585, 1997.
Stevens, J.F., Miranda, C.L., Buhler, D.R., Deinzer, M.L.: Chemistry and biology of hop flavonoids.-J. amer. Soc. Brew. Chem. 65: 136–145, 1998.
Yamazaki, Y., Suh, D.-Y., Sitthithaworn, W., Ishiguro, K., Kobayashi, Y., Shibuya, M., Ebizuka, Y., Sankawa, U.: Diverse chalcone synthase superfamily enzymes from the most primitive vascular plant, Psilotum nudum.-Planta 214: 75–84, 2001.
Zuurbier, K.W.M., Fung, S., Sheffer, J.J.C., Verpoorte, R.: Formation of aromatic intermediate in the biosynthesis of bitter acids in Humulus lupulus.-Phytochemistry 38: 77–82, 1995.
Zuurbier, K.W.M., Lesser, J., Berger, T., Hofte, A.J.P., Schroder, G., Verpoorte, R., Schroder, J.: 4-hydroxy-2-pyrone formation by chalcone synthase and stylbene synthase with nonphysiological substrates.-Phytochemistry 49: 1945–1951, 1998.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Novak, P., Krofta, K. & Matousek, J. Chalcone synthase homologues from Humulus lupulus: some enzymatic properties and expression. Biol Plant 50, 48–54 (2006). https://doi.org/10.1007/s10535-005-0073-y
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/s10535-005-0073-y