Abstract
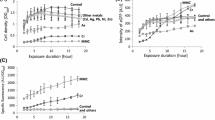
The significance of metal(oid)s as environmental pollutants has made them a priority in ecotoxicology, with the aim of minimizing exposure to animals or humans. Therefore, it is necessary to develop sensitive and inexpensive methods that can efficiently detect and monitor these pollutants in the environment. Conventional analytical techniques suffer from the disadvantages of high cost and complexity. Alternatively, prokaryotic or eukaryotic whole-cell biosensors (WCB) are one of the newest molecular tools employed in environmental monitoring that use the cell as an integrated reporter incorporating a reporter gene fused to a heavy metal responsive promoter. In the present paper, we report results from expressing, in the ciliate Tetrahymena thermophila, constructs consisting of the reporter gfp gene fused to the complete MTT1 or MTT5 protein coding regions under the transcriptional control of the MTT1 metallothionein promoter, which plays a critical role in heavy metal stress in this ciliate. When exposed to Cd2+, such cells overexpress both the GFP reporter transgene and the linked metallothionein gene. We report that, for the GFPMTT5 strain, this metallothionein overexpression results in marked resistance to cadmium toxicity (24 h LC50 ~15 μM of Cd2+), compared to wild type cells (24 h LC50 ~1.73 μM of Cd2+). These results provide the first experimental evidence that ciliate metallothioneins, like in other organisms, function to protect the cell against toxic metal ions. Because these strains may have novel advantages as WCBs, we have compared their properties to those of other previously reported Tetrahymena WCBs.





Similar content being viewed by others
Abbreviations
- GFP:
-
Green fluorescent protein
- IP:
-
Iodine propidium
- MT:
-
Metallothionein
- RT-PCR:
-
Retro-transcription-polymerase chain reaction
- TEM:
-
Transmission electron microscopy
- WCB:
-
Whole-cell biosensor
References
Amaro F, Turkewitz AP, Martin-Gonzalez A, Gutierrez JC (2011) Whole-cell biosensors for detection of heavy metal ions in environmental samples based on metallothionein promoters from Tetrahymena thermophila. Microb Biotechnol 4:513–522
Aury JM, Jaillon O, Duret L, Noel B, Jubin C, Porcel BM, Ségurens B, Daubin V, Anthouard V, Aiach N, Arnaiz O, Billaut A, Beisson J, Blanc I, Bouhouche K, Câmara F, Duharcourt S, Guigo R, Gogendeau D, Katinka M, Keller AM, Kissmehl R, Klotz C, Koll F, Le Mouël A, Lepère G, Malinsky S, Nowacki M, Nowak JK, Plattner H, Poulain J, Ruiz F, Serrano V, Zagulski M, Dessen P, Bétermier M, Weissenbach J, Scarpelli C, Schächter V, Sperling L, Meyer E, Cohen J, Wincker P (2006) Global trends of the whole-genome duplications revealed by the ciliate Paramecium tetraurelia. Nature 444:171–178
Belkin S (2003) Microbial whole-cell sensing systems of environmental pollutants. Curr Opin Microbiol 6:206–212
Boldrin F, Santovito G, Formigari A, Bisharyan Y, Cassidy-Hanley D, Clark TG, Piccinni E (2008) MTT2, a copper-inducible metallothionein gene from Tetrahymena thermophila. Comp Biochem Physiol C 147:232–240
Coppellotti O (1994) Effects of cadmium on Uronema marinum (Ciliophora, Scuticociliatida) from Antartica. Acta Protozool 33:159–167
Cowan AT, Bowman GR, Edwards KF, Emerson JJ, Turkewitz AP (2005) Genetic, genomic and functional analysis of the granule lattice proteins in Tetrahymena secretory granules. Mol Biol Cell 16:4046–4060
D′Souza SF (2001) Microbial biosensors. Biosens Bioelectron 49:337–353
Dentler W (2000) Fixation of Tetrahymena cells for electron microscopy. In: Asai DJ, Forney JD (eds) Tetrahymena thermophila: methods in cell biology, vol 62. Academic Press, London, pp 323–331
Díaz S, Amaro F, Rico D, Campos V, Benitez L, Martin-Gonzalez A, Hamilton EP, Orias E, Gutierrez JC (2007) Tetrahymena metallothioneins fall into two discrete subfamilies. PLoS ONE 2:e291
Dunlop S, Chapman G (1981) Detoxification of zinc and cadmium by the freshwater protozoan Tetrahymena pyriformis. II Growth experiments and ultrastructural studies on sequestration of heavy metals. Environ Research 24:264–274
Eisen JA, Coyne RS, Wu M, Wu D, Thiagarajan M, Wortman JR, Badger JH, Ren Q, Amedeo P, Jones KM, Tallon LJ, Delcher AL, Salzberg SL, Silva JC, Haas BJ, Majoros WH, Farzad M, Carlton JM, Smith RK Jr, Garg J, Pearlman RE, Karrer KM, Sun L, Manning G, Elde NC, Turkewitz AP, Asai DJ, Wilkes DE, Wang Y, Cai H, Collins K, Stewart BA, Lee SR, Wilamowska K, Weinberg Z, Ruzzo WL, Wloga D, Gaertig J, Frankel J, Tsao CC, Gorovsky MA, Keeling PJ, Waller RF, Patron NJ, Cherry JM, Stover NA, Krieger CJ, del Toro C, Ryder HF, Williamson SC, Barbeau RA, Hamilton EP, Orias E (2006) Macronuclear genome sequence of the ciliate Tetrahymena thermophila, a model eukaryote. PLoS Biol 4:c286
Gaertig J, Gorovsky MA (1992) Efficient mass transformation of Tetrahymena thermophila by electroporation of conjugants. Proc Natl Acad Sci USA 89:9196–9200
