Abstract
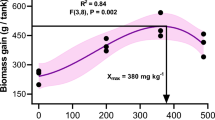
This study aimed to evaluate the effect of dietary supplementation of α-lipoic acid (LA) in the growth, blood chemistry, and liver antioxidant status of African catfish Clarias gariepinus. This study applied a completely randomised experimental design consisting of five experimental diets (30% protein and 4% lipid) with graded levels of LA, i.e. LA0.0 as the control (0.0 g LA kg−1 feed), LA0.5 (0.5g LA kg−1 feed), LA1.0 (1.0 g LA kg−1 feed), LA1.5 (1.5 g LA kg−1 feed), and LA2.0 (2.0 g kg−1 feed) with three replications. An orthogonal polynomial regression analysis was used to determine the optimum dose of LA for fish growth. Seventy-five catfish juveniles (14.65±0.05 g) were randomly distributed into each fibre tank (600 L in capacity) and maintained in zero water exchange condition for 60 days. The fish were fed twice a day to apparent satiation at 08.00 AM and 05.00 PM (GMT+7). Fish survival was at a range of 93.78–97.78% and was not significantly different amongst the treatments (P>0.05). The final body weight, biomass and specific growth rate of the fish fed with LA supplemented diets were generally higher than those of the control. Higher feed intake and feed protein retention were also higher in LA treatment groups compared to that of the control, but there were no significant differences in the feed conversion ratio. In the present study, visual observation on the liver colour was used to indicate the liver condition. Abnormal coloured fish liver (pale coloured) was found to be higher in lipid and malondialdehyde, and lower in superoxide dismutase (SOD) activity than those of normal coloured (red coloured) liver. The percentage of normal coloured liver (red coloured liver) in the control (28.8 %) was lower than that of the LA treatment groups (46.7–70 %). Antioxidant status of the fish in LA treatment groups was superior than that of the control as indicated by the lower levels of MDA and the higher SOD activities. Regardless of the dose of LA, all fish fed on LA supplemented diets demonstrated significantly lower plasma alanine aminotransferase and aspartate aminotransferase levels than those of the control (P<0.05). The fish in LA0.5, LA1.0, and LA1.5 treatments had significantly lower cholesterol and low-density lipoprotein (LDL) and higher high-density lipoprotein (HDL) levels compared to the control and LA2.0 group (P<0.05). In conclusion, the present study demonstrated that dietary supplementation of LA could enhance the growth, blood chemistry, and liver antioxidant status of African catfish juveniles. Based on the polynomial orthogonal analyses, an LA dietary supplementation level of 1.21 g kg−1 feed can be recommended for optimum growth of African catfish.


Similar content being viewed by others
References
AOAC (2012) Official method of analysis of AOAC International. Gaithersburg, Marylands
APHA-AWWA-WEF (1998) Standard method for the examination of water and wastewater. Washington D.C, Washington.
Ali M, Esawy BHE, Elmorsy EA (2015) Effect of combining an angiotensin-converting enzyme inhibitor and a vitamin D receptor activator on renal oxidative and nitrosative stress in diabetic rats. Natl J Physiol Pharm Pharmacol 5(3):222–231. https://doi.org/10.5455/njppp.2015.5.1401201522
Arvind A, Osganian SA, Cohen DE, Corey KE (2000) Lipid and lipoprotein metabolism in liver disease. Endotext. https://www.ncbi.nlm.nih.gov/books/NBK326742/. Accessed 20 June 2020
Baeeri M, Bahadar H, Rahimifard M, Navaei-Nigjeh M, Khorasani R, Rezvanfar MA, Gholami M, Abdollahi M (2019) α-Lipoic acid prevents senescence, cell cycle arrest, and inflammatory cues in fibroblasts by inhibiting oxidative stress. Pharmacol Res 141:214–223. https://doi.org/10.1016/j.phrs.2019.01.003
Butler JA, Hagen TM, Moreau R (2009) Lipoic acid improves hypertriglyceridemia by stimulating triacylglycerol clearance and downregulating liver triacylglycerol secretion. Arch Biochem Biophys 485:63–71. https://doi.org/10.1016/j.abb.2009.01.024
Chen WL, Kang CH, Wang SG, Lee HM (2012) α-Lipoic acid regulates lipid metabolism through induction of sirtuin 1 (SIRT1) and activation of AMP-activated protein kinase. Diabetologia 55:1824–1835. https://doi.org/10.1007/s00125-012-2530-4
Dyk JC, Cochrane MJ, Wagenaar GM (2012) Liver histopathology of the sharptooth catfish Clarias gariepinus as a biomarker of aquatic pollution. Chemosphere 87:301–311. https://doi.org/10.1016/j.chemosphere.2011.12.002
