Abstract
Purpose
To quantitatively examine changes in choroidal circulation hemodynamics in patients with acute posterior multifocal placoid pigment epitheliopathy (APMPPE).
Methods
Retrospective observational case series. Five eyes of 3 APMPPE patients were included. In all APMPPE eyes, laser speckle flowgraphy was conducted to evaluate the mean blur rate (MBR), a quantitative index of relative blood flow velocity. The changes in MBR at the initial visit and after 3 and 6 months were statistically analyzed. In 2 eyes, enhanced depth imaging optical coherence tomography was used to measure central choroidal thickness.
Results
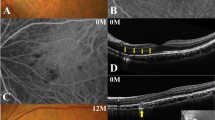
In all eyes, multiple subretinal placoid lesions spontaneously regressed with recovery of outer retinal morphology. The average MBR significantly increased at 3 and 6 months after baseline at the fovea by 49.6 and 54.2 % and at the lesion site by 65.2 and 70.8 %, respectively. These changes at both regions during the 6-month follow-up were statistically significant (P = 0.02 for each). The mean central choroidal thickness decreased with time (518.0 µm at baseline to 344.5 µm at 6 months).
Conclusions
Our data revealed the significant elevation of choroidal blood flow velocity and the substantial reduction of choroidal thickness with regression of APMPPE, suggesting that impaired choroidal circulation is involved in the pathogenesis of APMPPE.



Similar content being viewed by others
References
Gass JD. Acute posterior multifocal placoid pigment epitheliopathy. Arch Ophthalmol. 1968;80:177–85.
Agarwal A. Acute posterior multifocal placoid pigment epitheliopathy. In: Agarwal A, editor. Gass’ atlas of macular diseases. 5th ed. Edinburgh: Elsevier Saunders; 2012. p. 954–60.
Fiore T, Iaccheri B, Androudi S, Papadaki TG, Anzaar F, Brazitikos P, et al. Acute posterior multifocal placoid pigment epitheliopathy: outcome and visual prognosis. Retina. 2009;29:994–1001.
Goldenberg D, Habot-Wilner Z, Loewenstein A, Goldstein M. Spectral domain optical coherence tomography classification of acute posterior multifocal placoid pigment epitheliopathy. Retina. 2012;32:1403–10.
Mensah E, Vafidis GC. Acute posterior multifocal placoid pigment epitheliopathy in a 7-year-old girl. J Pediatr Ophthalmol Strabismus. 2002;39:239–41.
Taich A, Johnson MW. A syndrome resembling acute posterior multifocal placoid pigment epitheliopathy in older adults. Retina. 2009;29:149–56.
Furusho F, Imaizumi H, Takeda M. One case of Harada disease complicated by acute posterior multifocal placoid pigment epitheliopathy-like recurrence in both eyes. Jpn J Ophthalmol. 2001;45:117–8.
Tanigawa M, Tsukahara Y, Yamanaka H. A case of acute posterior multifocal placoid pigment epitheliopathy demonstrating Vogt-Koyanagi-Harada disease-like optical coherence tomography findings in the acute stage. Case Rep Ophthalmol. 2013;11:172–9.
Lee GE, Lee BW, Rao NA, Fawzi AA. Spectral domain optical coherence tomography and autofluorescence in a case of acute posterior multifocal placoid pigment epitheliopathy mimicking Vogt-Koyanagi-Harada disease: case report and review of literature. Ocul Immunol Inflamm. 2011;19:42–7.
Howe LJ, Woon H, Graham EM, Fitzke F, Bhandari A, Marshall J. Choroidal hypoperfusion in acute posterior multifocal placoid pigment epitheliopathy. An indocyanine green angiography study. Ophthalmology. 1995;102:790–8.
Spaide RF, Yannuzzi LA, Slakter J. Choroidal vasculitis in acute posterior multifocal placoid pigment epitheliopathy. Br J Ophthalmol. 1991;75:685–7.
El-Markaby HS, Mohammed TH, El-Raggal TM. Acute posterior multifocal placoid pigment epitheliopathy: role of TNF blocker in severe cases. Retina. 2012;32:2102–7.
Hirano Y, Yasukawa T, Nagai H, Ogura Y. Spatio-temporal understanding of the pathology of acute posterior multifocal placoid pigment epitheliopathy. Jpn J Ophthalmol. 2012;56:371–4.
