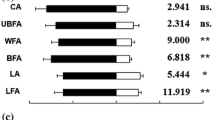
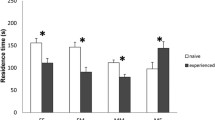
Abstract
Hymenopteran parasitoids can utilize substrate-borne semiochemicals released by conspecifics or by their hosts, increasing the likelihood of successful mating and host location. According to the literature, two substrate-borne chemo-orientation patterns can occur: (1) biased random searching, a non-directional reaction toward the chemicals (kinesis), and (2) trail-following searching, a directional response toward the source emitting the chemical compounds (taxis). These two different strategies can be adopted by parasitoids to locate hosts and mates. In host location, random searching is induced by allelochemicals indirectly associated with the host, whereas trail-following behavior is induced by allelochemicals directly emitted by the target organism. In mate finding, sex pheromones emitted by conspecifics can induce either the random searching or the trail-following behavior, although the spatial distribution of virgin conspecifics could be an important factor driving the evolution of substrate-borne chemo-orientation patterns. The chemical nature of substrate-borne semiochemicals has not yet been fully elucidated. Most studies have shown that crude extracts are biologically active for eliciting parasitoid arrestment response, but few studies have clearly characterized their chemical nature. However, experimental evidence indicates that cuticular lipids located in the external layer of insects’ bodies play a role in parasitoid–parasitoid and host–parasitoid communication. The ecological role of parasitoid chemo-orientation in host and mate location is discussed from a biological control perspective.


Similar content being viewed by others
References
Andow DA, Prokrym DR (1990) Plant structural complexity and host finding by a parasitoid. Oecologia 82:162–165
Ardeh M, De Jong PW, Loomans AJM, van Lenteren JC (2004) Inter- and intraspecific effects of volatile and nonvolatile sex pheromones on males, mating behavior, and hybridization in Eretmocerus mundus and and E. eremicus (Hymenoptera: Aphelinidae). J Insect Behav 17:745–759
Beevers M, Lewis WJ, Gross HR, Nordlund DA (1981) Kairomones and their use for management of entomophagous insects. X. Laboratory studies on manipulation of host-finding behavior of Trichogramma pretiosum Riley (Hymenoptera: Trichogrammatidae) with a kairomone extracted from Heliothis zea (Boddie) moth scales. J Chem Ecol 7:635–648
Bell WJ (1984) Chemo-orientation in walking insects. In: Bell WJ, Cardé RT (eds) Chemical ecology of insects, Sunderland. Sinauer Associates Inc., Mass, USA, pp 93–106
Bell WJ (1990) Searching behavior patterns in insects. Annu Rev Entomol 35:447–467
Bell W, Tobin TR (1981) Orientation to sex-pheromone in the American cockroach—Analysis of chemo-orientation mechanisms. J Insect Physiol 27:501–508
Bell WJ, Tobin TR (1982) Chemo-orientation. Biol Rev 57:219–260
Bell WJ, Kipp LR, Collins RD (1995) The role of chemo-orientation in search behavior. In: Cardé RT, Bell WJ (eds) Chemical ecology of insects 2. Springer, New York, USA, pp 105–152
Bernal JS, Luck RF (2007) Mate finding via a trail sex pheromone by Aphytis melinus DeBach (Hymenoptera: Aphelinidae) males. J Insect Behav 20:515–525
Blenn B, Bandoly M, Kueffner A, Otte T, Geiselhardt S, Fatouros NE, Hilker M (2012) Insect egg deposition induces indirect defense and epicuticular wax changes in Arabidopsis thaliana. J Chem Ecol 38:882–892
Borges M, Colazza S, Ramirez-Lucas P, Chauhan KR, Moraes MCB, Aldrich JR (2003) Kairomonal effect of walking traces from Euschistus heros (Heteroptera: Pentatomidae) on two strains of Telenomus podisi (Hymenoptera: Scelionidae). Physiol Entomol 28:349–355
