Abstract
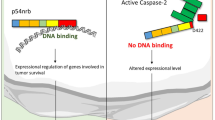
Sp1 is a ubiquitous transcription factor that regulates many genes involved in apoptosis and senescence. Sp1 also has a role in the DNA damage response; at low levels of DNA damage, Sp1 is phosphorylated by ATM and localizes to double-strand break sites where it facilitates DNA double-strand-break repair. Depletion of Sp1 increases the sensitivity of cells to DNA damage, whereas overexpression of Sp1 can drive cells into apoptosis. In response to a variety of stimuli, Sp1 can be regulated through proteolytic cleavage by caspases and/or degradation. Here, we show that activation of apoptosis through DNA damage or TRAIL-mediated activation of the extrinsic apoptotic pathway induces caspase-mediated cleavage of Sp1. Cleavage of Sp1 was coincident with the appearance of cleaved caspase 3, and produced a 70 kDa Sp1 product. In vitro analysis revealed a novel caspase cleavage site at aspartic acid 183. Mutation of aspartic acid 183 to alanine conferred resistance to cleavage, and ectopic expression of the Sp1 D183A rendered cells resistant to apoptotic stimuli, indicating that Sp1 cleavage is involved in the induction of apoptosis. The 70 kDa product resulting from caspase cleavage of Sp1 comprises amino acids 184–785. This truncated form, designated Sp1-70C, which retains transcriptional activity, induced apoptosis when overexpressed in normal epithelial cells, whereas Sp1D183A induced significantly less apoptosis. Together, these data reveal a new caspase cleavage site in Sp1 and demonstrate for the first time that caspase cleavage of Sp1 promotes apoptosis.








Similar content being viewed by others
References
Azizkhan JC, Jensen DE, Pierce AJ, Wade M (1993) Transcription from TATA-less promoters: dihydrofolate reductase as a model. Crit Rev Eukaryot Gene Express 3:229–254
Smale ST, Schmidt MC, Berk AJ, Baltimore D (1990) Transcriptional activation by Sp1 as directed through TATA or initiator: specific requirement for mammalian transcription factor IID. Proc Natl Acad Sci USA 87:4509–4513
Milavetz BI (2002) SP1 and AP-1 elements direct chromatin remodeling in SV40 chromosomes during the first 6 h of infection. Virology 294:170–179
Marin M, Karis A, Visser P, Grosveld F, Philipsen S (1997) Transcription factor Sp1 is essential for early embryonic development but dispensable for cell growth and differentiation. Cell 89:619–628
Tan NY, Khachigian LM (2009) Sp1 phosphorylation and its regulation of gene transcription. Mol Cell Biol 29:2483–2488
Swingler TE, Kevorkian L, Culley KL, Illman SA, Young DA, Parker AE, Lohi J, Clark IM (2010) MMP28 gene expression is regulated by Sp1 transcription factor acetylation. Biochem J 427:391
Majumdar G, Wright J, Markowitz P, Martinez-Hernandez A, Raghow R, Solomon SS (2004) Insulin stimulates and diabetes inhibits O-linked N-acetylglucosamine transferase and O-glycosylation of Sp1. Diabetes 53:3184–3192
Brasse-Lagnel C, Fairand A, Lavoinne A, Husson A (2003) Glutamine stimulates argininosuccinate synthetase gene expression through cytosolic O-glycosylation of Sp1 in Caco-2 cells. J Biol Chem 278:52504–52510
Kang HT, Ju JW, Cho JW, Hwang ES (2003) Down-regulation of Sp1 activity through modulation of O-glycosylation by treatment with a low glucose mimetic, 2-deoxyglucose. J Biol Chem 278:51223–51231
Majumdar G, Harmon A, Candelaria R, Martinez-Hernandez A, Raghow R, Solomon SS (2003) O-Glycosylation of Sp1 and transcriptional regulation of the calmodulin gene by insulin and glucagon. Am J Physiol Endocrinol Metab 285:E584–E591
