Abstract
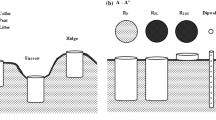
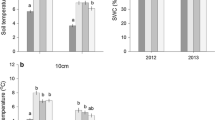
The two components of soil respiration, autotrophic respiration (from roots, mycorrhizal hyphae and associated microbes) and heterotrophic respiration (from decomposers), was separated in a root trenching experiment in a Norway spruce forest. In June 2003, cylinders (29.7 cm diameter) were inserted to 50 cm soil depth and respiration was measured both outside (control) and inside the trenched areas. The potential problems associated with the trenching treatment, increased decomposition of roots and ectomycorrhizal mycelia and changed soil moisture conditions, were handled by empirical modelling. The model was calibrated with respiration, moisture and temperature data of 2004 from the trenched plots as a training set. We estimate that over the first 5 months after the trenching, 45% of respiration from the trenched plots was an artefact of the treatment. Of this, 29% was a water difference effect and 16% resulted from root and mycelia decomposition. Autotrophic and heterotrophic respiration contributed to about 50% each of total soil respiration in the control plots averaged over the two growing seasons. We show that the potential problems with the trenching, decomposing roots and mycelia and soil moisture effects, can be handled by a modelling approach, which is an alternative to the sequential root harvesting technique.




Similar content being viewed by others
References
Betson NR, Göttlicher SG, Hall M, Wallin G, Richter A, Högberg P (2007) No diurnal variation in rate or carbon isotope composition of soil respiration in a boreal forest. Tree Physiol 27:749–756
Bhupinderpal-Singh, Nordgren A, Ottosson-Löfvenius M, Högberg MN, Mellander PE, Högberg P (2003) Tree root and soil heterotrophic respiration as revealed by girdling of boreal Scots pine forest: extending observations beyond the first year. Plant Cell Environ 26:1287–1296
Boone RD, Nadelhoffer KJ, Canary JD, Kaye JP (1998) Roots exert a strong influence on the temperature sensitivity of soil respiration. Nature 396:570–572
Borken W, Savage K, Davidson EA, Trumbore SE (2006) Effects of experimental drought on soil respiration and radiocarbon efflux from a temperate forest soil. Glob Change Biol 12:177–193
Boström B, Comstedt D, Ekblad A (2007) Isotope fractionation and 13C enrichment in soil profiles during the decomposition of soil organic matter. Oecologia 153:89–98
Bowden RD, Nadelhoffer KJ, Boone RD, Melillo JM, Garrison JB (1993) Contributions of above ground litter, below ground litter, and root respiration to total soil respiration in a temperate mixed hardwood forest. Can J For Res 23:1402–1407
Cheng W (2008) Rhizosphere priming effect: its functional relationships with microbial turnover, evapotranspiration, and C–N budgets. Soil Biol Biochem 41:1795–1801
Cheng W, Johnson DW, Fu S (2003) Rhizosphere effects on decomposition: controls of plant species, phenology and fertilization. Soil Sci Soc Am J 67:1418–1427
Comstedt D (2008) Explaining temporal variations in soil respiration rates and δ13C in coniferous forest ecosystems. Doctoral thesis, Örebro University (manuscript #3 in thesis)
Comstedt D, Boström B, Marshall JD, Holm A, Slaney M, Linder S, Ekblad A (2006) Effects of elevated [CO2] and temperature on soil respiration in a boreal forest using δ13C as a labelling tool. Ecosystems 9:1266–1277
Davidson EA, Belk E, Boone RD (1998) Soil water content and temperature as independent or confounded factors controlling soil respiration in a temperate mixed hardwood forest. Glob Change Biol 4:217–227
Dixon RK, Brown S, Houghton RA, Solomon AM, Trexler MC, Wisniewski J (1994) Carbon pools and flux of global forest ecosystems. Science 263:185–190
