Abstract
Objectives
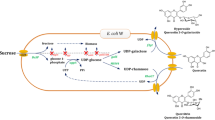
To produce flavonol and flavone 6-C-glucosides by bioconversion using recombinant Escherichia coli expressing a C-glucosyltransferase from wasabi (WjGT1).
Results
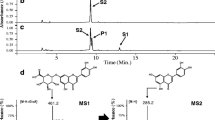
Escherichia coli expressing WjGT1 (Ec-WjGT1) converted flavones (apigenin and luteolin) and flavonols (quercetin and kaempferol) into their 6-C-glucosides in M9 minimal media supplemented with glucose, and released these products into the culture media. Ec-WjGT1 system also converts a flavanone (naringenin) into its C-glucoside at a conversion rate of 60% in 6 h. For scale-up production, apigenin, kaempferol, and quercetin were sequentially fed into the Ec-WjGT1 system at concentrations of 20–50 µM every 15–60 min, and the system was then able to produce isovitexin, kaempferol 6-C-glucoside, and quercetin 6-C-glucoside at an 89–99% conversion rate.
Conclusions
The Ec-WjGT1 system quickly and easily produces flavone and flavonol 6-C-glucosides at high conversion rates when using sequential administration to avoid precipitation of substrates.




Similar content being viewed by others
References
Brazier-Hicks M, Edwards R (2013) Metabolic engineering of the flavone-C-glycoside pathway using polyprotein technology. Metab Eng 16:11–20. https://doi.org/10.1016/j.ymben.2012.11.004
De Bruyn F, Van Brempt M, Maertens J, Van Bellegem W, Duchi D et al (2015) Metabolic engineering of Escherichia coli into a versatile glycosylation platform: production of bio-active quercetin glycosides. Microb Cell Fact 14:138. https://doi.org/10.1186/s12934-015-0326-1
Hamidullah KR, Saini KS, Kumar A, Kumar S et al (2015) Quercetin-6-C-β-d-glucopyranoside, natural analog of quercetin exhibits anti-prostate cancer activity by inhibiting Akt-mTOR pathway via aryl hydrocarbon receptor. Biochimie 119:68–79. https://doi.org/10.1016/j.biochi.2015.10.012
Harborne JB, Williams CA (2000) Advances in flavonoid research since 1992. Phytochemistry 55:481–504. https://doi.org/10.1016/s0031-9422(00)00235-1
He JB, Zhao P, Hu ZM, Liu S, Kuang Y et al (2019) Molecular and structural characterization of a promiscuous C-glycosyltransferase from Trollius chinensis. Angew Chem Int Ed 58:11513–11520. https://doi.org/10.1002/anie.201905505
Ito T, Fujimoto S, Shimosaka M, Taguchi G (2014) Production of C-glucosides of flavonoids and related compounds by Escherichia coli expressing buckwheat C-glucosyltransferase. Plant Biotechnol 31:519–524. https://doi.org/10.5511/plantbiotechnology.14.1016a
Ito T, Fujimoto S, Suito F, Shimosaka M, Taguchi G (2017) C-glycosyltransferases catalyzing the formation of di-C-glucosyl flavonoids in citrus plants. Plant J 91:187–198. https://doi.org/10.1111/tpj.13555
Jay M, Viricel MR, Gonnet JF (2006) C-glycosylflavonoids. In: Andersen ØM, Markham KR (eds) Flavonoids: chemistry, biochemistry and application. CRC Press, Boca Raton, FL, pp 858–903
Maatooq GT, El-Sharkawy SH, Afifi M, Rosazza JP (1997) C-p-hydroxybenzoylglycoflavones from Citrullus colocynthis. Phytochemistry 44:187–190. https://doi.org/10.1016/S0031-9422(96)00465-7
Markham K, Ternai B, Stanley R, Geiger H, Mabry T (1978) Carbon-13 NMR studies of flavonoids—III: naturally occurring flavonoid glycosides and their acylated derivatives. Tetrahedron 34:1389–1397. https://doi.org/10.1016/0040-4020(78)88336-7
Mashima K, Hatano M, Suzuki H, Shimosaka M, Taguchi G (2019) Identification and characterization of apigenin 6-C-glucosyltransferase involved in biosynthesis of isosaponarin in wasabi (Eutrema japonicum). Plant Cell Physiol 60:2733–2743. https://doi.org/10.1093/pcp/pcz164