Friedewald WT, Levy RI, Fredrickson DS (1972) Estimation of the concentration of low-density lipoprotein cholesterol in plasma without use of the preparative ultra-centrifuge. Clin Chem 18:499–502. https://doi.org/10.1093/clinchem/18.6.499
Ghahderijani MS, Hajimoradloo A, Ghorbani R, Roohi Z (2016) The effects of garlic-supplemented diets on skin mucosal immune responses, stress resistance and growth performance of the caspian roach (Rutilus rutilus) fry. Fish Shellfish Immunol 49:79–83. https://doi.org/10.1016/j.fsi.2015.12.021
Huang CC, Sun J, Ji H, Oku H, Chang ZG, Tian JJ, Yu EM, Xie J (2019) Influence of dietary alpha-lipoic acid and lipid level on the growth performance, food intake and gene expression of peripheral appetite regulating factors in juvenile grass carp (Ctenopharyngodon idellus). Aquaculture 505:412–422. https://doi.org/10.1016/j.aquaculture.2019.02.054
Islam MT (2009) Antioxidant activities of dithiol alpha-lipoic acid. Bangladesh Journal of. Med Sci 8:254–265. https://doi.org/10.3329/bjms.v8i3.3982
Kagan VE, Shvedova A, Serbinova E, Khan S, Swanson C, Powell R, Packer L (1992) Dihydrolipoic acid—a universal antioxidant both in the membrane and in the aqueous phase: reduction of peroxyl, ascorbyl and chromanoxyl radicals. Biochem Pharmacol 44:1637–1649. https://doi.org/10.1016/0006-2952(92)90482-X
Kütter MT, Monserrat JM, Primel EG, Caldas SS, Tesser MB (2012) Effects of dietary α-lipoic acid on growth, body composition and antioxidant status in the plata pompano Trachinotus marginatus (Pisces, Carangidae). Aquaculture 368:29–35. https://doi.org/10.1016/j.aquaculture.2012.09.010
Kütter MT, Romano LA, Venturalima J, Tesser MB, Monserrat JM (2014) Antioxidant and toxicological effects elicited by alpha-lipoic acid in aquatic organisms. Comparat Biochem Physiol Part C 162:70–76. https://doi.org/10.1016/j.cbpc.2014.03.008
Kuo YT, Lin TH, Chen WL, Lee HM (2012) Alpha-lipoic acid induces adipose triglyceride lipase expression and decreases intracellular lipid accumulation in HepG2 cells. Eur J Pharmacol 692:10–18. https://doi.org/10.1016/j.ejphar.2012.07.028
Lefevre S, Wang T, Jensen A, Cong NV, Huong DTT, Phuong NT, Bayley M (2014) Air-breathing fishes in aquaculture. What can we learn from physiology? J Fish Biol 84:705–731. https://doi.org/10.1111/jfb.12302
Longaray-Garcia M, Flores JA, Külkamp-Guerreiro IC, Guterres SS, Pereira TCB, Bogo MR, Monserrat JM (2013) Modulation of antioxidant and detoxifying capacity in fish Cyprinus carpio (Cyprinidae) after treatment with nanocapsules containing lipoic acid. Compar Biochem Physiol Part A 165:468–475. https://doi.org/10.1016/j.cbpa.2013.02.004
Lu DL, Limbu SM, Lv HB, Ma Q, Chen LQ, Zhang ML, Du ZY (2019) The comparisons in protective mechanisms and efficiencies among dietary α-lipoic acid, β-glucan and l-carnitine on Nile tilapia infected by Aeromonas hydrophila. Fish Shellfish Immunol 86:785–793. https://doi.org/10.1016/j.fsi.2018.12.023
Monserrat JM, Ventura JL, Ferreira R, Acosta D, Garcia ML, Ramos PB, Moraes TB, Dos-Santos LC, Amado LL (2008) Modulation of antioxidant and detoxification responses mediated by lipoic acid in the fish Corydoras paleatus (Callychthyidae). Compar Biochem Physiol C: Toxicol Pharmacol 148(3):287–292. https://doi.org/10.1016/j.cbpc.2008.06.011
Monserrat JM, Garcia ML, Ventura-Lima J, González M, Ballesteros ML, Miglioranza KSB, Amé MV, Wunderlin DA (2014) Antioxidant, phase II and III responses induced by lipoic acid in the fish Jenynsia multidentata (Anablapidae) and its influence on endolsulfan accumulation and toxicity. Pestic Biochem Physiol 108:8–15. https://doi.org/10.1016/j.pestbp.2013.10.009
Nwani CD, Lakra WS, Nagpure NS, Kumar R, Kushwaha B, Srivastava SK (2010) Toxicity of the herbicide atrazine: effects on lipid perodixidation and activites of antioxidant enzymes in the freshwater fish Channa punctatus (Bloch). Int J Environ Res Public Health 7(8):3298–3312. https://doi.org/10.3390/ijerph7083298
Odabasoglu F, Halici Z, Aygun H, Halici M, Atalay F, Cakir A, Suleyman H (2011) α-Lipoic acid has anti-inflammatory and anti-oxidative properties: an experimental study in rats with carrageenan induced acute and cotton pellet-induced chronic inflammations. Br J Nutr 105:31–43. https://doi.org/10.1017/S0007114510003107
