Abstract
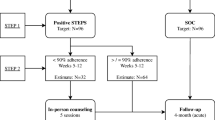
To assess the efficacy of an education- and strengths-based counselling programme to promote antiretroviral therapy (ART) adherence in a cohort of HIV-infected individuals with high prevalence of injection drug use in Estonia. Parallel-group randomized (1:1) controlled trial (RCT). Adults receiving ART in two clinics were followed for 12 months. The trial compared: (i) an intervention (three sessions) incorporated into routine clinic visits, providing education about HIV, ART, the role of adherence, and tailoring regimen to daily routines using problem-solving skills to address adherence barriers versus (ii) usual care (control). Primary and secondary outcomes were self-reported ART adherence (3-day recall) and viral load (respectively). 519 patients were randomized and 82% completed the study. Recent optimal ART adherence (3-day recall ≥95%) was reported by 75.6% in the intervention group and 72.9% of controls at baseline and 76.7% and 67.5%, respectively, at 12 months (RR 1.14, 95% CI 1.00–1.28; adjusted RR 1.13, 95% CI 1.00–1.27). There was no difference in the proportion of patients with undetectable viral load. At 12 months the intervention group reported significantly higher perceptions of ART necessity versus ART concerns [mean ART necessity-concerns differential: intervention group 1.32 (SD 1.22) vs control group 1.08 (SD 1.12); p = 0.048]. All-cause mortality among study participants was 27.7 per 1000 person years (95% CI 15.6–44.8). A brief, clinic-based adherence intervention alone may assist with adherence but lacks impact on viral load at 12 months.

Similar content being viewed by others
References
Langebeek N, Gisolf EH, Reiss P, et al. Predictors and correlates of adherence to combination antiretroviral therapy(ART) for chronic HIV infection: a meta-analysis. BMC Med. 2014;12:142.
Mills EJ, Nachega JB, Buchan I, et al. Adherence to antiretroviral therapy in sub-Saharan Africa and North America: a meta-analysis. JAMA. 2006;296(6):679–90.
European Centre for Disease Prevention and Control, WHO Regional Office for Europe. HIV/AIDS surveillance in Europe 2014. Stockholm: ECDC. http://ecdc.europa.eu/en/publications/Publications/hiv-aids-surveillance-in-Europe-2014.pdf (2015).
Soodla P, Rajasaar H, Avi R, et al. Design and structure of the Estonian HIV Cohort Study (E-HIV). Infect Dis (Lond). 2015;47(11):768–75.
Lõhmus L, Lemsalu L, Rüütel K, Laisaar KT, Uusküla A. Infektsionisti külastavate HIV-i nakatunud inimeste tervis [Health indicators of PLHIV in HIV care with infectious disease doctors]. National Institute for Health Development, Tallinn, Estonia. https://intra.tai.ee/images/prints/documents/142088556524_Infektsionisti_kylastavate_HIVi_nakatunud_inimeste_tervis.pdf (2014).
Amico KR, McMahan V, Goicochea P, et al. Developing an innovative approach to adherence counseling and assessment in a pre-exposure prophylaxis (PrEP) trial: next step counseling and neutral assessment in the iPrEX study. In: 5th International Conference on Treatment Adherence, Miami USA, May 23–25, 2010.
Grant RM, Lama JR, Anderson PL, et al. Preexposure chemoprophylaxis for HIV prevention in men who have sex with men. N Engl J Med. 2010;363:2587–99.
Fisher JD, Amico KR, Fisher WA, Harman JJ. The information-motivation-behavioral skills model of antiretroviral adherence and its applications. Curr HIV/AIDS Rep. 2008;5:193–203.
Munro S, Lewin S, Swart T, Volmink J. A review of health behaviour theories: how useful are these for developing interventions to promote long-term medication adherence for TB and HIV/AIDS? BMC Public Health. 2007;7:104.
Laisaar KT, Uusküla A, Sharma A, DeHovitz JA, Amico KR. Developing an adherence support intervention for patients on antiretroviral therapy in the context of the recent IDU-driven HIV/AIDS epidemic in Estonia. AIDS Care. 2013;25(7):863–73.
Estonian Society of Infectious Diseases 2013. Eesti Infektsioonhaiguste seltsi juhend “HIV patsiendi jälgimine [The guideline for monitoring of HIV infected patient]. http://www.esid.ee/cms/tl_files/failid/failid/HIV%20patsiendi%20jaelgimine.pdf.
