Abstract
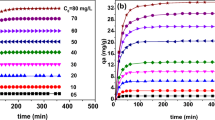
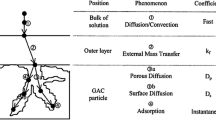
The aim of the present study was to clarify the physical meaning of the parameters used in fractal kinetic and generalised isotherm models of Brouers–Sotolongo. For this purpose, adsorption of methylene blue (MB) and methyl orange (MO) onto four activated carbons (ACs) was carried out. These ACs were characterised in terms of composition, surface area, pore volumes and pore size distributions, carbon nanotexture and surface chemistry. Adsorption isotherms were carried out at 25 °C, and at pH 2.5 and 8 for MO and MB, respectively, and fitted with Langmuir, Freundlich, Jovanovich, Hill–Sips, Brouers–Sotolongo, Brouers–Gaspard and General Brouers–Sotolongo (GBS) models. Adsorption kinetics were fitted by traditional pseudo-first and pseudo-second order models and compared to the Brouers–Sotolongo (BSf) fractal kinetic model. GBS and BSf were found to be the best models describing adsorption isotherms and kinetics, respectively. This finding suggests that MB and MO adsorption is probabilistic and closely correlated to the heterogeneous character of the adsorbent surface. Moreover, BSf and GBS parameters were correlated with surface area and amount of surface functional groups. In particular, higher surface area and amount of functional groups respectively decreased and increased the constants τc and α of the BSf stochastic model.








Similar content being viewed by others
References
Acosta, R., Fierro, V., Martinez de Yuso, A., Nabarlatz, D., Celzard, A.: Tetracycline adsorption onto activated carbons produced by KOH activation of tyre pyrolysis char. Chemosphere 149, 168–176 (2016). https://doi.org/10.1016/j.chemosphere.2016.01.093
Altenor, S., Ncibi, M.C., Emmanuel, E., Gaspard, S.: Textural characteristics, physiochemical properties and adsorption efficiencies of Caribbean alga Turbinaria turbinata and its derived carbonaceous materials for water treatment application. Biochem. Eng. J. 67, 35–44 (2012). https://doi.org/10.1016/j.bej.2012.05.008
Bandosz, T.J., Jagiello, J., Contescu, C., Schwarz, J.A.: Characterization of the surfaces of activated carbons in terms of their acidity constant distributions. Carbon 31(7), 1193–1202 (1993). https://doi.org/10.1016/0008-6223(93)90072-I
Bello, O.S., Bello, I.A., Adegoke, K.A.: Adsorption of dyes using different types of sand: a review. S. Afr. J. Chem. 66, 00–00 (2013)
Ben Hamissa, A.M., Brouers, F., Borhane, M., Seffen, M.: Adsorption of textile dyes using agave Americana (L.) fibres: equilibrium and kinetics modelling. Adsor. Sci. Technol. 25(5), 311–325 (2007). https://doi.org/10.1260/026361707783432533
Ben Hamissa, A.M., Brouers, F., Ncibi, M.C., Seffen, M.: Kinetic modeling study on methylene blue sorption onto agave Americana fibers: fractal kinetics and regeneration studies. Sep. Sci. Technol. 48(18), 2834–2842 (2013). https://doi.org/10.1080/01496395.2013.809104
Benaddi, H., Bandosz, T.J., Jagiello, J., Schwarz, J.A., Rouzaud, J.N., Legras, D., Béguin, F.: Surface functionality and porosity of activated carbons obtained from chemical activation of wood. Carbon 38(5), 669–674 (2000). https://doi.org/10.1016/S0008-6223(99)00134-7
Bouhamed, F., Elouear, Z., Bouzid, J., Ouddane, B.: Multi-component adsorption of copper, nickel and zinc from aqueous solutions onto activated carbon prepared from date stones. Environ. Sci. Pollut. Res. 23(16), 15801–15806 (2016). https://doi.org/10.1007/s11356-015-4400-3