Tamaki Y, Araie M, Kawamoto E, Eguchi S, Fujii H. Noncontact, two-dimensional measurement of retinal microcirculation using laser speckle phenomenon. Invest Ophthalmol Vis Sci. 1994;35:3825–34.
Sugiyama T. Basic technology and clinical applications of the updated model of laser speckle flowgraphy to ocular diseases. Photonics. 2014;1:220–34.
Aizawa N, Yokoyama Y, Chiba N, Omodaka K, Yasuda M, Otomo T, et al. Reproducibility of retinal circulation measurements obtained using laser speckle flowgraphy-NAVI in patients with glaucoma. Clin Ophthalmol. 2011;5:1171–6.
Isono H, Kishi S, Kimura Y, Hagiwara N, Konishi N, Fujii H. Observation of choroidal circulation using index of erythrocytic velocity. Arch Ophthalmol. 2003;121:225–31.
Saito M, Saito W, Hashimoto Y, Yoshizawa C, Fujiya A, Noda K, et al. Macular choroidal blood flow velocity decreases with regression of acute central serous chorioretinopathy. Br J Ophthalmol. 2013;97:775–80.
Saito M, Saito W, Hashimoto Y, Yoshizawa C, Shinmei Y, Noda K, et al. Correlation between decreased choroidal blood flow velocity and the pathogenesis of acute zonal occult outer retinopathy. Clin Exp Ophthalmol. 2014;42:139–50.
Hirooka K, Saito W, Namba K, Takemoto Y, Mizuuchi K, Uno T, et al. Relationship between choroidal blood flow velocity and choroidal thickness during systemic corticosteroid therapy for Vogt-Koyanagi-Harada disease. Graefes Arch Clin Exp Ophthalmol. 2015;253:609–17.
Hashimoto Y, Saito W, Saito M, Hirooka K, Mori S, Noda K, et al. Decreased choroidal blood flow velocity in the pathogenesis of multiple evanescent white dot syndrome. Graefes Arch Clin Exp Ophthalmol. 2015;253:1457–64.
Hashimoto Y, Saito W, Saito M, Hirooka K, Mori S, Noda K, et al. Increased choroidal blood flow velocity with regression of unilateral acute idiopathic maculopathy. Jpn J Ophthalmol. 2015;59:252–60.
Riva CE, Titze P, Hero M, Petrig BL. Effect of acute decreases of perfusion pressure on choroidal blood flow in humans. Invest Ophthalmol Vis Sci. 1997;38:1752–60.
Okuno T, Sugiyama T, Kohyama M, Kojima S, Oku H, Ikeda T. Ocular blood flow changes after dynamic exercise in humans. Eye. 2006;20:796–800.
Schneider U, Inhoffen W, Gelisken F. Indocyanine green angiography in a case of unilateral recurrent posterior acute multifocal placoid pigment epitheliopathy. Acta Ophthalmol Scand. 2003;81:72–5.
Watanabe G, Fujii H, Kishi S. Imaging of choroidal hemodynamics in eyes with polypoidal choroidal vasculopathy using laser speckle phenomenon. Jpn J Ophthalmol. 2008;52:175–81.
Takahashi A, Saito W, Hashimoto Y, Saito M, Ishida S. Impaired circulation in the thickened choroid of a patient with serpiginous choroiditis. Ocul Immunol Inflamm. 2014;22:409–13.
Hirooka K, Saito W, Hashimoto Y, Saito M, Ishida S. Increased macular choroidal blood flow velocity and decreased choroidal thickness with regression of punctate inner choroidopathy. BMC Ophthalmol. 2014;14:73.
Maruko I, Iida T, Sugano Y, Ojima A, Ogasawara M, Spaide RF. Subfoveal choroidal thickness after treatment of central serous chorioretinopathy. Ophthalmology. 2010;117:1792–9.
Yenerel NM, Gorgun E, Dinc UA, Oncel M. Treatment of cystoid macular edema due to acute posterior multifocal placoid pigment epitheliopathy. Ocul Immunol Inflamm. 2008;16:67–71.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflicts of interest
K. Hirooka, None; W. Saito, None; M. Saito, None; Y. Hashimoto, None; S. Mori, None; K. Noda, None; S. Ishida, None.
About this article
Cite this article
Hirooka, K., Saito, W., Saito, M. et al. Increased choroidal blood flow velocity with regression of acute posterior multifocal placoid pigment epitheliopathy. Jpn J Ophthalmol 60, 172–178 (2016). https://doi.org/10.1007/s10384-016-0440-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10384-016-0440-6