Bouchard Y, Cloutier C (1984) Honeydew as a source of host-searching kairomones for the aphid parasitoid Aphidius nigripes (Hymenoptera: Aphidiidae). Can J Zool 62:1513–1520
Budenberg W (1990) Honeydew as a contact kairomone for aphid parasitoids. Entomol Exp Appl 55:139–147
Chabi-Olaye A, Schulthess F, Poehling HM, Borgemeister C (2001) Host location and host discrimination behavior of Telenomus isis, an egg parasitoid of the African cereal stem borer Sesamia calamistis. J Chem Ecol 27:663–678
Chiu-Alvarado P, Valle-Mora J, Rojas JC (2010) Chemical cues from the coffee berry borer influence the locomotory behavior of its bethylid parasitoids. Bull Entomol Res 100:707–714
Chiu-Alvarado P, Santiesteban A, Rojas JC (2012) Host kairomones elicit area-restricted search by Prorops nasuta, a parasitoid of the coffee berry borer. Open Entomol J 6:17–21
Colazza S, Salerno G, Wajnberg E (1999) Volatile and contact chemicals released by Nezara viridula (Heteroptera: Pentatomidae) have a kairomonal effect on the egg parasitoid Trissolcus basalis (Hymenoptera: Scelionidae). Biol Control 16:310–317
Colazza S, Aquila G, De Pasquale C, Peri E, Millar J (2007) The egg parasitoid Trissolcus basalis uses n-nonadecane, a cuticular hydrocarbon from its stink bug host Nezara viridula, to discriminate between female and male hosts. J Chem Ecol 33:1405–1420
Colazza S, Lo Bue M, Lo Giudice D, Peri E (2009) The response of Trissolcus basalis to footprint contact kairomones from Nezara viridula females is mediated by leaf epicuticular waxes. Naturwissenschaften 96:975–981
Collatz J, Tolasch T, Steidle JL (2009) Mate finding in the parasitic wasp Cephalonomia tarsalis (Ashmead): more than one way to a female’s heart. J Chem Ecol 35:761–768
Conti E, Salerno G, Bin F, Williams HJ, Vinson SB (2003) Chemical cues from Murgantia histrionica eliciting host location and recognition in the egg parasitoid Trissolcus brochymenae. J Chem Ecol 29:115–130
Conti E, Salerno G, Bin F, Vinson SB (2004) The role of host semiochemicals in parasitoid specificity: a case study with Trissolcus brochymenae and Trissolcus simoni on pentatomid bugs. Biol Control 29:435–444
Conti E, Salerno G, Leombruni B, Frati F, Bin F (2010) Short-range allelochemicals from a plant-herbivore association: a singular case of oviposition-induced synomone for an egg parasitoid. J Exp Biol 213:3911–3919
Danci A, Gries R, Schaefer W, Gries G (2006) Evidence for four-component close range sex pheromone in the parasitic wasp Glyptapanteles flavicoxis. J Chem Ecol 32:1539–1554
Deshpande SA, Kainoh Y (2012) Herbivore egg deposition induces tea leaves to arrest the egg-larval parasitoid Ascogaster reticulata. Entomol Exp Appl 144:172–180
Eigenbrode SD (2004) The effects of plant epicuticular waxy blooms on attachment and effectiveness of predatory insects. Arthropod Struct Dev 33:91–102
Eigenbrode SD, Jetter R (2002) Attachment to plant surface waxes by an insect predator. Integr Comp Biol 42:1091–1099
Fatouros NE, Bukovinszkine‘Kiss G, Kalkers LA, Soler Gamborena R, Dicke M, Hilker M (2005) Oviposition induced plant cues: do they arrest Trichogramma wasps during host location? Entomol Exp Appl 115:207–215
Fatouros NE, Bukovinszkine‘kiss G, Dicke M, Hilker M (2007) The response specificity of Trichogramma egg parasitoids towards infochemicals during host location. J Insect Behav 20:53–65
Fatouros NE, Broekgaarden C, Bukovinszkine‘Kiss G, van Loon JJA, Mumm R, Huigens ME, Dicke M, Hilker M (2008) Male-derived butterfly anti-aphrodisiac mediates induced indirect plant defense. Proc Natl Acad Sci USA 105:10033–10038
Fatouros NE, Pashalidou FG, Aponte Cordero WV, van Loon JJA, Mumm R, Dicke M, Hilker M, Huigens ME (2009) Anti-aphrodisiac compounds of male butterflies increase the risk of egg parasitoid attack by inducing plant synomone production. J Chem Ecol 35:1373–1381
