Abstract
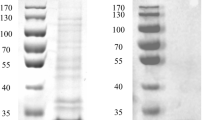
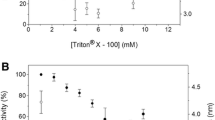
A 60 kDa phospholipase D (PLD) was obtained from Streptomyces olivochromogenes by one-step chromatography on Sepharose CL-6B. Maximal activity was at pH 8 and 75°C and the enzyme was stable from pH 7 to 13 and from 55 to 75°C. Thermal and pH stability with temperature optimum of the enzyme were highest among Streptomyces PLDs reported so far. The activity was Ca2+-dependent and enhanced by detergents. The Km and Vmax values for phosphatidylcholine were 0.6 mM and 650 μmol min−1 mg−1, respectively. In addition, the enzyme also revealed transphosphatidylation activity, which was optimum at pH 8 and 50°C. The first 15 amino acid residues of the N terminal sequence were ADYTPGAPGIGDPYY, which are significantly different from the other known PLDs. The enzyme may therefore be a novel PLD with potential application in the lipid industry.







Similar content being viewed by others
References
Carrea G, D’Arrigo P, Piergianni V, Roncaglio S, Secundo F, Servi S (1995) Purification and properties of two phospholipases D from Streptomyces sp. Biochim Biophys Acta 1255:273–279
Hatanaka T, Negishi T, Kubota-Akizawa M, Hagishita T (2002) Purification, characterization, cloning and sequencing of phospholipase D from Streptomyces septatus TH-2. Enzyme Microb Technol 31:233–241
Imamura S, Horiuti Y (1978) Enzymatic determination of phospholipase D activity with choline oxidase. J Biochem 83:677–680
Juneja LR, Kazuoka T, Goto N, Yamane T, Shimizu S (1989) Conversion of phosphatidylcholine to phosphatidylserine by various phospholipase D in the presence of L- or D-serine. Biochim Biophys Acta 1003:277–283
Leiros I, Secundo F, Zambonelli C, Servi S, Hough E (2000) The first crystal structure of a phospholipase D. Structure 8:655–667
Lerchner A, Mansfeld J, Schaffner I, Schops R, Beer HK, Ulbrich-Hofmann R (2005) Two highly homologous phospholipase D isoenzymes from Papaver somniferum L. with different transphosphatidylation potential. Biochim Biophys Acta 1737:94–101
Lim S, Choi J, Chung M, Lee E, Khang Y, Kim S, Nam D (2002) Production and characterization of extracellular phospholipase D from Streptomyces sp. YU 100. J Microbiol Biotechnol 12:189–195
Liscovitch M, Czarny M, Fiucci G, Tang X (2000) Phospholipase D: molecular and cell biology of a novel gene family. Biochem J 345:401–415
Morris AJ, Frohman MA, Engebrecht J (1997) Measurement of phospholipase D activity. Anal Biochem 252:1–9
Okawa Y, Yamaguchi T (1975) Studies on phospholipases from Streptomyces II. Purification and properties of Streptomyces hachijoensis phospholipase D2. J Biochem 78:363–372
Qin W, Pappan K, Wang X (1997) Molecular heterogeneity of phospholipase D (PLD): cloning of PLDγ and regulation of plant PLDγ,-β, and -α by polyphosphoinositides and calcium. J Biol Chem 272:28267–28273
Shimbo K, Yano H, Miyamoto Y (1990) Purification and properties of phospholipase D from Streptomyces lydicus. Agric Biol Chem 54:1189–1193
Simkhada JR, Cho SS, Lee HJ, Yoo JC (2007) Purification and biochemical properties of phospholipase D (PLD57) produced by Streptomyces sp. CS-57. Arch Pharm Res 30:1302–1308
Ulbrich-Hofmann R, Lerchner A, Oblozinsky M, Bezakova L (2005) Phospholipase D and its application in biocatalysis. Biotechnol Lett 27:535–543
Acknowledgements
This work was supported by a grant RT104-03-03 from the Regional Technology Innovation Program of Ministry of Commerce, Industry, and Energy (MOCIE), Republic of Korea.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Simkhada, J.R., Lee, H.J., Jang, S.Y. et al. A novel alkalo- and thermostable phospholipase D from Streptomyces olivochromogenes . Biotechnol Lett 31, 429–435 (2009). https://doi.org/10.1007/s10529-008-9890-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10529-008-9890-3