Abstract
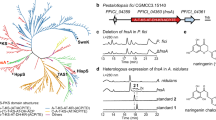
Biosynthesis of flavonoid derivatives requires enzyme(s) having high reactivity as well as regioselectivity. We have synthesized 3-O-kaempferol from naringenin using two enzymes. The first reaction, in which naringenin is converted to kaempferol, is mediated by flavonol synthase (FLS). An FLS (PFLS) with strong catalytic activity was cloned and characterized from the genome sequence of the poplar (Populus deltoides). PFLS consists of a 1,008 bp ORF encoding a 38 kDa protein. PFLS was expressed in Escherichia coli with a glutathione-S-transferase (GST) tagging. The purified recombinant PFLS was characterized. Catalytically, it was more efficient than the previously characterized FLSs. A mixture of two E. coli transformants harboring either PFLS or ROMT9 (a kaempferol 3-O-methyltransferase) converted naringenin into 3-O-methylkaempferol.



Similar content being viewed by others
References
Chua CS, Biermann D, Goo KS, Sim T-S (2008) Elucidation of active site residues of Arabidopsis thaliana flavonol synthase provides a molecular platform for engineering flavonols. Phytochemistry 69:66–75
Chung ST, Jang DS, Han A-R, Jang JO, Kwon Y, Seo E-K, Lee HJ (2007) Modulation of P-glycoprotein-mediated resistance by kaempferol derivatives isolated from Zingiber zerumbet. Phytother Res 21:565–569
Clifton IJ, McDonough MA, Ehrismann D, Kershqw NJ, Granatino N, Schofield CJ (2006) Structural studies on 2-oxoglutarate oxygenases and related double-stranded β-helix fold proteins. J Inorg Biochem 100:644–669
Cushine TPT, Hamilton VES, Lamb AJ (2003) Assessment of the antibacterial activity of selected flavonoids and consideration of discrepancies between previous reports. Microbiol Res 158:281–289
Harborne JB, Williams CA (2000) Advances in flavonoid research since 1992. Phytochemistry 55:481–504
Holton TA, Brugliera F, Tanaka Y (1993) Cloning and expression of flavonol synthase from Petunia hybrida. Plant J 4:1003–1010
Jeon YM, Kim BG, Ahn J-H (2009) Biological synthesis of 7-O-methyl apigenin from naringenin using Escherchia coli expressing two genes. J Microbiol Biotechnol 19:491–494
Katsuyama Y, Funa N, Miyahisa I, Horinouchi S (2007) Synthesis of unnatural flavonoids and stilbenes by exploiting the plant biosynthetic pathway in Escherichia coli. Chem Biol 14:613–621
Kim DH, Kim B-G, Lee Y, Ryu JY, Lim Y, Hur H-G, Ahn J-H (2005) Regiospecific methylation of naringenin to ponciretin by soybean O-methyltransferase expressed in Escherichia coli. J Biotechnol 138:155–162
Kim BG, Kim H, Hur H-G, Lim Y, Ahn J-H (2006a) Regioselectivity of 7-O-methyltransferase of poplar to flavones. J Biotechnol 138:52–162
Kim BG, Lee Y, Hur H-G, Lim Y, Ahn J-H (2006b) Flavonoid 3′-O-methyltransferase from rice: cDNA cloning, characterization and functional expression. Phytochemistry 67:387–394
Kim BG, Kim JH, Kim J, Lee C, Ahn J-H (2008) Accumulation of flavonols in response to ultraviolet-B irradiation in soybean is related to induction of flavanone 3-β-hydroxylase and flavonol synthase. Mol Cells 25:247–252
Kim S-K, Kim DH, Kim BG, Jeon YM, Hong BS, Ahn J-H (2009) Cloning and characterization of the UDP glucose/galactose epimerases of Oryza sativa. J Kor Soc Appl Biol Chem 52:315–320
Ko JH, Kim BG, Ahn J-H (2006) Glycosylation of flavonoids with a glycosyltransferase from Bacillus cereus. FEMS Microbiol Lett 258:263–268
Leonard E, Yan Y, Koffas MAG (2006) Functional expression of a P450 flavonoid hydroxylase for the biosynthesis of plant-specific hydroxylated flavonols in Escherichia coli. Metab Eng 8:172–181
Lim G-Z, Lian Y-J, Ryu J-H, Sung M-K, Park J-S, Park H-J, Park B-K, Shin J-S, Lee M-S, Cheon C-I (2007) Expression and purification of His-tagged flavonol synthase of Camellia sinensis from Escherichia coli. Protein Expr Purif 52:287–292
Lukacin R, Wellmann F, Britsch L, Martens S, Matern U (2003) Flavonol synthase gene from Citrus unshiu is a bifunctional dioxygenase. Phytochemistry 62:287–292
Nakatani N, Jitoe A, Masuda T, Yonemori S (1991) Constituents of Zingiberaceae. Part I1. Flavonoid constituents of Zingiber zerumbet Smith. Agric Biol Chem 55:455–460
Tahara S (2007) A journey of twenty-five years through the ecological biochemistry of flavonoids. Biosci Biotechnol Biochem 71:1387–1404
Winkel-Shirley B (2001) Flavonoid biosynthesis. A colorful model for genetics, biochemistry, cell biology, and biotechnology. Plant Physiol 126:485–493
Wisman U, Hartmann M, Sagasser E, Baumann K, Palme K, Hahlbrock H, Weisshaar B (1998) Knock-out mutants from an En-1 mutagenized Arabidopsis thaliana population generate phenylpropanoid biosynthesis phenotypes. Proc Natl Acad Sci 95:12432–12437
Yan Y, Li Z, Koffa MA (2008) High-yield anthocyanin biosynthesis in engineered Escherichia coli. Biotechnol Bioeng 100:126–140
Acknowledgments
This work was supported by a grant from the Biogreen 21 Program, Rural Development Administration, Republic of Korea, and Agenda program (NIAS, 11-30-68) and also partially by Priority Research Centers Program through the National Research Foundation of Korea (NRF) funded by the Ministry of Education, Science and Technology (2009-0093824).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kim, BG., Joe, E.J. & Ahn, JH. Molecular characterization of flavonol synthase from poplar and its application to the synthesis of 3-O-methylkaempferol. Biotechnol Lett 32, 579–584 (2010). https://doi.org/10.1007/s10529-009-0188-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10529-009-0188-x