Fauvergue X, Hopper KR, Antolint MF (1995) Mate finding via a trail sex pheromone by a parasitoid wasp. Proc Natl Acad Sci USA 92:900–904
Fauvergue X, Fouillet P, Mesquita ALM, Bouletréau M (1998) Male orientation to trail sex pheromones in parasitoid wasps: does the spatial distribution of virgin females matter? J Insect Physiol 44:667–675
Frati F, Salerno G, Conti E (2013) Cabbage waxes affect Trissolcus brochymenae response to short-range synomones. Insect Sci. doi:10.1111/j.1744-7917.2012.01575.x
Gardner S, Dixon AFG (1985) Plant structure and the foraging success of Aphidius rhopalosiphi (Hymenoptera: Aphidiidae). Ecol Entomol 10:171–179
Gardner S, van Lenteren JC (1986) Characterization of the arrestment responses of Trichogramma evanescens. Oecologia 68:265–270
Gazit Y, Lewis WJ, Tumlison JH (1996) Arrestment of Telenomus remus (Hymenoptera: Scelionidae) by a kairomone associated with eggs of its host, Spodoptera frugiperda (Lepidoptera: Noctuidae). Biol Control 6:283–290
Godfray HCJ (1994) Parasitoids: behavioral and evolutionary ecology. Princeton University Press, Princeton, USA
González JM, Cusumano A, Williams HJ, Colazza S, Vinson SB (2011) Behavioral and chemical investigations of contact kairomones released by the mud dauber wasp Trypoxylon politum, a host of the parasitoid Melittobia digitata. J Chem Ecol 37:629–639
Gross HR (1981) Employment of kairomones in the management of parasitoids. In: Nordlund DA, Jones RL, Lewis WJ (eds) Semiochemicals: their role in pest control. Wiley, New York, USA, pp 137–150
Gueldner R, Nordlund DA, Lewis WJ, Thean JE, Wilson DM (1984) Kairomones and their use for management of entomophagous insects. XV. Identification of several acids in scales of Heliothis zea (Lepidoptera: Noctuidae) moths and comments on their possible role as kairomones for Trichogramma pretiosum (Hymenoptera: Trichogrammatidae). J Chem Ecol 10:245–251
Hardy ICW, Ode PJ, Siva-Jothy M (2005) Mating systems. In: Jervis M (ed) Insects as natural enemies. Springer, Dordrecht, The Netherlands, pp 261–306
Hoffmeister T, Roitberg BD, Lalonde RG (2000) Catching Ariadne by her thread: how a parasitoid exploits the herbivore’s marking trails to locate its host? Entomol Exp Appl 95:77–85
Howard RW, Flinn PW (1990) Larval trails of Cryptolestes ferrugineus (Coleoptera: Cucujidae) as kairomonal host-finding cues for the parasitoid Cephalonomia waterstoni (Hynenoptera: Bethylidae). Ann Entomol Soc Am 83:239–245
Howard RW, Charlton M, Charlton RE (1998) Host-finding, host recognition, and host-acceptance behavior of Cephalonomia tarsalis (Hynenoptera: Bethylidae). Ann Entomol Soc Am 91:879–889
Jander R (1963) Insect orientation. Annu Rev Entomol 8:95–114
Jones R, Lewis WJ, Bowman MC, Beroza M, Bierl BA (1971) Host-seeking stimulant for parasite of corn earworm—isolation, identification, and synthesis. Science 173:842–843
Jones R, Lewis WJ, Beroza M, Bierl BA, Sparks AN (1973) Host-seeking stimulants (kairomones) for the egg parasite, Trichogramma evanescens. Environ Entomol 2:593–596
Kainoh Y, Nemoto T, Shimizu K, Tatsuki S, Kusano T, Kuwahara Y (1991) Mating-behavior of Ascogaster reticulatus Watanabe (Hymenoptera: Braconidae), an egg-larval parasitoid of the smaller tea tortrix, Adoxophyes sp. (Lepidoptera: Tortricidae). 3. Identification of a sex-pheromone. Appl Entomol Zool 26:543–549
Kamano Y, Shimizu K, Kainoh Y, Tatsuki S (1989) Mating-behavior of Ascogaster reticulatus Watanabe (Hymenoptera: Braconidae), an egg-larval parasitoid of the smaller tea tortrix, Adoxophyes sp. (Lepidoptera: Tortricidae). 2. Behavioral sequence and a role of sex-pheromone. Appl Entomol Zool 24:372–378