Chesney MA, Ickovics JR, Chambers DB, et al. Self-reported adherence to antiretroviral medications among participants in HIV clinical trials: the AACTG adherence instruments. Patient Care Committee & Adherence Working Group of the Outcomes Committee of the Adult AIDS Clinical Trials Group (AACTG). AIDS Care. 2000;12(3):255–66.
Mannheimer S, Thackeray L, Huppler Hullsiek K, et al. A randomized comparison of two instruments for measuring self-reported antiretroviral adherence. AIDS Care. 2008;20(2):161–9.
Simoni JM, Kurth AE, Pearson CR, Pantalone DW, Merrill JO, Frick PA. Self-report measures of antiretroviral therapy adherence: a review with recommendations for HIV research and clinical management. AIDS Behav. 2006;10(3):227–45.
Uusküla A, Laisaar KT, Raag M, et al. Antiretroviral therapy (ART) adherence and correlates to nonadherence among people on ART in Estonia. AIDS Care. 2012;24(12):1470–9.
Ware JE, Snow KK, Kosinski M, Gandek B. SF-36 health survey manual & interpretation guide. Boston: New England Medical Center; 1993.
Ewing J, Rouse BA. Identifying the hidden alcoholic. Paper presented at: the 29th International Congress on Alcohol and Drug Dependence, 1970; Sydney Australia, February 3. 1970.
Horne R, Weinman J, Hankins M. The Beliefs about Medicines Questionnaire: the development and evaluation of a new method for assessing the cognitive representation of medication. Psychology and Health. 1999;14:1–24.
Oppenheim AN. Questionnaire design, interviewing and attitude measurement. London: Pinter; 1992.
Zou G. A modified Poisson regression approach to prospective studies with binary data. Am J Epid. 2004;159(7):702–6.
White IR, Royston P, Wood AM. Multiple imputation using chained equations: issues and guidance for practice. Stat Med. 2011;30(4):377–99.
R Core Team. R: a language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria. https://www.R-project.org/ (2015).
van Buuren S, Groothuis-Oudshoorn Karin. Mice: multivariate imputation by chained equations in R. J Stat Softw. 2011;45(3):1–67.
Mbuagbaw L, Sivaramalingam B, Navarro T, et al. Patient adherence review (PAR) team. Interventions for enhancing adherence to antiretroviral therapy (ART): a systematic review of high quality studies. AIDS Patient Care STDS. 2015;29(5):248–66.
de Bruin M, Viechtbauer W, Schaalma HP, Kok G, Abraham C, Hospers HJ. Standard care impact on effects of highly active antiretroviral therapy adherence interventions: a meta-analysis of randomized controlled trials. Arch Intern Med. 2010;170(3):240–50.
Charania MR, Marshall KJ, Lyles CM, et al. Identification of evidence-based interventions for promoting HIV medication adherence: findings from a systematic review of U.S.-based studies, 1996–2011. AIDS Behav. 2014;9:646–60.
Amico KR, Harman JJ, Johnson BT. Efficacy of antiretroviral therapy adherence interventions: a research synthesis of trials, 1996 to 2004. JAIDS. 2006;41(3):285–97.
Simoni JM, Pearson CR, Pantalone DW, Marks G, Crepaz N. Efficacy of interventions in improving highly active antiretroviral therapy adherence and HIV-1 RNA viral load, A meta-analytic review of randomized controlled trials. JAIDS. 2006;43(Suppl1):S23–35.
Rueda S, Park-Wyllie LY, Bayoumi AM, et al. Patient support and education for promoting adherence to highly active antiretroviral therapy for HIV/AIDS. Cochrane Database Syst Rev. 2006;3:CD001442.
Mathes T, Pieper D, Antoine SL, Eikermann M. Adherence-enhancing interventions for highly active antiretroviral therapy in HIV-infected patients. A systematic review. HIV Med. 2013;14:583–95.
CDC. Complete listing of medication adherence evidence-based behavioral interventions. http://www.cdc.gov/hiv/research/interventionresearch/compendium/ma/complete.html (2016).
Kurth AE, Spielberg F, Cleland CM, et al. Computerized counseling reduces HIV-1 viral load and sexual transmission risk: findings from a randomized controlled trial. J Acquir Immune Defic Syndr. 2014;65(5):611–20.
Richardson JL, Milam J, McCutchan A, et al. Effect of brief safer-sex counseling by medical providers to HIV-1 seropositive patients: a multi-clinic assessment. AIDS. 2004;18(8):1179–86.
Kalichman SC, Hudd K, Diberto G. Operational fidelity to an evidence-based HIV prevention intervention for people living with HIV/AIDS. J Prim Prev. 2010;31(4):235–45.