Pei J, Sun Q, Zhao L, Shi H, Tang F et al (2018) Efficient biotransformation of luteolin to isoorientin through adjusting induction strategy, controlling acetic acid, and increasing UDP-glucose supply in Escherichia coli. J Agric Food Chem 67:331–340. https://doi.org/10.1021/acs.jafc.8b05958
Sasaki N, Nishizaki Y, Yamada E, Tatsuzawa F, Nakatsuka T et al (2015) Identification of the glucosyltransferase that mediates direct flavone C-glucosylation in Gentiana triflora. FEBS Lett 589:182–187. https://doi.org/10.1016/j.febslet.2014.11.045
Shrestha A, Pandey RP, Dhakal D, Parajuli P, Sohng JK (2018) Biosynthesis of flavone C-glucosides in engineered Escherichia coli. Appl Microbiol Biotechnol 102:1251–1267. https://doi.org/10.1007/s00253-017-8694-6
Sun Y, Chen Z, Yang J, Mutanda I, Li S et al (2020) Pathway-specific enzymes from bamboo and crop leaves biosynthesize anti-nociceptive C-glycosylated flavones. Commun Biol 3:1–11. https://doi.org/10.1038/s42003-020-0834-3
Talhi O, Silva A (2012) Advances in C-glycosylflavonoid research. Curr Org Chem 16:859–896. https://doi.org/10.2174/138527212800194791
Vanegas KG, Larsen AB, Eichenberger M, Fischer D, Mortensen UH et al (2018) Indirect and direct routes to C-glycosylated flavones in Saccharomyces cerevisiae. Microb Cell Fact 17:107. https://doi.org/10.1186/s12934-018-0952-5
Williams CA (2006) Flavone and flavonol O-glycosides. In: Andersen ØM, Markham KR (eds) Flavonoids: chemistry, biochemistry and application. CRC Press, Boca Raton, FL, pp 749–824
Xiao J, Muzashvili TS, Georgiev MI (2014) Advances in the biotechnological glycosylation of valuable flavonoids. Biotechnol Adv 32:1145–1156. https://doi.org/10.1016/j.biotechadv.2014.04.006
Zhang M, Li F-D, Li K, Wang Z-L, Wang Y-X et al (2020) Functional characterization and structural basis of an efficient di-C-glycosyltransferase from Glycyrrhiza glabra. J Am Chem Soc 142:3506–3512. https://doi.org/10.1021/jacs.9b12211
Acknowledgements
We are indebted to the Division of Gene Research, Research Center for Advanced Science and Technology, Shinshu University, and the Research Center for Advanced Plant Factory, Shinshu University, for supplying the necessary research facilities. We also thank the Global leader program for fiber renaissance, Shinshu University for essential support to NJ, and Dr. Makoto Shimosaka, Shinshu University for a critical evaluation of the manuscript.
Supplementary Information
Supplementary Table 1—NMR assignment of the purified compounds produced by bioconversion (400 MHz, dimethyl sulfoxide-d6 (DMSO-d6)).
Supplementary Figure 1—Bioconversion of flavones by the Ec-WjGT1 system.
Supplementary Figure 2—Effect of glucose concentration on the bioconversion of apigenin by the Ec-WjGT1 system.
Supplementary Figure 3—Heteronuclear multiple bond correlation (HMBC) analysis of the purified compounds produced by bioconversion.
Supplementary Figure 4—Bioconversion of resveratrol by the Ec-WjGT1 system.
Funding
This work was supported in part by Japan Society for the Promotion of Science (JSPS) KAKENHI Grant Numbers JP26450120 and JP20K05825 to GT.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors have no conflicts of interest to declare.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Dorjjugder, N., Hatano, M. & Taguchi, G. Production of flavonol and flavone 6-C-glucosides by bioconversion in Escherichia coli expressing a C-glucosyltransferase from wasabi (Eutrema japonicum). Biotechnol Lett 43, 1913–1919 (2021). https://doi.org/10.1007/s10529-021-03165-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10529-021-03165-3