Oliva-Teles A (2012) Nutrition and health of aquaculture fish. J Fish Dis 35(2):83–108. https://doi.org/10.1111/j.1365-2761.2011.01333.x
Palm HW, Knaus U, Wasenitz B, Bischoff AA, Strauch SM (2018) Proportional up scaling of African catfish (Clarias gariepinus Burchell, 1822) commercial recirculating aquaculture systems disproportionally affects nutrient dynamics. Aquaculture 491:155–168. https://doi.org/10.1016/j.aquaculture.2018.03.021
Schram E, Roques JA, Abbink W, Spanings T, De Vries P, Bierman S, van de Vis H, Flik G (2010) The impact of elevated water ammonia concentration on physiology, growth and feed intake of African catfish (Clarias gariepinus). Aquaculture 306:108–115. https://doi.org/10.1016/j.aquaculture.2010.06.005
Schumann G, Klauke R (2003) New IFCC reference procedures for the determination of catalytic activity concentrations of five enzymes in serum: preliminary upper reference limits obtained in hospitalized subjects. Clin Chim Acta 327:69–79. https://doi.org/10.1016/S0009-8981(02)00341-8
Seo EY, Ha AW, Kim WK (2012) α-Lipoic acid reduced weight gain and improved the lipid profile in rats fed with high fat diet. Nutr Res Pract 6:195–200. https://doi.org/10.4162/nrp.2012.6.3.195
Shay KP, Moreau RF, Smith EJ, Smith AR, Hagen TM (2009) Alpha-lipoic acid as a dietary supplement: molecular mechanisms and therapeutic potential. Biochim Biophys Acta 1790:1149–1160. https://doi.org/10.1016/j.bbagen.2009.07.026
Shi XC, Jin A, Sun J, Yang Z, J-j T, Ji H, Yu HB, Li Y, Zhou JS, Du ZY, Chen LQ (2017) Asam alpha-lipoic acid ameliorates n-3 highly-unsaturated fatty acids induced lipid peroxidation via regulating antioxidant defenses in grass carp (Ctenopharyngodon idellus). Fish Shellfish Immunol 67:359–367
Thurston RV (1979) Aqueous ammonia equilibrium: tabulation of percent un-ionized ammonia. Environmental Research Laboratory-Duluth, Office of Research and Development, US Environmental Protection Agency
Trattner S, Pickova J, Park KH, Rinchard J, Dabrowski K (2007) Effects of alpha-lipoic and ascorbic acid on the muscle and brain fatty acids and antioxidant profile of the South American pacu Piaractus mesopotamicus. Aquaculture 273:158–164. https://doi.org/10.1016/j.aquaculture.2007.09.025
Xu F, Xu C, Xiao S, Lu M, Limbu SM, Wang X, Du Z, Qin JG, Chen L (2018) Effects of α-lipoic acid on growth performance, body composition, antioxidant profile and lipid metabolism of the GIFT tilapia (Oreochromis niloticus) fed high-fat diets. Aquac Nutr 25:585–596. https://doi.org/10.1111/anu.12881
Zhang J, Zhou X, Wu W, Wang J, Xie H, Wu Z (2018) Regeneration of glutathione by alpha-lipoic acid via Nrf2/ARE signaling pathway alleviates cadmium-induced HepG2 cell toxicity. Environ Toxicol Pharmacol 51:30–37. https://doi.org/10.1016/j.etap.2017.02.022
Availability of data and material
The data that support the findings of this study are available in corresponding author upon reasonable request.
Code availability
Not applicable.
Funding
This research was funded by the Directorate General of Higher Education of The Ministry of Research, Technology and Higher Education, Indonesia grant number 1/E1/KP.PTNBH/2020/dated 12 Mei 2020.
Author information
Authors and Affiliations
Contributions
Desi Rahmadani Siagian: experimental designing, research implementation, data analyses, and manuscript preparation. Dedi Jusadi: conceptualization, experimental designing, data analyses, and manuscript preparation. Julie Ekasari: conceptualization, data analyses, and manuscript preparation. Mia Setiawati: conceptualization, data analysis, and manuscript preparation.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
The experimental procedures have been following the ethical guidelines from Animal Care and Use Committee of IPB University, Indonesia. All authors consented to participate in all aspects of this research and publication.
Consent for publication
All authors agree to publish this manuscript in Aquaculture International journal.
Conflict of interest
The authors declare no competing interests.
Additional information
Handling Editor: Gavin Burnell
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Siagian, D.R., Jusadi, D., Ekasari, J. et al. Dietary α-lipoic acid supplementation to improve growth, blood chemistry, and liver antioxidant status of African catfish Clarias gariepinus. Aquacult Int 29, 1935–1947 (2021). https://doi.org/10.1007/s10499-021-00726-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10499-021-00726-1