Braghiroli, F.L., Fierro, V., Parmentier, J., Vidal, L., Gadonneix, P., Celzard, A.: Hydrothermal carbons produced from tannin by modification of the reaction medium: addition of H+ and Ag+. Ind. Crops Prod. 77, 364–374 (2015). https://doi.org/10.1016/j.indcrop.2015.09.010
Brouers, F.: The fractal (BSf) kinetics equation and its approximations. J. Mod. Phys. 5(16), 1594 (2014a). https://doi.org/10.4236/jmp.2014.516160
Brouers, F.: Statistical foundation of empirical isotherms. Open J. Stat. 4(09), 687–701 (2014b). https://doi.org/10.4236/ojs.2014.49064
Brouers, F., Al-Musawi, T.J.: On the optimal use of isotherm models for the characterization of biosorption of lead onto algae. J. Mol. Liq. 212, 46–51 (2015). https://doi.org/10.1016/j.molliq.2015.08.054
Brouers, F., Sotolongo-Costa, O.: Generalized fractal kinetics in complex systems (application to biophysics and biotechnology). Physica A 368(1), 165–175 (2006). https://doi.org/10.1016/j.physa.2005.12.062
Brouers, F., Sotolongo-Costa, O., Weron, K.: Burr, Lévy: Tsallis. Physica A 344(3), 409–416 (2004). https://doi.org/10.1016/j.physa.2004.06.008
Brouers, F., Sotolongo, O., Marquez, F., Pirard, J.P.: Microporous and heterogeneous surface adsorption isotherms arising from Levy distributions. Physica A 349(1–2), 271–282 (2005). https://doi.org/10.1016/j.physa.2004.10.032
Centeno, T.A., Stoeckli, F.: The assessment of surface areas in porous carbons by two model-independent techniques, the DR equation and DFT. Carbon 9(48), 2478–2486 (2010)
Choi, Y.-K., Cho, M.-H., Kim, J.-S.: Air gasification of dried sewage sludge in a two-stage gasifier. Part 4: application of additives including Ni-impregnated activated carbon for the production of a tar-free and H2-rich producer gas with a low NH3 content. Int. J. Hydrog. Energy 41(3), 1460–1467 (2016). https://doi.org/10.1016/j.ijhydene.2015.11.125
Dubinin, M.M.: In homogeneous microporous structures of carbonaceous adsorbents. Carbon 19, 321–324 (1981)
Enaime, G., Ennaciri, K., Ounas, A., Baçaoui, A., Seffen, M., Selmi, T., Yaacoubi, A.: Preparation and characterization of activated carbons from olive wastes by physical and chemical activation: application to Indigo carmine adsorption. J. Mater. Environ. Sci. 8(11), 4125–4137 (2017)
Francois, B., Francisco, M.-M.: Dubinin isotherms versus the Brouers–Sotolongo family isotherms: a case study. Adsor. Sci. Technol. 34(9–10), 552–564 (2016). https://doi.org/10.1177/0263617416670909
Freundlich, H.: Over the adsorption in solution. J. Phys. Chem. 57, 385–471 (1906)
Gaspard, S., Altenor, S., Passe-Coutrin, N., Ouensanga, A., Brouers, F.: Parameters from a new kinetic equation to evaluate activated carbons efficiency for water treatment. Water Res. 40(18), 3467–3477 (2006). https://doi.org/10.1016/j.watres.2006.07.018
Ho, Y.S., McKay, G.: Sorption of dye from aqueous solution by peat. Chem. Eng. J. 70(2), 115–124 (1998). https://doi.org/10.1016/S0923-0467(98)00076-1
Ho, Y.S., McKay, G.: Pseudo-second order model for sorption processes. Process Biochem. 34(5), 451–465 (1999). https://doi.org/10.1016/S0032-9592(98)00112-5
Húmpola, P., Odetti, H., Moreno-Piraján, J.C., Giraldo, L.: Activated carbons obtained from agro-industrial waste: textural analysis and adsorption environmental pollutants. Adsorption 22(1), 23–31 (2016). https://doi.org/10.1007/s10450-015-9728-y
