Abstract
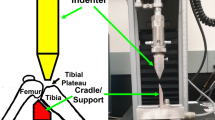
The onset of post-traumatic osteoarthritis (PTOA) remains prevalent following traumatic joint injury such as anterior cruciate ligament (ACL) rupture, and animal models are important for studying the pathomechanisms of PTOA. Noninvasive ACL injury using the tibial compression model in the rat has not been characterized, and it may represent a more clinically relevant model than the common surgical ACL transection model. This study employed four loading profiles to induce ACL injury, in which motion capture analysis was performed, followed by quantitative joint laxity testing. High-speed, high-displacement loading repeatedly induces complete ACL injury, which causes significant increases in anterior-posterior and varus laxity. No loading protocol induced valgus laxity. Tibial internal rotation and anterior subluxation occurs up to the point of ACL failure, after which the tibia rotates externally as it subluxes over the femoral condyles. High displacement was more determinative of ACL injury compared to high speed. Low-speed protocols induced ACL avulsion from the femoral footprint whereas high-speed protocols caused either midsubstance rupture, avulsion, or a combination injury of avulsion and midsubstance rupture. This repeatable, noninvasive ACL injury protocol can be utilized in studies assessing PTOA or ACL reconstruction in the rat.




Similar content being viewed by others
References
Altman, R., et al. Biomechanical and biochemical properties of dog cartilage in experimentally induced osteoarthritis. Ann. Rheum. Dis. 43:83–90, 1984.
Anderson, D. D., et al. Post-traumatic osteoarthritis: improved understanding and opportunities for early intervention. J. Orthop. Res. 29:802–809, 2011.
Boden, B. P., et al. Mechanisms of anterior cruciate ligament injury. Orthopedics 23:573–578, 2000.
Boden, B. P., et al. Noncontact anterior cruciate ligament injuries: mechanisms and risk factors. J. Am. Acad. Orthop. Surg. 18:520–527, 2010.
Brophy, R. H., et al. Effect of short-duration low-magnitude cyclic loading versus immobilization on tendon-bone healing after ACL reconstruction in a rat model. J. Bone Joint Surg. 93:381–393, 2011.
Brown, T. D., et al. Posttraumatic osteoarthritis: a first estimate of incidence, prevalence, and burden of disease. J. Orthop. Trauma 20:739–744, 2006.
Buckwalter, J. A., and T. D. Brown. Joint injury, repair, and remodeling: roles in post-traumatic osteoarthritis. Clin. Orthop. Relat. Res. 423:7–16, 2004.
Christiansen, B., et al. Musculoskeletal changes following non-invasive knee injury using a novel mouse model of post-traumatic osteoarthritis. Osteoarthr. Cartil. 20:773–782, 2012.
Dirschl, D. R., et al. Articular fractures. J. Am. Acad. Orthop. Surg. 12:416–423, 2004.
Gillquist, J., and K. Messner. Anterior cruciate ligament reconstruction and the long term incidence of gonarthrosis. Sports Med. 27:143–156, 1999.
Guilak, F., et al. Mechanical and biochemical changes in the superficial zone of articular cartilage in canine experimental osteoarthritis. J. Orthop. Res. 12:474–484, 1994.
Hashimoto, S., et al. Development and regulation of osteophyte formation during experimental osteoarthritis. Osteoarthr. Cartil. 10:180–187, 2002.
Jansen, H., et al. Effects of low-energy NMR on posttraumatic osteoarthritis: observations in a rabbit model. Arch. Orthop. Trauma Surg. 131:863–868, 2011.
Jansen, H., et al. Detection of vascular endothelial growth factor (VEGF) in moderate osteoarthritis in a rabbit model. Ann. Anat. 194:452–456, 2012.
Kadonishi, Y., et al. Acceleration of tendon–bone healing in anterior cruciate ligament reconstruction using an enamel matrix derivative in a rat model. J. Bone Joint Surg. Br. 94:205–209, 2012.
Koga, H., et al. Mechanisms for noncontact anterior cruciate ligament injuries knee joint kinematics in 10 injury situations from female team handball and basketball. Am. J. Sports Med. 38:2218–2225, 2010.
Lee, J. K., et al. Anterior cruciate ligament tears: MR imaging compared with arthroscopy and clinical tests. Radiology 166:861–864, 1988.
Levine, J. W., et al. Clinically relevant injury patterns after an anterior cruciate ligament injury provide insight into injury mechanisms. Am. J. Sports Med. 41:385–395, 2013.
Linko, E., et al. Surgical versus conservative interventions for anterior cruciate ligament ruptures in adults. Cochrane Database Syst. Rev. 2:CD001356, 2005.