Gallego A, Martin-Gonzalez A, Ortega R, Gutierrez JC (2007) Flow cytometry assessment of cytotoxicity and reactive oxygen species generation by single and binary mixtures of cadmium, zinc and copper on populations of the ciliated protozoan Tetrahymena thermophila. Chemosphere 68:647–661
Gutierrez JC, Martin-Gonzalez A, Diaz S, Ortega R (2003) Ciliates as potential source of cellular and molecular biomarker/biosensors for heavy metal pollution. Eur J Protistol 39:461–467
Gutierrez JC, Martin-Gonzalez A, Diaz S, Amaro F, Ortega R, Gallego A, Lucas MP (2008) Ciliates as cellular tools to study the eukaryotic cell–heavy metal interactions. In: Brown SE, Welton WC (eds) Heavy metal pollution. Nova Science Publishers, New York, pp 1–44
Gutierrez JC, Amaro F, Martin-Gonzalez A (2009) From heavy metal-binder to biosensors: ciliate metallothioneins discussed. BioEssays 31:805–816
Gutierrez JC, Amaro F, Diaz S, de Francisco P, Cubas LL, Martin-Gonzalez A (2011) Ciliate metallothioneins: unique microbial eukaryotic heavy-metal-binder molecules. J Biol Inorg Chem 16:1025–1034
Hakkila K, Maksimow M, Karp M, Virta M (2002) Reporter genes lucFF, luxCDABE, gfp and dsred have different characteristics in whole-cell bacterial sensors. Anal Biochem 301:235–242
Hamilton EP, Orias E (2000) Genetically mapping new mutants and cloned genes. In: Asai DJ, Forney JD (eds) Tetrahymena thermophila: methods in cell biology, vol 62. Academic Press, London, pp 265–280
Keith LH, Telliard WA (1979) Priority pollutants: a perspective view. Environ Sci Technol 13:416–423
Kohlmeier S, Mancuso M, Tecon R, Harms H, van der Meer JR, Wells M (2007) Bioreporters: gfp versus lux revisited and single-cell response. Biosens Bioelectron 22:1578–1585
Larionov A, Krause A, Miller W (2005) A standard curve based method for relative real time PCR data processing. BMC Bioinform 6:62
Lovley DR (2000) Environmental microbe–metal interactions. ASM Press, Washington DC
Magrisso S, Erel Y, Belkin S (2008) Microbial reporters of metal bioavailability. Microb Biotechnol 1:320–330
Martin-Gonzalez A, Diaz S, Borniquel S, Gallego A, Gutierrez JC (2006) Cytotoxicity and bioaccumulation of heavy metals by ciliated protozoa isolated from urban wastewater treatment plants. Res Microb 157:108–118
Nassiri Y, Mansot JL, Wery J, Ginsburger-Vogel T, Amiard JC (1997) Ultrastructural and electron energy loss spectroscopy studies of sequestration mechanisms of Cd and Cu in the marine diatom Skeletonema costatum. Arch Environ Contam Toxicol 33:147–155
Park M, Tsai S-L, Chen W (2013) Microbial biosensors: engineered microorganisms as the sensing machinery. Sensors 13:5777–5795
Piccinni E, Albergoni V (1996) Cadmium detoxification in Protists. Comp Biochem Physiol C 113C:141–147
Pyne CK, Iftode F, Curgy J (1983) The effects of cadmium on the growth pattern and ultrastructure of the ciliate Tetrahymena thermophila. Biol Cel 48:121–132
Sambrook J, Russell DW (2001) Molecular cloning: a laboratory manual. Cold Spring Harbor Laboratory Press, New York
Shang Y, Song X, Bowen J, Corstanje R, Gao Y, Gaertig J, Gorovsky MA (2002) A robust inducible-repressible promoter greatly facilitates gene knockouts, conditional expression and overexpression of homologous and heterologous genes in Tetrahymena thermophila. Proc Natl Acad Sci USA 97:3734–3739
Tsien RY (1998) The green fluorescent protein. Annu Rev Biochem 67:509–544
Valko M, Morris H, Cronin MT (2005) Metals, toxicity and oxidative stress. Curr Med Chem 12:1161–1208
van der Meer JR, Belkin S (2010) Where microbiology meets microengineering: design and applications of reporter bacteria. Nat Rev Microb 8:511–522
van der Meer JR, Tropel D, Jasper M (2004) Illuminating the detection chain of bacterial bioreporters. Environ Microbiol 6:1005–1020
Visviki I, Rachlin JW (1994) Acute and chronic exposure of Dunaliella salina and Chlamydomonas bullosa to copper and cadmium: effects on growth. Arch Environ Contam Toxicol 26:149–153
Walmsley RM, Keenan P (2000) The eukaryote alternative: advantages of using yeasts in place of bacteria in microbial biosensor development. Biotechnol Bioprocess Eng 5:387–394
Yao M-C, Yao C-H (1991) Transformation of Tetrahymena to cycloheximide resistance with a ribosomal protein gene through sequence replacement. Proc Natl Acad Sci USA 88:9493–9497
Acknowledgments
This work was supported by the Grant CGL2008-00317/BOS (MINECO) to JCG and by NIH GM077607 to APT. A PhD scholarship from Complutense University (UCM) was awarded to FA.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Amaro, F., Turkewitz, A.P., Martín-González, A. et al. Functional GFP-metallothionein fusion protein from Tetrahymena thermophila: a potential whole-cell biosensor for monitoring heavy metal pollution and a cell model to study metallothionein overproduction effects. Biometals 27, 195–205 (2014). https://doi.org/10.1007/s10534-014-9704-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10534-014-9704-0