Kapranas A, Lo Giudice D, Peri E, Millar JG, Colazza S (2013) Emergence, dispersal, and mate finding via a substrate-borne sex pheromone in the parasitoid Metaphycus luteolus (Hymenoptera: Encyrtidae). Entomol Exp Appl 148:74–83
Klomp H (1981) Parasitic wasps as sleuth hounds—response of an ichneumon wasp to the trail of its host. Neth J Zool 31:762–772
Laing J (1937) Host-finding by insect parasites. 1. Observations on the finding of hosts by Alysia manducator, Mormoniella vitripennis and Trichogramma evanescens. J Anim Ecol 6:298–317
Lewis W, Jones RL (1971) Substance that stimulates host-seeking by Microplitis croceipes (Hymenoptera: Braconidae), a parasite of Heliothis species (Lepidoptera: Noctuidae). Ann Entomol Soc Am 64:471–473
Lewis WJ, Jones RL, Nordlund DA, Gross HR Jr (1975a) Kairomones and their use for management of entomophagous insects: II. Mechanisms causing increase in rate of parasitization by Trichogramma spp. J Chem Ecol 1:349–360
Lewis WJ, Jones RL, Nordlund DA, Sparks AN (1975b) Kairomones and their use for management of entomophagous insects: I. Evaluation for increasing rates of parasitization by Trichogramma spp. in the field. J Chem Ecol 1:343–347
Lewis WJ, Beevers M, Nordlund DA, Gross HR Jr, Hagen KS (1979) Kairomones and their use for management of entomophagous insects. IX. Investigations of various kairomone-treatment patterns for Trichogramma spp. J Chem Ecol 5:673–680
Lo Giudice D, Riedel M, Rostás M, Peri E, Colazza S (2011) Host sex discrimination by an egg parasitoid on brassica leaves. J Chem Ecol 37:622–628
Lockey K (1988) Lipids of the insect cuticle—Origin, composition and function. Comp Biochem Physiol Part B 89:595–645
Loke WH, Ashley TR (1984a) Sources of fall armyworm, Spodoptera frugiperda (Lepidoptera: Noctuidae), kairomones eliciting host-finding behavior in Cotesia (=Apanteles) marginiventris (Hymenoptera: Braconidae). J Chem Ecol 10:1019–1027
Loke WH, Ashley TR (1984b) Behavioral and biological responses of Cotesia marginiventris to kairomones of the Fall Armyworm, Spodoptera frugiperda. J Chem Ecol 10:521–529
Mandour NS, Ren SX, Qiu BL (2007) Effect of Bemisia tabaci honeydew and its carbohydrates on search time and parasitization of Encarsia bimaculata. J Appl Entomol 131:645–651
Mehrnejad MR, Copland MJW (2006) Behavioral responses of the parasitoid Psyllaephagus pistaciae (Hymenoptera: Encyrtidae) to host plant volatiles and honeydew. Entomol Sci 9:31–37
Meiners T, Peri E (2013) Chemical ecology of insect parasitoids: essential elements for developing effective biological control programmes. In: Wajnberg E, Colazza S (eds) Chemical ecology of insect parasitoids. Wiley-Blackwell, UK, pp 193–224
Meiners T, Kopf A, Stein C, Hilker M (1997) Chemical signals mediating interactions between Galeruca tanaceti L. (Coleoptera, Chrysomelidae) and its egg parasitoid Oomyzus galerucivorus (Hedqvits) (Hymenoptera: Eulophidae). J Insect Behav 10:523–539
Noldus LPJJ (1989) Semiochemicals, foraging behaviour and quality of entomophagous insects for biological control. J Appl Entomol 108:425–451
Noldus LPJJ, van Lenteren JC (1985) Kairomones for the egg parasite Trichogramma evanescens Westwood, (Hymenoptera: Trichogrammatidae). II. Effect of contact chemicals produced by two of its hosts, Pieris brassicae L., (Lepidoptera: Pieridae) and Pieris rapae L., (Lepidoptera: Pieridae). J Chem Ecol 11:793–800
Obrycki JJ (1986) The influence of foliar pubescence on entomophagous species. In: Boethel DJ, Eikenbarry RD (eds) Interactions of plant resistance and parasitoids and predators of insects. Wiley, New York, USA, pp 61–83
Pashalidou FG, Huigens ME, Dicke M, Fatouros NE (2010) The use of oviposition-induced plant cues by Trichogramma egg parasitoids. Ecol Entomol 35:748–753