CampBinford M, Kahana SY, Altice FL. A systematic review of antiretroviral adherence interventions for HIV-infected people who use drugs. Curr HIV/AIDS Rep. 2012;9(4):287–312.
Nosyk B, Min JE, Colley G, et al. The causal effect of opioid substitution treatment on HAART medication refill adherence. AIDS. 2015;29(8):965–73.
Luborsky L, Singer B, Luborsky L. Comparative studies of psychotherapies is it true that “Everyone Has Won and All Must Have Prizes”? Arch Gen Psychiatry. 1975;32(8):995–1008.
Metsch LR, Feaster DJ, Gooden L, et al. Effect of risk-reduction counseling with rapid HIV testing on risk of acquiring sexually transmitted infections The AWARE Randomized Clinical Trial. JAMA. 2013;310(16):1701–10.
National Collaborating Centre for Primary Care (UK). Medicines adherence: involving patients in decisions about prescribed medicines and supporting adherence. London: Royal College of General Practitioners (UK); (NICE Clinical Guidelines, No. 76:7, Assessment of adherence). http://www.ncbi.nlm.nih.gov/books/NBK55447/ (2009).
Bangsberg DR. Less than 95% adherence to nonnucleoside reverse-transcriptase inhibitor therapy can lead to viral suppression. Clin Infect Dis. 2006;43(7):939–41.
Shuter J, Sarlo JA, Kanmaz TJ, Rode RA, Zingman BS. HIV-infected patients receiving lopinavir/ritonavir-based antiretroviral therapy achieve high rates of virologic suppression despite adherence rates less than 95%. J Acquir Immune Defic Syndr. 2007;45(1):4–8.
Yurin OG. Principal approaches to HIV treatment in the Russian Federation. Presentation at the 5th Eastern Europe and Central Asia AIDS Conference, Moscow Russian Federation, March 23–25, 2016.
Abishev AT. Applying modern treatment strategies in Kazakhstan. Presentation at the 5th Eastern Europe and Central Asia AIDS Conference, Moscow Russian Federation, March 23-25, 2016.
Dettori JR. Loss to follow-up. Evid-Based Spine-Care J. 2011;2(1):7–10.
Antiretroviral Therapy Cohort Collaboration (ART-CC), May MT, Gill MJ, Wittkop L, et al. Mortality of treated HIV-1 positive individuals according to viral subtype in Europe and Canada: collaborative cohort analysis. IDS. 2016;30(3):503–13.
Smith CJ, Ryom L, Weber R, et al. Trends in underlying causes of death in people with HIV from 1999 to 2011 (D:A:D): a multicohort collaboration. Lancet. 2014;384(9939):241–8.
Statistics Estonia. http://pub.stat.ee/px-web.2001/dialog/statfile1.asp.
Acknowledgements
This work was supported by the European Union through the European Regional Development Fund within the research project „Bridging the gap in knowledge and practice of prevention and care for HIV in Estonia” (HIV-BRIDGE), and by Grant # TARTH15017I from the Estonian Ministry of Education and Research.
Collaborators for the HIV-BRIDGE Study Group
Dr Kai Zilmer, dr Tiiu Aug, dr Kerstin Kase, dr Helen Mülle, dr Kersti Kink, Merike Meindok, Irina Milaševitš, Irina Sorokina, Olev Lumiste ja Liudmila Ponomarenko at the West Tallinn Central Hospital; and dr Jelena Šmidt, dr Svetlana Semjonova, dr Natalia Nikitina, dr Julia Kogan, Anne Junolainen, Marjana Golubnitšija ja Tatiana Fishkova at the Ida-Viru Central Hospital (Kohtla-Järve).
Author information
Authors and Affiliations
Consortia
Contributions
AU, KTL and RA had the original idea for the article and outlined the design. KTL and KR were responsible for data collection, LL for data management; MR for data analysis. AU wrote the first draft of the manuscript to which all other authors contributed.
Corresponding author
Ethics declarations
Conflict of interest
The authors report no conflict of interests.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Additional information
Collaborators of the HIV-BRIDGE Study Group is listed in “Acknowledgment”.
Rights and permissions
About this article
Cite this article
Uusküla, A., Laisaar, KT., Raag, M. et al. Effects of Counselling on Adherence to Antiretroviral Treatment Among People with HIV in Estonia: A Randomized Controlled Trial. AIDS Behav 22, 224–233 (2018). https://doi.org/10.1007/s10461-017-1859-6
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10461-017-1859-6