Abstract
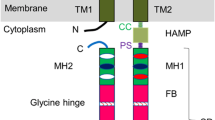
The mechanism of TonB dependent siderophore uptake through outer membrane transporters in Gram-negative bacteria is poorly understood. In an effort to expand our knowledge of the interaction between TonB and the outer membrane transporters, we have cloned and expressed the FepA cork domain (11–154) from Salmonella typhimurium and characterized its interaction with the periplasmic C-terminal domain of TonB (103–239) by isotope assisted FTIR and NMR spectroscopy. For comparison we also performed similar experiments using the FecA N-terminal domain (1–96) from Escherichia coli which includes the conserved TonB box. The FepA cork domain was completely unfolded in solution, as observed for the E. coli cork domain previously [Usher et al. (2001) Proc Natl Acad Sci USA 98, 10676–10681]. The FepA cork domain was found to bind to TonB, eliciting essentially the same chemical shift changes in TonB C-terminal domain as was observed in the presence of TonB box peptides. The FecA construct did not cause this same structural change in TonB. The binding of the FepA cork domain to TonB-CTD was found to decrease the amount of ordered secondary structure in TonB-CTD. It is likely that the FecA N-terminal domain interferes with TonB-CTD binding to the TonB box. Binding of the FepA cork domain induces a loss of secondary structure in TonB, possibly exposing TonB surface area for additional intermolecular interactions such as potential homodimerization or additional interactions with the barrel of the outer membrane transporter.
Similar content being viewed by others
References
A Aiyar (2000) ArticleTitleThe use of CLUSTAL W and CLUSTAL X for multiple sequence alignment Methods Mol Biol 132 221–241 Occurrence Handle10547838 Occurrence Handle1:CAS:528:DyaK1MXmslKqsLg%3D
A Barth (2000) ArticleTitleThe infrared absorption of amino acid side chains Prog Biophys Mol Biol 74 141–173 Occurrence Handle11226511 Occurrence Handle1:CAS:528:DC%2BD3MXhsVKmurg%3D Occurrence Handle10.1016/S0079-6107(00)00021-3
M Bonhivers M Desmadril GS Moeck (2001) ArticleTitle2001 Stability studies of FhuA, a two-domain outer membrane from Escherichia Coli. Biochemistry 40 2606–2613 Occurrence Handle11327884 Occurrence Handle1:CAS:528:DC%2BD3MXnsFKjtw%3D%3D Occurrence Handle10.1021/bi001725i
V Braun (2003) ArticleTitleIron uptake by Escherichia coli Front Biosci 8 s1409–s1421 Occurrence Handle12957834 Occurrence Handle1:CAS:528:DC%2BD3sXntlCqs7k%3D
V Braun S Mahren (2005) ArticleTitleTransmembrane transcriptional control (surface signalling) of the Escherichia coli Fec type FEMS Microbiol Rev 29 673–684 Occurrence Handle16102597 Occurrence Handle1:CAS:528:DC%2BD2MXos1Wkt7Y%3D Occurrence Handle10.1016/j.femsre.2004.10.001
SK Buchanan BS Smith L Venkatramani et al. (1999) ArticleTitleCrystal structure of the outer membrane active transporter FepA from Escherichia coli Nat Struct Biol 6 56–63 Occurrence Handle9886293 Occurrence Handle1:CAS:528:DyaK1MXjvVOjuw%3D%3D Occurrence Handle10.1038/4931
DM Byler H Susi (1986) ArticleTitleExamination of the secondary structure of proteins by deconvolved FTIR spectra Biopolymers 25 469–487 Occurrence Handle3697478 Occurrence Handle1:CAS:528:DyaL28Xht1GqtLc%3D Occurrence Handle10.1002/bip.360250307
C Chang A Mooser A Pluckthun A Wlodawer (2001) ArticleTitleCrystal structure of the dimeric C-terminal domain of TonB reveals a novel fold J Biol Chem 276 27535–27540 Occurrence Handle11328822 Occurrence Handle1:CAS:528:DC%2BD3MXlsV2isLg%3D Occurrence Handle10.1074/jbc.M102778200
DP Chimento RJ Kadner MC Wiener (2005) ArticleTitleComparative structural analysis of TonB-dependent outer membrane transporters: implications for the transport cycle Proteins 59 240–251 Occurrence Handle15739205 Occurrence Handle1:CAS:528:DC%2BD2MXksl2jsr8%3D Occurrence Handle10.1002/prot.20416