Peri E, Sole MA, Wajnberg E, Colazza S (2006) Effect of host kairomones and oviposition experience on the arrestment behavior of an egg parasitoid. J Exp Biol 209:3629–3635
Pompanon F, De Schepper B, Mourer Y, Fouillet P, Boulétreau M (1997) Evidence for a substrate-borne sex pheromone in the parasitoid wasp Trichogramma brassicae. J Chem Ecol 23:1349–1360
Powell W, Pickett JA (2003) Manipulation of parasitoids for aphid pest management: progress and prospects. Pest Manag Sci 59:149–155
Puente ME, Kennedy GG, Gould F (2008) The impact of herbivore-induced plant volatiles on parasitoid foraging success: a general deterministic model. J Chem Ecol 34:945–958
Quicke DLJ (1997) Parasitic wasps. Chapman and Hall, London, UK
Rogers M, Potter DA (2002) Kairomones from scarabaeid grubs and their frass as cues in below-ground host location by the parasitoids Tiphia vernalis and Tiphia pygidialis. Entomol Exp Appl 102:307–314
Romani R, Isidoro N, Bin F (2010) Antennal structures used in communication by egg parasitoids. In: Parra JRP, Consôli FL, Zucchi RA (eds) Egg parasitoids in agroecosystems with emphasis on Trichogramma. Springer, Dordrecht, The Netherlands, pp 57–96
Romeis J, Zebitz CPW (1997) Searching behaviour of Encarsia formosa as mediated by colour and honeydew. Entomol Exp Appl 82:299–309
Romeis J, Shanower TG, Zebitz CPW (2003) Physical and chemical plant characters inhibiting the searching behavior of Trichogramma chilonis. Entomol Exp Appl 87:275–284
Rostás M, Wölfling M (2009) Caterpillar footprints as host location kairomones for Cotesia marginiventris: persistence and chemical nature. J Chem Ecol 35:20–27
Rostás M, Ruf D, Zabka V, Hildebrandt U (2008) Plant surface wax affects parasitoid’s response to host footprints. Naturwissenschaften 95:997–1002
Ruther J (2013) Novel insights into pheromone-mediated communication in parasitic Hymenopterans. In: Wajnberg E, Colazza S (eds) Chemical ecology of insect parasitoids. Wiley-Blackwell, UK, pp 112–144
Ruther J, Steidle JLM (2000) Mites as matchmakers: semiochemicals from host-associated mites attract both sexes of the parasitoid Lariophagus distinguendus. J Chem Ecol 26:1205–1217
Ruther J, Meiners T, Steidle JL (2002) Rich in phenomena-lacking in terms. A classification of kairomones. Chemoecology 12:161–167
Salerno G, De Santis F, Iacovone A, Bin F, Conti E (2013) Short-range cues mediate parasitoid searching behavior on maize: the role of oviposition-induced plant synomones. Biol Control 64:247–254
Schmidt J, Carter MH (1992) The locomotory response of the egg parasitoid Trichogramma evanescens Westwood to hexane extracts of Eastern Spruce Budworm scales Choristoneura fumiferana (Clemens). Can J Zool 70:941–949
Shaltiel L, Ayal Y (1998) The use of kairomones for foraging decisions by an aphid parasitoid in small host aggregations. Ecol Entomol 2:319–329
Shimron O, Heftz A, Gerling D (1992) Arrestment responses of Eretmocerus species and Encarsia deserti (Hymenoptera: Aphelinidae) to Bemisia tabaci honeydew. J Insect Behav 5:517–526
Shorey HH (1976) Mechanisms of movement and orientation to pheromone sources. In: Shorey HH (ed) Animal communication by pheromones. Academic Press, New York, USA, pp 249–284
Shorey HH (1977) Interaction of insects with their chemical environment. In: Shorey HH, McKelvey JJ (eds) Chemical control of insect behavior: theory and application. Wiley, New York, USA, pp 1–5
Shu S, Jones RL (1989) Kinetic effects of a kairomone in moth scales of the European corn borer on Trichogramma nubilale Ertle and Davis (Hymenoptera:Trichogrammatidae). J Insect Behav 2:123–131
Shu S, Swedenborg PD, Jones RL (1990) A kairomone for Trichogramma nubilale (Hymenoptera: Trichogrammatidae)—Isolation, identification, and synthesis. J Chem Ecol 16:521–529
