Abstract
Objectives
To realize a practical technology for recycling both cyclodextrin and resting-cells at the same time in phytosterol biotransformation using mycobacterial resting cells.
Results
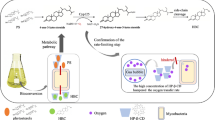
In order to produce 22-hydroxy-23,24-bisnorchol-4-ene-3-one (HBC) efficiently and low-costly, a recycled phytosterols (PS) biotransformation process using mycobacterial resting cells was developed. By optimizing the ratio of hydroxypropyl-β-cyclodextrin (HP-β-CD) and PS to 1:1 (w/w), most products crystallized during the biotransformation process. So, the HBC was easily separated by low-speed (900×g) centrifugation with yield of 92%. The resting cells, HP-β-CD and the residual products and substrates left in the reaction system were reused for another bioconversion cycle after PS supplement. Three continuous cycles were achieved without the supplement of cells and HP-β-CD. In each batch, 80 g L−1 of PS was transformed to HBC with the space–time yield of HBC of 8.9–12.8 g L−1 day−1.
Conclusions
This strategy reduced the cost of HBC production and simplified the purification process.


Similar content being viewed by others
References
Donova MV, Egorova OV (2012) Microbial steroid transformations: current state and prospects. Appl Microbiol Biotechnol 94:1423–1447. https://doi.org/10.1007/s00253-012-4078-0
Donova MV, Dovbnya DV, Sukhodolskaya GV, Khomutov SM, Nikolayeva VM, Kwon I, Han K (2005) Microbial conversion of sterol-containing soybean oil production waste. J Chem Technol Biotechnol 80(1):55–60. https://doi.org/10.1002/jctb.1156
Fernandes P, Cruz A, Angelova B, Pinheiro HM, Cabral JMS (2003) Microbial conversion of steroid compounds: recent developments. Enzym Microb Technol 32:688–705. https://doi.org/10.1016/s0141-0229(03)00029-2
Gao XQ, Feng JX, Hua Q, Wei DZ, Wang XD (2014) Investigation of factors affecting biotransformation of phytosterols to 9-hydroxyandrost-4-ene-3,17-dione based on the HP-β-CD-resting cells reaction system. Biocatal Biotransform 32:343–347. https://doi.org/10.3109/10242422.2014.976633
Gao XQ, Feng JX, Wang XD, Hua Q, Wei DZ (2015) Enhanced steroid metabolites production by resting cell phytosterol bioconversion. Chem Biochem Eng Quart 29:567–573. https://doi.org/10.15255/CABEQ.2014.2098
Heipieper HJ, Neumann G, Cornelissen S, Meinhardt F (2007) Solvent-tolerant bacteria for biotransformations in two-phase fermentation systems. Appl Microbiol Biotechnol 74:961–973. https://doi.org/10.1007/s00253-006-0833-4
Hesselink P, Vliet S, Vries H, Witholt B (1989) Optimization of steroid side chain cleavage by Mycobacterium sp. in the presence of cyclodextrins. Enzym Microb Technol 11:398–404. https://doi.org/10.1016/0141-0229(89)90133-6
Jambhekar S, Breen P (2016) Cyclodextrins in pharmaceutical formulations II: solubilization, binding constant, and complexation efficiency. Drug Discov Today 21:363–368. https://doi.org/10.1016/j.drudis.2015.11.016
Manosroi A, Saowakhon S, Manosroi J (2008) Enhancement of androstadienedione production from progesterone by biotransformation using the hydroxypropyl-beta-cyclodextrin complexation technique. J Steroid Biochem Mol Biol 108:132–136. https://doi.org/10.1016/j.jsbmb.2007.05.032
Mancilla RA, Little C, Amoroso A (2018) Efficient bioconversion of high concentration phytosterol microdispersion to 4-androstene-3,17-dione (AD) by Mycobacterium sp. B3805. Appl Biochem Biotechnol 185:494–506. https://doi.org/10.1007/s12010-017-2665-3
Stefanov S, Yankov D, Beschkov V (2006) Biotransformation of phytosterols to androstenedione in two phase water-oil systems. Chem Biochem Eng Quart 20:421–427
Shtratnikova VY, Schelkunov MI, Dovbnya DV, Bragin EY, Donova MV (2017) Effect of methyl-β-cyclodextrin on gene expression in microbial conversion of phytosterol. Appl Microbiol Biotechnol 101:4659–4667. https://doi.org/10.1007/s00253-017-8288-3
Shen YB, Yu ZQ, Yang X, Wang F, Luo JM, Wang M (2017) A new technique for promoting cyclic utilization of cyclodextrins in biotransformation. J Ind Microbiol Biotechnol 44:1–7. https://doi.org/10.1007/s10295-016-1856-1
Saab HB, Fouchard S, Boulanger A, Llopiz P, Neunlist S (2013) Luffa cylindrica and phytosterols bioconversion: from shake flask to jar bioreactor. J Ind Microbiol Biotechnol 40:315–1320. https://doi.org/10.1007/s10295-013-1315-1
Toró A, Ambrus G (1990) Oxidative decarboxylation of 17(20)-dehydro-23,24-dinorcholanoic acids. Tetrahedron Lett 31:3475–3476. https://doi.org/10.1016/S0040-4039(00)97426-4
Wang M, Zhang L, Shen Y, Ma Y, Zheng Y, Luo J (2009) Effects of hydroxypropyl-β-cyclodextrin on steroids 1-en-dehydrogenation biotransformation by Arthrobacter simplex TCCC 11037. J Mol Catal B 59:58–63. https://doi.org/10.1016/j.molcatb.2008.12.017
Xu LQ, Liu YJ, Yao K, Liu HH, Tao XY, Wang FQ, Wei DZ (2016) Unraveling and engineering the production of 23,24-bisnorcholenic steroids in sterol metabolism. Sci Rep 6:21928. https://doi.org/10.1038/srep21928
Zhou X, Zhang Y, Shen YB, Zhang X, Xu SP, Shang ZH, Xia ML, Wang M (2019) Efficient production of androstenedione by repeated batch fermentation in waste cooking oil media through regulating NAD+/NADH ratio and strengthening cell vitality of Mycobacterium neoaurum. Bioresour Technol 279:209–217. https://doi.org/10.1016/j.biortech.2019.01.144
Acknowledgements
This work was supported by the National Natural Science Foundation of China (Grant Numbers 31570079, 21276083).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Hu, Y., Wang, D., Wang, X. et al. A recycled batch biotransformation strategy for 22-hydroxy-23,24-bisnorchol-4-ene-3-one production from high concentration of phytosterols by mycobacterial resting cells. Biotechnol Lett 42, 2589–2594 (2020). https://doi.org/10.1007/s10529-020-02991-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10529-020-02991-1