IUPAC: International Union of Pure and Applied Chemistry; Korean Chemical Society. 45th IUPAC World Chemestry Congress, Busan, Korea, 9–14 August 2015. Elseviers, Amsterdam (2015)
Jagiello, J.: Stable numerical solution of the adsorption integral equation using splines. Langmuir 10(8), 2778–2785 (1994). https://doi.org/10.1021/la00020a045
Jagiello, J., Olivier, J.P.: 2D-NLDFT adsorption models for carbon slit-shaped pores with surface energetical heterogeneity and geometrical corrugation. Carbon 55, 70–80 (2013). https://doi.org/10.1016/j.carbon.2012.12.011
Jagiello, J., Bandosz, T.J., Putyera, K., Schwarz, J.A.: Determination of proton affinity distributions for chemical systems in aqueous environments using a stable numerical solution of the adsorption intergral equation. J. Colloid Interface Sci. 172, 341–346 (1995)
Jagiello, J., Bandosz, T.J., Schwarz, J.A.: Carbon surface characterization in terms of its acidity constant distribution. Letters to the editor. Carbon 32, 1026–1028 (2000)
Jagiello, J., Ania, C., Parra, J.B., Cook, C.: Dual gas analysis of microporous carbons using 2D-NLDFT heterogeneous surface model and combined adsorption data of N2 and CO2. Carbon 91, 330–337 (2015). https://doi.org/10.1016/j.carbon.2015.05.004
Jaramillo, M.M., Mendoza, A., Vaquero, S., Anderson, M., Palma, J., Marcilla, R.: Role of textural properties and surface functionalities of selected carbons on the electrochemical behaviour of ionic liquid based-supercapacitors. RSC Adv. 2(22), 8439–8446 (2012). https://doi.org/10.1039/C2RA21035E
Jovanović, D.S.: Physical adsorption of gases. Kolloid-Zeitschrift und Zeitschrift für Polymere 235(1), 1203–1213 (1969). https://doi.org/10.1007/bf01542530
Kesraoui, A., Selmi, T., Seffen, M., Brouers, F.: Influence of alternating current on the adsorption of indigo carmine. Environ. Sci. Pollut. Res. 24(11), 1–11 (2016). https://doi.org/10.1007/s11356-016-7201-4
Kopelman, R.: Fractal Reaction Kinetics. Science 241, 1620–1626 (1988). https://doi.org/10.1126/science.241.4873.1620
Lagergren, S.: Zur Theorie der Sogenannten Adsorption Gelöster Stoffe, Kungliga Svenska Vetenskapsakade-miens. Handlingar 24(4), 1–39 (1898)
Langmuir, I.: The adsorption of gases on plane surfaces of glass, mica, and platinum. J. Am. Chem. Soc. 40, 1361 (1918)
Mailler, R., Gasperi, J., Coquet, Y., Buleté, A., Vulliet, E., Deshayes, S., Zedek, S., Mirande-Bret, C., Eudes, V., Bressy, A.: Removal of a wide range of emerging pollutants from wastewater treatment plant discharges by micro-grain activated carbon in fluidized bed as tertiary treatment at large pilot scale. Sci. Total Environ. 542, 983–996 (2016). https://doi.org/10.1016/j.scitotenv.2015.10.153
Meilanov, R.P., Sveshnikova, D.A., Shabanov, O.M.: Fractal nature of sorption kinetics. J. Phys. Chem. A 106(48), 11771–11774 (2002). https://doi.org/10.1021/jp0216575
Miao, M.-S., Liu, Q., Shu, L., Wang, Z., Liu, Y.-Z., Kong, Q.: Removal of cephalexin from effluent by activated carbon prepared from alligator weed: kinetics, isotherms, and thermodynamic analyses. Process. Saf. Environ. Prot. 104(Part B), 481–489 (2016). https://doi.org/10.1016/j.psep.2016.03.017
Ncibi, M., Altenor, S., Seffen, M., Brouers, F., Gaspard, S.: Modelling single compound adsorption onto porous and non-porous sorbents using a deformed Weibull exponential isotherm. Chem. Eng. J. 145(2), 196–202 (2008)