YN Chirgadze OV Fedorov NP Trushina (1975) ArticleTitleEstimation of amino acid residue side-chain absorption in the infrared spectra of protein solutions in heavy water Biopolymers 14 679–694 Occurrence Handle1156632 Occurrence Handle1:CAS:528:DyaE2MXksF2htLw%3D Occurrence Handle10.1002/bip.1975.360140402
D Cobessi H Celia N Folschweiller et al. (2005a) ArticleTitleThe crystal structure of the pyoverdine outer membrane receptor FpvA from Pseudomonas aeruginosa at 3.6 angstroms resolution J Mol Biol 347 121–134 Occurrence Handle1:CAS:528:DC%2BD2MXhslChtLY%3D Occurrence Handle10.1016/j.jmb.2005.01.021
D Cobessi H Celia F Pattus (2005b) ArticleTitleCrystal structure at high resolution of ferric-pyochelin and its membrane receptor FptA from Pseudomonas aeruginosa J Mol Biol 352 893–904 Occurrence Handle1:CAS:528:DC%2BD2MXhtVWit7nO Occurrence Handle10.1016/j.jmb.2005.08.004
F Delaglio S Grzesiek GW Vuister et al. (1995) ArticleTitleNMRPipe: a multidimensional spectral processing system based on UNIX pipes J Biomol NMR 6 277–293 Occurrence Handle8520220 Occurrence Handle1:CAS:528:DyaK2MXhtVSmurfK Occurrence Handle10.1007/BF00197809
HJ Dyson PE Wright (2005) ArticleTitleIntrinsically unstructured proteins and their functions Nat Rev Mol Cell Biol 6 197–208 Occurrence Handle15738986 Occurrence Handle1:CAS:528:DC%2BD2MXhslSlsLo%3D Occurrence Handle10.1038/nrm1589
HA Eisenhauer S Shames PD Pawelek JW Coulton (2005) ArticleTitleSiderophore transport through Escherichia coli outer membrane receptor FhuA with disulfide-tethered cork and barrel domains J Biol Chem 280 30574–30580 Occurrence Handle15994322 Occurrence Handle1:CAS:528:DC%2BD2MXos1Cqtrs%3D Occurrence Handle10.1074/jbc.M506708200
H Fabian HJ Vogel (2002) ArticleTitleFourier transform infrared spectroscopy of calcium-binding proteins Methods Mol Biol 173 57–74 Occurrence Handle11859779 Occurrence Handle1:CAS:528:DC%2BD38XhsVWns74%3D
H Fabian T Yuan HJ Vogel HH Mantsch (1996) ArticleTitleComparative analysis of the amino- and carboxy-terminal domains of calmodulin by Fourier transform infrared spectroscopy Eur Biophys J 24 195–201 Occurrence Handle8665835 Occurrence Handle1:CAS:528:DyaK28XjsVaqsrw%3D Occurrence Handle10.1007/BF00205100
JD Faraldo-Gomez GR Smith MS Sansom (2003) ArticleTitleMolecular dynamics simulations of the bacterial outer membrane protein FhuA: a comparative study of the ferrichrome-free and bound states Biophys J 85 1406–1420 Occurrence Handle12944258 Occurrence Handle1:CAS:528:DC%2BD3sXntFemt78%3D
AD Ferguson V Braun HP Fiedler et al. (2000) ArticleTitleCrystal structure of the antibiotic albomycin in complex with the outer membrane transporter FhuA Protein Sci 9 956–963 Occurrence Handle10850805 Occurrence Handle1:CAS:528:DC%2BD3cXjs12hsr0%3D Occurrence Handle10.1110/ps.9.5.956
AD Ferguson R Chakraborty BS Smith et al. (2002) ArticleTitleStructural basis of gating by the outer membrane transporter FecA Science 295 1715–1719 Occurrence Handle11872840 Occurrence Handle1:CAS:528:DC%2BD38Xhslehs7g%3D Occurrence Handle10.1126/science.1067313
AD Ferguson E Hofmann JW Coulton K Diederichs W Welte (1998) ArticleTitleSiderophore-mediated iron transport: crystal structure of FhuA with bound lipopolysaccharide Science 282 2215–2220 Occurrence Handle9856937 Occurrence Handle1:CAS:528:DyaK1cXotFCrtbk%3D Occurrence Handle10.1126/science.282.5397.2215
A Garcia Herrero HJ Vogel (2005) ArticleTitleNuclear magnetic resonance solution structure of the periplasmic signalling domain of the TonB-dependent outer membrane transporter FecA from Escherichia coli Mol Microbiol 58 1226–1237 Occurrence Handle16313612 Occurrence Handle1:CAS:528:DC%2BD2MXhtlWmsLzJ Occurrence Handle10.1111/j.1365-2958.2005.04889.x
E Goormaghtigh V Cabiaux JM Ruysschaert (1994) ArticleTitleDetermination of soluble and membrane protein structure by Fourier transform infrared spectroscopy. III. Secondary structures Subcell Biochem 23 405–450 Occurrence Handle7855879 Occurrence Handle1:CAS:528:DyaK2MXmvVWlu7s%3D
Guex N 2001 DeepView – The Swiss-PDBViewer User Guide.