Spengler ML, Brattain MG (2006) Sumoylation inhibits cleavage of Sp1 N-terminal negative regulatory domain and inhibits Sp1-dependent transcription. J Biol Chem 281:5567–5574
Beishline K, Kelly CM, Olofsson BA, Koduri S, Emrich J, Greenberg RA, Azizkhan-Clifford J (2012) Sp1 Facilitates DNA double-strand break repair through a nontranscriptional mechanism. Mol Cell Biol 32:3790–3799
Olofsson BA, Kelly CM, Kim J, Hornsby SM, Azizkhan-Clifford J (2007) Phosphorylation of Sp1 in response to DNA damage by ataxia telangiectasia-mutated kinase. Mol Cancer Res 5:1319–1330
Deniaud E, Baguet J, Mathieu AL, Pages G, Marvel J, Leverrier Y (2006) Overexpression of Sp1 transcription factor induces apoptosis. Oncogene 25:7096–7105
Deniaud E, Baguet J, Chalard R, Blanquier B, Brinza L, Meunier J, Michallet MC, Laugraud A, Ah-Soon C, Wierinckx A, Castellazzi M, Lachuer J, Gautier C, Marvel J, Leverrier Y (2009) Overexpression of transcription factor Sp1 leads to gene expression perturbations and cell cycle inhibition. PLoS ONE 4:e7035
Chuang JY, Wu CH, Lai MD, Chang WC, Hung JJ (2009) Overexpression of Sp1 leads to p53-dependent apoptosis in cancer cells. Int J Cancer 125:2066–2076
Kavurma MM, Santiago FS, Bonfoco E, Khachigian LM (2001) Sp1 phosphorylation regulates apoptosis via extracellular FasL-Fas engagement. J Biol Chem 276:4964–4971
Chou CF, Peng HW, Wang CY, Yang YT, Han SH (2000) An Sp1 binding site involves the transcription of the Fas ligand gene induced by PMA and ionomycin in Jurkat cells. J Biomed Sci 7:136–143
Black AR, Black JD, Azizkhan-Clifford J (2001) Sp1 and kruppel-like factor family of transcription factors in cell growth regulation and cancer. J Cell Physiol 188:143–160
Xu R, Zhang P, Huang J, Ge S, Lu J, Qian G (2007) Sp1 and Sp3 regulate basal transcription of the survivin gene. Biochem Biophys Res Commun 356:286–292
Li Y, Xie M, Yang J, Yang D, Deng R, Wan Y, Yan B (2006) The expression of antiapoptotic protein survivin is transcriptionally upregulated by DEC1 primarily through multiple sp1 binding sites in the proximal promoter. Oncogene 25:3296–3306
Saitoh Y, Yaginuma Y, Ishikawa M (1999) Analysis of Bcl-2, Bax and Survivin genes in uterine cancer. Int J Oncol 15:137–141
Falleni M, Pellegrini C, Marchetti A, Roncalli M, Nosotti M, Palleschi A, Santambrogio L, Coggi G, Bosari S (2005) Quantitative evaluation of the apoptosis regulating genes Survivin, Bcl-2 and Bax in inflammatory and malignant pleural lesions. Lung Cancer 48:211–216
Dasari A, Bartholomew JN, Volonte D, Galbiati F (2006) Oxidative stress induces premature senescence by stimulating caveolin-1 gene transcription through p38 mitogen-activated protein kinase/Sp1-mediated activation of two GC-rich promoter elements. Cancer Res 66:10805–10814
Volonte D, Zhang K, Lisanti MP, Galbiati F (2002) Expression of caveolin-1 induces premature cellular senescence in primary cultures of murine fibroblasts. Mol Biol Cell 13:2502–2517
Brown JP, Wei W, Sedivy JM (1997) Bypass of senescence after disruption of p21CIP1/WAF1 gene in normal diploid human fibroblasts. Science 277:831–834
Su K, Roos MD, Yang X, Han I, Paterson AJ, Kudlow JE (1999) An N-terminal region of Sp1 targets its proteasome-dependent degradation in vitro. J Biol Chem 274:15194–15202
Han I, Kudlow JE (1997) Reduced O glycosylation of Sp1 is associated with increased proteasome susceptibility. Mol Cell Biol 17:2550–2558