Lockwood, K. A., et al. Comparison of loading rate-dependent injury modes in a murine model of post-traumatic osteoarthritis. J. Orthop. Res. 32:79–88, 2014.
Lohmander, L., et al. High prevalence of knee osteoarthritis, pain, and functional limitations in female soccer players twelve years after anterior cruciate ligament injury. Arthritis Rheum. 50:3145–3152, 2004.
Lohmander, L. S., et al. The long-term consequence of anterior cruciate ligament and meniscus injuries osteoarthritis. Am. J. Sports Med. 35:1756–1769, 2007.
Mankin, H., A. Grodzinsky, and J. A. Buckwalter. Articular cartilage and osteoarthritis. In: Orthopaedic Basic Science: Foundations of Clinical Practice, edited by T. Einhorn. Chicago, IL: American Academy of Orthopaedic Surgeons, 2007.
Martin, J., and J. Buckwalter. Post-traumatic osteoarthritis: the role of stress induced chondrocyte damage. Biorheology 43:517–521, 2006.
Mazzocca, A. D., et al. Valgus medial collateral ligament rupture causes concomitant loading and damage of the anterior cruciate ligament. J. Knee Surg. 16:148–151, 2003.
Meyer, E. G., and R. C. Haut. Excessive compression of the human tibio-femoral joint causes ACL rupture. J. Biomech. 38:2311–2316, 2005.
Meyer, E. G., and R. C. Haut. Anterior cruciate ligament injury induced by internal tibial torsion or tibiofemoral compression. J. Biomech. 41:3377–3383, 2008.
Mifune, Y., et al. Tendon graft revitalization using adult anterior cruciate ligament (ACL)-derived CD34+ cell sheets for ACL reconstruction. Biomaterials 34:5476–5487, 2013.
Myklebust, G., and R. Bahr. Return to play guidelines after anterior cruciate ligament surgery. Br. J. Sports Med. 39:127–131, 2005.
Onur, T. S., et al. Joint instability and cartilage compression in a mouse model of posttraumatic osteoarthritis. J. Orthop. Res. 32:318–323, 2014.
Quatman, C. E., and T. E. Hewett. The anterior cruciate ligament injury controversy: is “valgus collapse” a sex-specific mechanism? Br. J. Sports Med. 43:328–335, 2009.
Quatman, C. E., et al. Preferential loading of the ACL compared with the MCL during landing a novel in sim approach yields the multiplanar mechanism of dynamic valgus during ACL injuries. Am. J. Sports Med. 42:177–186, 2013. doi:10.1177/0363546513506558.
Roos, H., et al. Osteoarthritis of the knee after injury to the anterior cruciate ligament or meniscus: the influence of time and age. Osteoarthr. Cartil. 3:261–267, 1995.
Ruan, M. Z., et al. Quantitative imaging of murine osteoarthritic cartilage by phase-contrast micro-computed tomography. Arthritis Rheum. 65:388–396, 2013.
Sankar, W. N., et al. Combined anterior cruciate ligament and medial collateral ligament injuries in adolescents. J. Pediatr. Orthop. 26:733–736, 2006.
Shin, C. S., A. M. Chaudhari, and T. P. Andriacchi. The effect of isolated valgus moments on ACL strain during single-leg landing: a simulation study. J. Biomech. 42:280–285, 2009.
Tang, Z., et al. Contributions of different intraarticular tissues to the acute phase elevation of synovial fluid MMP-2 following rat ACL rupture. J. Orthop. Res. 27:243–248, 2009.
Von Porat, A., E. M. Roos, and H. Roos. High prevalence of osteoarthritis 14 years after an anterior cruciate ligament tear in male soccer players: a study of radiographic and patient relevant outcomes. Ann. Rheum. Dis. 63:269–273, 2004.
Wu, Y., et al. The profile of MMP and TIMP in injured rat ACL. Mol. Cell Biomech. 7:115–1124, 2009.
Yoon, K. H., J. H. Yoo, and K.-I. Kim. Bone contusion and associated meniscal and medial collateral ligament injury in patients with anterior cruciate ligament rupture. J. Bone Joint Surg. 93:1510–1518, 2011.
Author information
Authors and Affiliations
Corresponding author
Additional information
Associate Editor Michael R. Torry oversaw the review of this article.
Rights and permissions
About this article
Cite this article
Maerz, T., Kurdziel, M.D., Davidson, A.A. et al. Biomechanical Characterization of a Model of Noninvasive, Traumatic Anterior Cruciate Ligament Injury in the Rat. Ann Biomed Eng 43, 2467–2476 (2015). https://doi.org/10.1007/s10439-015-1292-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10439-015-1292-9