PI Haris D Chapman (1995) ArticleTitleThe conformational analysis of peptides using Fourier transform IR spectroscopy Biopolymers 37 251–263 Occurrence Handle7540054 Occurrence Handle1:CAS:528:DyaK2MXlvFarurg%3D Occurrence Handle10.1002/bip.360370404
PI Haris GT Robillard AA Dijk ParticleVan D Chapman (1992) ArticleTitlePotential of 13C and 15N labeling for studying protein-protein interactions using Fourier transform infrared spectroscopy Biochemistry 31 6279–6284 Occurrence Handle1320934 Occurrence Handle1:CAS:528:DyaK38XksVersrk%3D Occurrence Handle10.1021/bi00142a016
M Hollosi Z Majer AZ Ronai et al. (1994) ArticleTitleCD and Fourier transform ir spectroscopic studies of peptides. II. Detection of beta-turns in linear peptides Biopolymers 34 177–185 Occurrence Handle8142587 Occurrence Handle1:CAS:528:DyaK2cXitFymsbk%3D Occurrence Handle10.1002/bip.360340204
M Jackson HH Mantsch (1995) ArticleTitleThe use and misuse of FTIR spectroscopy in the determination of protein structure Crit Rev Biochem Mol Biol 30 95–120 Occurrence Handle7656562 Occurrence Handle1:CAS:528:DyaK2MXlvFGnsrw%3D
BA Johnson RA Blevins (1994) ArticleTitleNMRView – A computer program for the visualization and analysis of NMR data J Biomol NMR 4 603–614 Occurrence Handle1:CAS:528:DyaK2cXmt1Gkurw%3D Occurrence Handle10.1007/BF00404272
N Kalnin S Venyaminov (1987) ArticleTitleQuantitative measurements of water solution IR spectra J Appl Spectrosc 4 592–597
CM Khursigara G Crescenzo ParticleDe PD Pawelek JW Coulton (2004) ArticleTitleEnhanced binding of TonB to a ligand-loaded outer membrane receptor: Role of the oligomeric state of TonB in formation of a functional FhuA-TonB complex J Biol Chem 279 7405–7412 Occurrence Handle14668326 Occurrence Handle1:CAS:528:DC%2BD2cXhsFaqu70%3D Occurrence Handle10.1074/jbc.M311784200
CM Khursigara G De Crescenzo PD Pawelek JW Coulton (2005) ArticleTitleDeletion of the proline-rich region of TonB disrupts formation of a 2:1 complex with FhuA, an outer membrane receptor of Escherichia coli Protein Sci 14 1266–1273 Occurrence Handle15802653 Occurrence Handle1:CAS:528:DC%2BD2MXjvFSqtrs%3D Occurrence Handle10.1110/ps.051342505
PE Klebba (2003) ArticleTitleThree paradoxes of ferric enterobactin uptake Front Biosci 8 s1422–s1436 Occurrence Handle12957833 Occurrence Handle1:CAS:528:DC%2BD3sXntlGqsro%3D
R Koebnik (2005) ArticleTitleTonB-dependent trans-envelope signalling: the exception or the rule? Trends Microbiol 13 343–347 Occurrence Handle15993072 Occurrence Handle1:CAS:528:DC%2BD2MXmvFSisL0%3D Occurrence Handle10.1016/j.tim.2005.06.005
J Koedding F Killig P Polzer et al. (2005) ArticleTitleCrystal structure of a 92-residue C-terminal fragment of TonB from Escherichia coli reveals significant conformational changes compared to structures of smaller TonB fragments J Biol Chem 280 3022–3028 Occurrence Handle1:CAS:528:DC%2BD2MXmt12isA%3D%3D Occurrence Handle10.1074/jbc.M411155200
G Kurisu SD Zakharov MV Zhalnina et al. (2003) ArticleTitleThe structure of BtuB with bound colicin E3 R-domain implies a translocon Nat Struct Biol 10 948–954 Occurrence Handle14528295 Occurrence Handle1:CAS:528:DC%2BD3sXosVCjtbg%3D Occurrence Handle10.1038/nsb997
S Letoffe K Wecker M Delepierre P Delepelaire C Wandersman (2005) ArticleTitleActivities of the Serratia marcescens heme receptor HasR and isolated plug and beta-barrel domains: the beta-barrel forms a heme-specific channel J Bacteriol 187 4637–4645 Occurrence Handle15968075 Occurrence Handle1:CAS:528:DC%2BD2MXlslGgsro%3D Occurrence Handle10.1128/JB.187.13.4637-4645.2005