Mortensen ER, Marks PA, Shiotani A, Merchant JL (1997) Epidermal growth factor and okadaic acid stimulate Sp1 proteolysis. J Biol Chem 272:16540–16547
Piedrafita FJ, Pfahl M (1997) Retinoid-induced apoptosis and Sp1 cleavage occur independently of transcription and require caspase activation. Mol Cell Biol 17:6348–6358
Rickers A, Peters N, Badock V, Beyaert R, Vandenabeele P, Dorken B, Bommert K (1999) Cleavage of transcription factor SP1 by caspases during anti-IgM-induced B-cell apoptosis. Eur J Biochem 261:269–274
Wang YT, Yang WB, Chang WC, Hung JJ (2011) Interplay of posttranslational modifications in Sp1 mediates Sp1 stability during cell cycle progression. J Mol Biol 414:1–14
Dynan WS, Saffer JD, Lee WS, Tjian R (1985) Transcription factor Sp1 recognizes promoter sequences from the monkey genome that are simian virus 40 promoter. Proc Natl Acad Sci USA 82:4915–4919
Enomoto M, Bunge MB, Tsoulfas P (2013) A multifunctional neurotrophin with reduced affinity to p75NTR enhances transplanted Schwann cell survival and axon growth after spinal cord injury. Exp Neurol 248:170–182
Yang J, Liu X, Bhalla K, Kim CN, Ibrado AM, Cai J, Peng TI, Jones DP, Wang X (1997) Prevention of apoptosis by Bcl-2: release of cytochrome c from mitochondria blocked. Science 275:1129–1132
Yang XH, Sladek TL, Liu X, Butler BR, Froelich CJ, Thor AD (2001) Reconstitution of caspase 3 sensitizes MCF-7 breast cancer cells to doxorubicin- and etoposide-induced apoptosis. Cancer Res 61:348–354
Xue LY, Chiu SM, Oleinick NL (2001) Photodynamic therapy-induced death of MCF-7 human breast cancer cells: a role for caspase-3 in the late steps of apoptosis but not for the critical lethal event. Exp Cell Res. 263;145–155
Blanc C, Deveraux QL, Krajewski S, Janicke RU, Porter AG, Reed JC, Jaggi R, Marti A (2000) Caspase-3 is essential for procaspase-9 processing and cisplatin-induced apoptosis of MCF-7 breast cancer cells. Cancer Res 60:4386–4390
Kurokawa H, Nishio K, Fukumoto H, Tomonari A, Suzuki T, Saijo N (1999) Alteration of caspase-3 (CPP32/Yama/apopain) in wild-type MCF-7, breast cancer cells. Oncol Rep 6:33–37
Astrinidis A, Kim J, Kelly CM, Olofsson BA, Torabi B, Sorokina EM, Azizkhan-Clifford J (2010) The transcription factor SP1 regulates centriole function and chromosomal stability through a functional interaction with the mammalian target of rapamycin/raptor complex, Genes Chromosomes Cancer 49:282–297
Dynan WS, Sazer S, Tjian R, Schimke RT (1986) Transcription factor Sp1 recognizes a DNA sequence in the mouse dihydrofolate reductase promoter. Nature 319:246–248
Chowdhury I, Tharakan B, Bhat GK (2008) Caspases: an update. Comp Biochem Phys B 151:10–27
Earnshaw WC, Martins LM, Kaufmann SH (1999) Mammalian caspases: structure, activation, substrates, and functions during apoptosis. Annu Rev Biochem 68:383–424
Nicholson DW (1999) Caspase structure, proteolytic substrates, and function during apoptotic cell death. Cell Death Differentiation 6:1028–1042
Mahrus S, Trinidad JC, Barkan DT, Sali A, Burlingame AL, Wells JA (2008) Global sequencing of proteolytic cleavage sites in apoptosis by specific labeling of protein N termini. Cell 134:866–876
Courey AJ, Tjian R (1988) Analysis of Sp1 in vivo reveals multiple transcriptional domains, including a novel glutamine-rich activation motif. Cell 55:887–898