Smith GS, Allison JCS, Pammenter NW (1994) Bioassay study of response by a parasitoid to frass and feeding substrates of its host, the Stalk Borer Eldana saccharina. Ann Appl Biol 125:439–446
Strand M, Williams HJ, Vinson SB, Mudd A (1989) Kairomonal activities of 2-acylcyclohexane-1,3 diones produced by Ephestia kuehniella Zeller in eliciting searching behavior by the parasitoid Bracon hebetor (Say). J Chem Ecol 15:1491–1500
Takabayashi J, Takahashi S (1986a) Effect of kairomones in the host searching behavior of Apanteles kariyai Watanabe (Hymenoptera: Braconidae), a parasitoid of the common armyworm, Pseudaletia separata Walker (Lepidoptera: Noctuidae). 2. Isolation and identification of arrestants produced by the host larvae. Appl Entomol Zool 21:114–118
Takabayashi J, Takahashi S (1986b) Effect of kairomones in the host searching behavior of Apanteles kariyai Watanabe (Hymenoptera: Braconidae), a parasitoid of the common armyworm, Pseudaletia separata Walker (Lepidoptera: Noctuidae). 3. Synthesis and bioassay of arrestants and related-compounds. Appl Entomol Zool 21:519–524
Takabayashi J, Noda T, Takahashi S (1985) Effect of kairomones in the host searching behavior of Apanteles kariyai Watanabe (Hymenoptera: Braconidae), a parasitoid of the common armyworm, Pseudaletia separata Walker (Lepidoptera: Noctuidae). 1. Presence of arresting stimulants produced by the host larvae. Appl Entomol Zool 20:484–489
Thibout E (2005) Role of caterpillar silk thread in location of host pupae by the parasitoid Diadromus pulchellus. J Insect Behav 18:817–826
Vinson SB (1977) Behavioural chemicals in the augmentation of natural enemies. In: Ridgway RL, Vinson SB (eds) Biological control by augmentation of natural enemies. Plenum, New York, USA, pp 237–279
Vinson SB (1998) The general host selection behavior of parasitoid Hymenoptera and a comparison of initial strategies utilized by larvaphagous and oophagous species. Biol Control 11:79–96
Vinson SB (2010) Nutritional ecology of insect egg parasitoids. In: Consôli FL, Parra JRP, Zucchi RA (eds) Egg parasitoids in agroecosystems with emphasis on Trichogramma. Springer, Dordrecht, The Netherlands, pp 25–56
Vinson SB, Lewis WJ (1965) A method of host selection by Cardiochiles nigriceps. J Econ Entomol 58:869–871
Vinson SB, Jones RL, Sonnet PE, Bierl BA, Beroza M (1975) Isolation, identification and synthesis of host-seeking stimulants for Cardiochiles nigriceps, a parasitoid of tobacco budworm. Entomol Exp Appl 18:443–450
Vinson S, Harlan DP, Hart WG (1978) Response of the parasitoid Microterys flavus to the brown soft scale and its honeydew. Environ Entomol 7:874–878
Waage JK (1978) Arrestment responses of parasitoid, Nemeritis canescens, to a contact chemical produced by its host, Plodia interpunctella. Physiol Entomol 3:135–146
Zaborski E, Teal PEA, Laing JE (1987) Kairomone mediated host finding by spruce budworm egg parasite, Trichogramma minutum. J Chem Ecol 13:113–122
Acknowledgments
We thank John Ruberson, Department of Entomology Kansas State University, USA, for critically reading and commenting on an earlier version of this manuscript. This work was supported by the University of Palermo—“Fondo Finalizzato alla Ricerca (FFR) bando 2012”.
Author information
Authors and Affiliations
Corresponding author
Additional information
Handling Editor: Dirk Babendreier.
Rights and permissions
About this article
Cite this article
Colazza, S., Cusumano, A., Lo Giudice, D. et al. Chemo-orientation responses in hymenopteran parasitoids induced by substrate-borne semiochemicals. BioControl 59, 1–17 (2014). https://doi.org/10.1007/s10526-013-9546-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10526-013-9546-7