Ekblad A, Högberg P (2000) Analysis of 13C of CO2 distinguishes between microbial respiration of added C4-sucrose and other soil respiration in a C3-ecosystem. Plant Soil 219:197–209
Ekblad A, Högberg P (2001) Natural abundance of 13C in CO2 respired from forest soils reveals speed of link between tree photosynthesis and root respiration. Oecologia 127:305–308
Ekblad A, Boström B, Holm A, Comstedt D (2005) Forest soil respiration rate and δ13C is regulated by recent above ground weather conditions. Oecologia 143:136–142
Epron D, Farque L, Lucot E, Badot P-M (1999) Soil CO2 efflux in a beech forest: the contribution of root respiration. Ann For Sci 56:289–295
Epron D, le Dantec V, Dufrene E, Granier A (2001) Seasonal dynamics of soil carbon dioxide efflux and simulated rhizosphere respiration in a beech forest. Tree Physiol 21:145–152
Ewel KC, Cropper WP Jr, Gholz HL (1987) Soil CO2 evolution in Florida slash pine plantations. II. Importance of root respiration. Can J For Res 17:330–333
Gadgil RL, Gadgil PD (1971) Mycorrhiza and litter decomposition. Nature 233:133
Göttlicher SG, Steinmann K, Betson NR, Högberg P (2006) The dependence of soil microbial activity on recent photosynthate from trees. Plant Soil 287:85–94
Hanson PJ, Edwards NT, Garten CT, Andrews JA (2000) Separating root and soil microbial contributions to soil respiration: a review of methods and observations. Biogeochemistry 48:115–148
Heinemeyer A, Hartley IP, Evans SP, Carreira de la Fuente JA, Ineson P (2007) Forest soil CO2 flux: uncovering the contribution and environmental responses of ectomycorrhizas. Glob Change Biol 13:1786–1797
Högberg MN, Högberg P (2002) Extramatrical ectomycorrhizal mycelium contributes one-third of microbial biomass and produces, together with associated roots, half the dissolved organic carbon in a forest soil. New Phytol 154:791–795
Högberg P, Nordgren A, Buchmann N, Taylor AFS, Ekblad A, Högberg MN, Nyberg G, Ottosson-Löfvenius M, Read DJ (2001) Large scale forest girdling shows that current photosynthesis drives soil respiration. Nature 411:789–792
Högberg P, Nordgren A, Högberg MN, Ottosson-Löfvenius M, Bhupinderpal-Singh, Olsson P, Linder S (2005) Fractional contributions by autotrophic and heterotrophic respiration to soil-surface CO2 efflux in Boreal forests. In: Griffiths H, Jarvis PG (eds) The carbon balance of forest biomes. Taylor & Francis, Oxford, pp 251–267
James FC, McCulloch CE (1990) Multivariate analysis in ecology and systematics: Panacea or Pandora’s box? Annu Rev Ecol Syst 21:129–166
Kätterer T, Reichstein M, Andrén O, Lomander A (1998) Temperature dependence of organic matter decomposition: a critical review using literature data analyzed with different models. Biol Fertil Soils 27:258–262
Koide RT, Malcolm GM (2009) N concentration controls decomposition rates of different strains of ectomycorrhizal fungi. Fungal Ecol 2:197–202
Kuzyakov Y (2002) Review: factors affecting rhizosphere priming effects. J Plant Nutr Soil Sci 165:382–396
Kuzyakov Y (2006) Sources of CO2 efflux from soil and review of partitioning methods. Soil Biol Biochem 38:425–448
Kuzyakov Y, Gavrichkova O (2010) Time lag between photosynthesis and CO2 efflux from soil: a review. Glob Change Biol (in press)
Lavigne MB, Ryan MG, Anderson DE, Baldocchi DD, Crill PM, Fitzjarrald DR, Goulden ML, Gower ST, Massheder JMM, McCaughey JH, Rayment MB, Striegl RG (1997) Comparing nocturnal eddy covariance measurements to estimates of ecosystem respiration made by scaling chamber measurements at six coniferous boreal sites. J Geophys Res 102:28977–28985