Pascal E, Tjian R (1991) Different activation domains of Sp1 govern formation of multimers and mediate transcriptional synergism. Genes Dev 5:1646–1656
Kadonaga JT, Courey AJ, Ladika J, Tjian R (1988) Distinct regions of Sp1 modulate DNA binding and transcriptional activation. Science 242:1566–1570
Wang J, Kang M, Qin YT, Wei ZX, Xiao JJ, Wang RS (2015) Sp1 is over-expressed in nasopharyngeal cancer and is a poor prognostic indicator for patients receiving radiotherapy. Int J Clin Exp Pathol 8:6936–6943
Hsu TI, Wang MC, Chen SY, Yeh YM, Su WC, Chang WC, Hung JJ (2012) Sp1 expression regulates lung tumor progression. Oncogene 31:3973–3988
Guan H, Cai J, Zhang N, Wu J, Yuan J, Li J, Li M (2012) Sp1 is upregulated in human glioma, promotes MMP-2-mediated cell invasion and predicts poor clinical outcome. Int J Cancer 130:593–601
Jiang NY, Woda BA, Banner BF, Whalen GF, Dresser KA, Lu D (2008) Sp1, a new biomarker that identifies a subset of aggressive pancreatic ductal adenocarcinoma. Cancer Epidemiol Biomark Prev 17:1648–1652
Hanahan D, Weinberg RA (2011) Hallmarks of cancer: the next generation. Cell 144:646–674
Beishline K, Azizkhan-Clifford J (2015) Sp1 and the ‘hallmarks of cancer’. FEBS J 282:224–258
Kaina B, Christmann M, Naumann S, Roos WP (2007) MGMT: key node in the battle against genotoxicity, carcinogenicity and apoptosis induced by alkylating agents. DNA Repair 6:1079–1099
Aas T, Borresen AL, Geisler S, Smith-Sorensen B, Johnsen H, Varhaug JE, Akslen LA, Lonning PE (1996) Specific P53 mutations are associated with de novo resistance to doxorubicin in breast cancer patients. Nat Med 2:811–814
Asanuma K, Tsuji N, Endoh T, Yagihashi A, Watanabe N (2004) Survivin enhances Fas ligand expression via up-regulation of specificity protein 1-mediated gene transcription in colon cancer cells. J Immunol 172:3922–3929
Wu J, Ling X, Pan D, Apontes P, Song L, Liang P, Altieri DC, Beerman T, Li F (2005) Molecular mechanism of inhibition of survivin transcription by the GC-rich sequence-selective DNA binding antitumor agent, hedamycin: evidence of survivin down-regulation associated with drug sensitivity. J Biol Chem 280:9745–9751
Iwahori S, Yasui Y, Kudoh A, Sato Y, Nakayama S, Murata T, Isomura H, Tsurumi T (2008) Identification of phosphorylation sites on transcription factor Sp1 in response to DNA damage and its accumulation at damaged sites. Cell Signal 20:1795–1803
Chen F, Arseven OK, Cryns VL (2004) Proteolysis of the mismatch repair protein MLH1 by caspase-3 promotes DNA damage-induced apoptosis. J Biol Chem 279:27542–27548
Zhang J, Powell SN (2005) The role of the BRCA1 tumor suppressor in DNA double-strand break repair. Mol Cancer Res 3:531–539
Cressman VL, Backlund DC, Avrutskaya AV, Leadon SA, Godfrey V, Koller BH (2006) Growth retardation, DNA repair defects, and lack of spermatogenesis in BRCA1-deficient mice. Mol Cell Biol 26:9571
Reid LJ, Shakya R, Modi AP, Lokshin M, Cheng JT, Jasin M, Baer R, Ludwig T (2008) E3 ligase activity of BRCA1 is not essential for mammalian cell viability or homology-directed repair of double-strand DNA breaks. Proc Natl Acad Sci USA. 105:20876–20881
Martin SA, Ouchi T (2005) BRCA1 phosphorylation regulates caspase-3 activation in UV-induced apoptosis. Cancer Res 65:10657–10662
Gu M, Li H, Shen C, Wu L, Liu W, Miao L, Zheng C (2010) Cloning and characterization of a new BRCA1 variant: a role for BRCT domains in apoptosis. Cancer Lett 295:205–213