Neimark, A.: A new approach to the determination of the surface fractal dimension of porous solids. Physica A 191(1), 258–262 (1992). https://doi.org/10.1016/0378-4371(92)90536-Y
Pereira, L.M.: Fractal pharmacokinetics. Comput. Math. Methods Med. 11(2), 161–184 (2010). https://doi.org/10.1080/17486700903029280
Rodríguez, A., García, J., Ovejero, G., Mestanza, M.: Adsorption of anionic and cationic dyes on activated carbon from aqueous solutions: equilibrium and kinetics. J. Hazard. Mater. 172(2–3), 1311–1320 (2009). https://doi.org/10.1016/j.jhazmat.2009.07.138
Sandro, A., Carene, B., Emmanuel, E., Lambert, J., Ehrhardt, J.-J., Gaspard, S.: Adsorption studies of methylene blue and phenol onto vetiver roots activated carbon prepared by chemical activation. J. Hazard. Mater. 165, 1029–1039 (2009). https://doi.org/10.1016/j.jhazmat.2008.10.133
Seredych, M., Biggs, M.J., Bandosz, T.J.: Oxygen reduction on chemically heterogeneous iron-containing nanoporous carbon: the effects of specific surface functionalities. Microporous Mesoporous Mater. 221, 137–149 (2016). https://doi.org/10.1016/j.micromeso.2015.09.032
Sethia, G., Sayari, A.: Activated carbon with optimum pore size distribution for hydrogen storage. Carbon 99, 289–294 (2016). https://doi.org/10.1016/j.carbon.2015.12.032
Sips, R.: The structure of a catalyst surface. J. Chem. Phys. 16(5), 490–495 (1948). https://doi.org/10.1063/1.1746922
Sokołowska, Z., Hajnos, M., Hoffmann, C., Renger, M., Sokołowski, S.: Comparison of fractal dimensions of soils estimated from adsorption isotherms,mercury intrusion,and particle size distribution. J. Plant Nutr. Soil Sci. 164(5), 591–599 (2001). https://doi.org/10.1002/1522-2624(200110)164:5<591::AIDLPLN591>3.0.CO;2-Y
Stanislavsky, A., Weron, K.: Is there a motivation for a universal behaviour in molecular populations undergoing chemical reactions? Phys. Chem. Chem. Phys. 15(37), 15595–15601 (2013). https://doi.org/10.1039/C3CP52272E
Stoeckli, F.: Characterization of microporous carbons by adsorption and immersion techniques. Porosity in carbons–characterization and applications. London: Arnold (1995)
Tian, S., Mo, H., Zhang, R., Ning, P., Zhou, T.: Enhanced removal of hydrogen sulfide from a gas stream by 3-aminopropyltriethoxysilane-surface-functionalized activated carbon. Adsorption 15(5), 477 (2009). https://doi.org/10.1007/s10450-009-9198-1
Washburn, E.W.: The dynamics of capillary flow. Phys. Rev. 17(3), 273–283 (1921). https://doi.org/10.1103/PhysRev.17.273
Zhang, Z., Pfefferle, L., Haller, G.L.: Characterization of functional groups on oxidized multi-wall carbon nanotubes by potentiometric titration. Catal. Today 249, 23–29 (2015). https://doi.org/10.1016/j.cattod.2014.12.013
Acknowledgements
The Tunisian group gratefully acknowledges the financial support of the EU-METALIC Erasmus Mundus project, and of the Tunisian Ministry of Higher Education and Scientific Research. The French group gratefully acknowledges the financial support of the CPER 2007-2013 “Structuring the Competitiveness Fibre Cluster”, through local (Conseil Général des Vosges), regional (Région Lorraine), national (DRRT and FNADT) and European (FEDER) funds.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Selmi, T., Seffen, M., Sammouda, H. et al. Physical meaning of the parameters used in fractal kinetic and generalised adsorption models of Brouers–Sotolongo. Adsorption 24, 11–27 (2018). https://doi.org/10.1007/s10450-017-9927-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10450-017-9927-9