Abstract
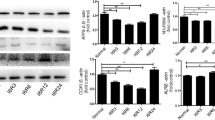
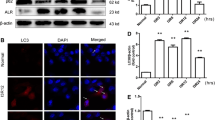
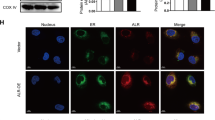
Oxidative stress plays an important role in cellular destruction. Augmenter of liver regeneration (ALR) is an anti-apoptotic factor that is expressed in all mammalian cells and functions as an anti-oxidant by stimulating the expression of a secretory isoform of clusterin and inhibiting reactive oxygen species (ROS) generation. Previous work from our group showed that ALR expression is upregulated in acute kidney injury (AKI) rats, and recombinant human ALR reduces tubular injury. In the present study, we used small interfering RNA (siRNA) silencing of ALR to examine its role in H2O2 induced mitochondrial injury and apoptosis. Knockdown of ALR increased ROS levels, reduced mitochondrial membrane potential, and increased the release of mitochondrial proteins and the rate of apoptosis in response to H2O2. In addition, the ratio of Bax/Bcl-2 was increased in siRNA/ALR groups treated with H2O2. These data confirm the protective role of ALR against oxidative stress-induced mitochondrial injury and suggest a potential mechanism underlying the protective role of ALR in AKI.








Similar content being viewed by others
Abbreviations
- ALR:
-
Augmenter of liver regeneration
- AKI:
-
Acute kidney injury
- ROS:
-
Reactive oxygen species
- MMP:
-
Mitochondrial membrane potential
- DCFH-DA:
-
2-,7-Dichlorofluorescin diacetate
- DAPI:
-
4′,6-Diamidino-2-phenylindole
- H2O2 :
-
Hydrogen peroxide
- MTS:
-
3-(4,5-Dimethylthiazol-2-yl)-5-(3-carboxymethoxyphenyl)-2-(4-sulfophenyl)-2H-tetrazolium, inner salt
- COX IV:
-
Cytochrome c oxidase IV
References
Linkermann A, Chen G, Dong G, Kunzendorf U, Krautwald S, Dong Z (2014) Regulated cell death in AKI. J Am Soc Nephrol. doi:10.1681/ASN030262
Pathak E, MacMillan-Crow LA, Mayeux PR (2012) Role of mitochondrial oxidants in an in vitro model of sepsis-induced renal injury. J Pharmacol Exp Ther 340:192–201
Bonventre JV, Yang L (2011) Cellular pathophysiology of ischemic acute kidney injury. J Clin Invest 121:4210–4221
Chan PH (1996) Role of oxidants in ischemic brain damage. Stroke 27(6):1124–1129
Zhang L, Zhao B, Yew DT, Kusiak JW (1997) Processing of Alzheimer’s amyloid precursor protein during H2O2-induced apoptosis in human neuronal cells. Biochem Biophys Res Commun 235(3):845–848
Yu M, Wang J, Li W et al (2008) Proteomic screen defines the hepatocyte nuclear factor 1alpha-binding partners and identifies HMGB1 as a new cofactor of HNF1alpha. Nucleic Acids Res 36:1209–1219
Liu X, Kim CN, Yang J, Jemmerson R (1996) Induction of apoptotic program in cell free extracts requirement for dATP and cytochrome c. Cell 86:147–157
Richter C (1998) Oxidative stress, mitochondria, and apoptosis. Restor Neurol Neurosci 12:59–62
Hagiya M, Francavilla A, Polimeno L, Ihara I, Sakai H, Seki T (1994) Cloning and sequence analysis of the rat augmenter of liver regeneration (ALR) gene expression of biologically active recombinant ALR and demonstration of tissue distribution. Proc Natl Acad Sci USA 91:8142–8146
Kallergi E, Kalef-Ezra E, Karagouni-Dalakoura K, Tokatlidis K (2014) Common players in mitochondria biogenesis and neuronal protection against stress-induced apoptosis. Neurochem Res 39:546–555
Liao XH, Zhang L, Liu Q, Sun H, Peng CM, Guo H (2010) Augmenter of liver regeneration protects kidneys from ischaemia/reperfusion injury in rats. Nephrol Dial Transplant 25:2921–2929
Liao XH, Zhang L, Liu Q, Sun H, Peng CM, Guo H (2012) Augmenter of liver regeneration attenuates tubular cell apoptosis in acute kidney injury in rats:the possible mechanisms. Ren Fail 34:590–599
Farrell SR, Thorpe C (2005) Augmenter of liver regeneration a flavin dependent sulfhydryl oxidase with cytochrome c reductase activity. Biochemistry 44:1532–1541
Gandhi CR (2012) Augmenter of liver regeneration. Fibrogenesis Tissue Repair 5:10
Banci L, Bertini I, Cefaro C (2010) Molecular chaperone function of Mia40 triggers consecutive induced folding steps of the substrate in mitochondrial protein import. Proc Natl Acad Sci 107:20190–20195