Su F, Smilenov LB, Ludwig T, Zhou L, Zhu J, Zhou G, Hall EJ (2010) Hemizygosity for Atm and Brca1 influence the balance between cell transformation and apoptosis. Radiat Oncol 5:15
Okita N, Yoshimura M, Watanabe K, Minato S, Kudo Y, Higami Y, Tanuma S (2013) CHK1 cleavage in programmed cell death is intricately regulated by both caspase and non-caspase family proteases. Biochim Biophys Acta 1830:2204–2213
Zhan Q, Jin S, Ng B, Plisket J, Shangary S, Rathi A, Brown KD, Baskaran R (2002) Caspase-3 mediated cleavage of BRCA1 during UV-induced apoptosis. Oncogene 21:5335–5345
Matsuura K, Wakasugi M, Yamashita K, Matsunaga T (2008) Cleavage-mediated activation of Chk1 during apoptosis. J Biol Chem 283:25485–25491
Solier S, Pommier Y (2011) MDC1 cleavage by caspase-3: a novel mechanism for inactivating the DNA damage response during apoptosis. Cancer Res 71:906–913
Huang Y, Nakada S, Ishiko T, Utsugisawa T, Datta R, Kharbanda S, Yoshida K, Talanian RV, Weichselbaum R, Kufe D, Yuan ZM (1999) Role for caspase-mediated cleavage of Rad51 in induction of apoptosis by DNA damage. Mol Cell Biol 19:2986–2997
Sakai W, Sugasawa K (2014) FANCD2 is a target for caspase 3 during DNA damage-induced apoptosis. FEBS Lett 588:3778–3785
Goodarzi AA, Noon AT, Deckbar D, Ziv Y, Shiloh Y, Lobrich M, Jeggo PA (2008) ATM signaling facilitates repair1 of DNA double-strand breaks associated with heterochromatin. Mol Cell 31:167–77
Morrison C, Sonoda E, Takao N, Shinohara A, Yamamoto K, Takeda S (2000) The controlling role of ATM in homologous recombinational repair of DNA damage. EMBO J 19:463–471
Kuhne M, Riballo E, Rief N, Rothkamm K, Jeggo PA, Lobrich M (2004) A double-strand break repair defect in ATM-deficient cells contributes to radiosensitivity. Cancer Res 64:500–508
Carlessi L, De Filippis L, Lecis D, Vescovi A, Delia D (2009) DNA-damage response, survival and differentiation in vitro of a human neural stem cell line in relation to ATM expression. Cell Death Differ 16:795–806
Lai JM, Chang JT, Wen CL, Hsu SL (2009) Emodin induces a reactive oxygen species-dependent and ATM-p53-Bax mediated cytotoxicity in lung cancer cells. Eur J Pharmacol 623:1–9
Chong MJ, Murray MR, Gosink EC, Russell HR, Srinivasan A, Kapsetaki M, Korsmeyer SJ, McKinnon PJ (2000) Atm and bax cooperate in ionizing radiation-induced apoptosis in the central nervous system. Proc Natl Acad Sci USA 97:889–894
Demontis S, Rigo C, Piccinin S, Mizzau M, Sonego M, Fabris M, Brancolini C, Maestro R (2006) Twist is substrate for caspase cleavage and proteasome-mediated degradation. Cell Death Diff 13:335–345
Borges HL, Bird J, Wasson K, Cardiff RD, Varki N, Eckmann L, Wang JY (2005) Tumor promotion by caspase-resistant retinoblastoma protein. Proc Natl Acad Sci USA 102:15587–15592
Puissant A, Fenouille N, Robert G, Jacquel A, Bassil CF, Colosetti P, Luciano F, Auberger P (2014) A new posttranslational regulation of REDD1/DDIT4 through cleavage by caspase 3 modifies its cellular function. Cell Death Dis 5:e1349
Yeh TC, Bratton SB (2013) Caspase-dependent regulation of the ubiquitin-proteasome system through direct substrate targeting. Proc Natl Acad Sci USA 110:14284–14289
Funding
This study was funded by College of Medicine, Drexel University.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Torabi, B., Flashner, S., Beishline, K. et al. Caspase cleavage of transcription factor Sp1 enhances apoptosis. Apoptosis 23, 65–78 (2018). https://doi.org/10.1007/s10495-017-1437-4
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10495-017-1437-4