Lee MS, Nakane K, Nakatsubo T, Koizumi H (2003) Seasonal changes in the contribution of root respiration to total soil respiration in a cool-temperate deciduous forest. Plant Soil 255:311–318
Liski J, Korotkov AV, Prins CFL, Karjalainen T, Victor DG, Kauppi PE (2003) Increased carbon sink in temperate and boreal forests. Clim Change 61:89–99
Majdi H, Damm E, Nylund J-E (2001) Longevity of mycorrhizal roots depends on branching order and nutrient availability. New Phytol 150:195–202
Mencuccini M, Hölttä T (2009) The significance of phloem transport for the speed with which canopy photosynthesis and belowground respiration are linked. New Phytol 185:189–203
Moorhead DL, Westerfield MM, Zak JC (1998) Plants retard litter decay in a nutrient-limited soil: a case study of exploitative competition? Oecologia 113:530–536
Ngao J, Longdoz B, Granier A, Epron D (2007) Estimation of autotrophic and heterotrophic components of soil respiration by trenching is sensitive to corrections for root decomposition and changes in soil water content. Plant Soil 301:99–110
Olsson P, Linder S, Giesler R, Högberg P (2005) Fertilization of boreal forest reduces both autotrophic and heterotrophic soil respiration. Glob Change Biol 11:1–9
Pritchard SG, Strand AE, McCormack ML, Davis MA, Oren R (2008) Mycorrhizal and rhizomorph dynamics in a loblolly pine forest during 5 years of free-air-CO2-enrichment. Glob Change Biol 14:1–13
Rey A, Pegoraro E, Tedeschi V (2002) Annual variation in soil respiration and its components in a coppice oak forest in Central Italy. Glob Change Biol 8:851–866
Rustad LE, Campbell JL, Marion GM, Norby RJ, Mitchell MJ, Hartley AE, Cornelissen JHC, Gurevitch J, GCTE-NEWS (2001) A meta-analysis of the response of soil respiration, net nitrogen mineralization, and aboveground plant growth to experimental ecosystem warming. Oecologia 126:543–562
Silver WL, Miya RK (2001) Global patterns in root decomposition: comparisons of climate and litter quality effects. Oecologia 129:407–419
Six J, Frey SD, Thiet RK, Batten KM (2006) Bacterial and fungal contributions to C-sequestration in agroecosystems. Soil Sci Soc Am J 70:555–569
Subke JA, Hahn V, Battipaglia G, Linder S, Buchmann N, Cotrufo MF (2004) Feedback interactions between needle litter decomposition and rhizosphere activity. Oecologia 139:551–559
Tang J, Baldocchi DD, Xu L (2005) Tree photosynthesis modulates soil respiration on a diurnal time scale. Glob Change Biol 11:1–7
Treseder KK, Allen MF, Ruess RW, Pregitzer KS, Hendrick RL (2005) Lifespans of fungal rhizomorphs under nitrogen fertilization in a pinyon-juniper woodland. Plant Soil 270:249–255
Unestam T (1991) Water repellency, mat formation, and leaf-stimulated growth of some ectomycorrhizal fungi. Mycorrhiza 1:13–20
Wallander H, Nilsson LO, Hagerberg D, Bååth E (2001) Estimation of the biomass and seasonal growth of external mycelium of ectomycorrhizal fungi in the field. New Phytol 151:753–760
Acknowledgements
This study was financially supported by the Swedish National Energy Administration (STEM), the Swedish Environmental Protection Agency (SNV), the Swedish Research council (VR), the Swedish Research council for Environment, Agricultural Sciences and Spatial Planning (Formas) and by the Swedish Research Council for Forestry and Agriculture. We thank Anders Holm for practical assistance.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Comstedt, D., Boström, B. & Ekblad, A. Autotrophic and heterotrophic soil respiration in a Norway spruce forest: estimating the root decomposition and soil moisture effects in a trenching experiment. Biogeochemistry 104, 121–132 (2011). https://doi.org/10.1007/s10533-010-9491-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10533-010-9491-9