HH Mantsch A Perczel M Hollosi GD Fasman (1993) ArticleTitleCharacterization of beta-turns in cyclic hexapeptides in solution by Fourier transform IR spectroscopy Biopolymers 33 201–207 Occurrence Handle8485294 Occurrence Handle1:CAS:528:DyaK3sXhs1Cms70%3D Occurrence Handle10.1002/bip.360330202
M Oke R Sarra R Ghirlando et al. (2004) ArticleTitleThe plug domain of a neisserial TonB-dependent transporter retains structural integrity in the absence of its transmembrane beta-barrel FEBS Lett 564 294–300 Occurrence Handle15111112 Occurrence Handle1:CAS:528:DC%2BD2cXjsVemsbw%3D Occurrence Handle10.1016/S0014-5793(04)00196-6
RS Peacock AM Weljie S.P. Howard FD Price HJ Vogel (2005) ArticleTitleThe solution structure of the C-terminal domain of TonB and interaction studies with TonB box peptides J Mol Biol 345 1185–1197 Occurrence Handle1:CAS:528:DC%2BD2MXis1ajtA%3D%3D Occurrence Handle10.1016/j.jmb.2004.11.026
K Postle RJ Kadner (2003) ArticleTitleTouch and go: tying TonB to transport Mol Microbiol 49 869–882 Occurrence Handle12890014 Occurrence Handle1:CAS:528:DC%2BD3sXmslKlt7Y%3D Occurrence Handle10.1046/j.1365-2958.2003.03629.x
C Ratledge LG Dover (2000) ArticleTitleIron metabolism in pathogenic bacteria Annu Rev Microbiol 54 881–941 Occurrence Handle11018148 Occurrence Handle1:CAS:528:DC%2BD3cXotFWrsbo%3D Occurrence Handle10.1146/annurev.micro.54.1.881
IJ Schalk WW Yue SK Buchanan (2004) ArticleTitleRecognition of iron-free siderophores by TonB-dependent iron transporters Mol Microbiol 54 14–22 Occurrence Handle15458401 Occurrence Handle1:CAS:528:DC%2BD2cXoslyqtb4%3D Occurrence Handle10.1111/j.1365-2958.2004.04241.x
WK Surewicz HH Mantsch (1988) ArticleTitleNew insight into protein secondary structure from resolution-enhanced infrared spectra Biochim Biophys Acta 952 115–130 Occurrence Handle3276352 Occurrence Handle1:CAS:528:DyaL1cXhtVyqtLY%3D
KC Usher E Ozkan KH Gardner J Deisenhofer (2001) ArticleTitleThe plug domain of FepA, a TonB-dependent transport protein from Escherichia coli, binds its siderophore in the absence of the transmembrane barrel domain Proc Natl Acad Sci USA 98 10676–10681 Occurrence Handle11526207 Occurrence Handle1:CAS:528:DC%2BD3MXntVGmsr8%3D Occurrence Handle10.1073/pnas.181353398
MC Wiener (2005) ArticleTitleTonB-dependent outer membrane transport: going for Baroque? Curr Opin Struct Biol 15 394–400 Occurrence Handle16039843 Occurrence Handle1:CAS:528:DC%2BD2MXmvVKhurg%3D Occurrence Handle10.1016/j.sbi.2005.07.001
WW Yue S Grizot SK Buchanan (2003) ArticleTitleStructural evidence for iron-free citrate and ferric citrate binding to the TonB-dependent outer membrane transporter FecA J Mol Biol 332 353–368 Occurrence Handle12948487 Occurrence Handle1:CAS:528:DC%2BD3sXms1OqtbY%3D Occurrence Handle10.1016/S0022-2836(03)00855-6
M Zhang H Fabian HH Mantsch HJ Vogel (1994) ArticleTitleIsotope-edited Fourier transform infrared spectroscopy studies of calmodulin’s interaction with its target peptides Biochemistry 33 10883–10888 Occurrence Handle7522050 Occurrence Handle1:CAS:528:DyaK2cXmvFGkt7k%3D Occurrence Handle10.1021/bi00202a006
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Peacock, R.S., Andrushchenko, V.V., Demcoe, A.R. et al. Characterization of TonB Interactions with the FepA Cork Domain and FecA N-terminal Signaling Domain. Biometals 19, 127–142 (2006). https://doi.org/10.1007/s10534-005-5420-0
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/s10534-005-5420-0