Bihlmaier K, Mesecke N, Terziyska N, Bien M, Hell K, Herrmann JM (2007) The disulfide relay system of mitochondria is connected to the respiratory chain. J Cell Biol 179:389–395
Giorda R, Hagiya M, Seki T, Shimonishi M, HarumiSakai A (1996) Analysis of the structure and expression of the augmenter of liver regeneration (ALR) gene. Mol Med 2:97–108
Polimeno L, Pesetti B, Lisowsky T et al (2009) Protective effect of augmenter of liver regeneration on hydrogen peroxide-induced apoptosis in SH-SY5Y human neuroblastoma cells. Free Radic Res 43:865–875
Polimeno L, Pesetti B, De Santis F et al (2012) Decreased expression of the augmenter of liver regeneration results in increased apoptosis and oxidative damage in human-derived glioma cells. Cell Death Dis 3:e289
Polimeno L, Pesetti B, Annoscia E (2011) Alrp, a survival factor that controls the apoptotic process of regenerating liver after partial hepatectomy in rats. Free Radic Res 45:534–549
Shen Y, Liu Q, Sun H, Li X, Wang N, Guo H (2013) Protective effect of augmenter of liver regeneration on vincristine-induced cell death in Jurkat T leukemia cells. Int Immunopharmacol 17:162–167
Ilowski M, Kleespies A, de Toni EN et al (2011) Augmenter of liver regeneration (ALR) protects human hepatocytes against apoptosis. Biochem Biophys Res Commun 404:148–152
Satoh T, Sakai N, Enokido Y, Uchiyama Y, Hatanaka H (1996) Free radical-independent protection by nerve growth factor and Bcl-2 of PC12 cells from hydrogen peroxide-triggered apoptosis. J Biochem 120:540–546
Zhang L, Yu H, Sun Y et al (2007) Protective effects of salidroside on ‘-induced apoptosis in SH-SY5Y human neuroblastoma cells. Eur J Pharmacol 564:18–25
Cao Y, Fu YL, Yu M et al (2009) Human augmenter of liver regeneration is important for hepatoma cell viability and resistance to radiation-induced oxidative stress. Free Radic Biol Med 47:1057–1066
Hahn P, Lindsten T, Lyubarsky A et al (2004) Deficiency of Bax and Bak protects photoreceptors from light damage in vivo. Cell Death Differ 11:1192–1197
Bae EH, Cho S, Joo SY et al (2011) 4-Hydroxy-2-hexenal-induced apoptosis in human renal proximal tubular epithelial cells. Nephrol Dial Transplant 26:3866–3873
Basile DP, Liapis H, Hammerman MR (1997) Expression of bcl-2 and bax in regenerating rat renal tubules following ischemic injury. Am J Physiol 272:F640–F647
Kluck R, Bossy-Wetzel E, Green D, Newmeyer D (1997) The release of cytochrome c from mitochondria: a primary site for Bcl-2 regulation of apoptosis. Science 275:1132–1136
Gross A, Yin XM, Wang K et al (1999) Caspase cleaved BID targets mitochondria and is required for cytochrome c release, while BCL-XL prevents this release but not tumor necrosis factor-R1/Fas death. J Biol Chem 274:1156–1163
Lange H, Lisowsky T, Gerber J (2001) An essential function of the mitochondrial sulfhydryl oxidase Erv1pALR in the maturation of cytosolic FeS proteins. EMBO Rep 2:715–720
Polimeno L, Capuano F, Marangi L, Margiotta M, Lisowsky T, Ierardi E (2000) The augmenter of liver regeneration induces mitochondrial gene expression in rat liver and enhances oxidative phosphorylation capacity of liver mitochondria. Dig Liver Dis 32:510–517
Hell K (2008) The Erv1-Mia40 disulfide relay system in the intermembrane space of mitochondria. Biochim Biophys Acta 1783:601–609
van Gurp M, Festjens N, van Loo G, Saelens X, Vandenabeele P (2003) Mitochondrial intermembrane proteins in cell death. Biochem Biophys Res Commun 304:487–497
Acknowledgments
This work was supported by a Grant from the National Natural Science Foundation of China (39071364) and clinical medicine research special funds of the Chinese medical association (14050370574), and Grants from medical scientific research Projects of Chongqing health and family planning commission (20142031).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Xia, N., Yan, Ry., Liu, Q. et al. Augmenter of liver regeneration plays a protective role against hydrogen peroxide-induced oxidative stress in renal proximal tubule cells. Apoptosis 20, 423–432 (2015). https://doi.org/10.1007/s10495-015-1096-2
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10495-015-1096-2