Abstract
Purpose
To determine whether water-dispersible hesperetin (WD-Hpt) can prevent degeneration of ganglion cell neurons in the ischemic retina.
Methods
Ischemia reperfusion (I/R) injury was induced by increasing the intraocular pressure of mice to 110 mmHg for 40 min. Mice received daily intraperitoneal injections with either normal saline (NS, 0.3 ml/day) or WD-Hpt (0.3 ml, 200 mg/kg/day). Reactive oxygen species (ROS) was assessed by dihydroethidium and nitrotyrosine formation. Inflammation was estimated by microglial morphology in the retina. Lipopolysaccharide (LPS)-stimulated BV-2 cells were used to explore the anti-inflammatory effect of WD-Hpt on activated microglia by quantifying the expression of IL-1β using real-time quantitative reverse transcription-polymerase chain reaction. Ganglion cell loss was assessed by immunohistochemistry of NeuN. Glial activation was quantified with glial fibrillary acidic protein (GFAP) immunoreactivity. Apoptosis was evaluated with a terminal deoxynucleotidyl transferase (TUNEL) assay and immunohistochemistry of cleaved caspase-3. Phosphorylation of extracellular signal-regulated kinase (p-ERK) was surveyed by western blotting.
Results
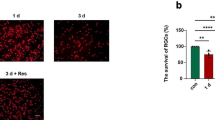
WD-Hpt decreased I/R-induced ROS formation. WD-Hpt alleviated microglial activation induced by I/R and reduced mRNA levels of IL-1β in LPS-stimulated BV-2. I/R resulted in a 37 % reduction in the number of ganglion cells in the NS-treated mice, whereas the reduction was only 5 % in the WD-Hpt-treated mice. In addition, WD-Hpt mitigated the immunoreactivity of GFAP, increased expression of cleaved caspase-3, increased number of TUNEL positive cells and p-ERK after I/R.
Conclusions
WD-Hpt protected ganglion cells from I/R injury by inhibiting oxidative stress and modulating cell death signaling. Moreover, WD-Hpt had an anti-inflammatory effect through the suppression of activated microglia.







Similar content being viewed by others
References
Joussen AM, Poulaki V, Le ML, Koizumi K, Esser C, Janicki H, et al. A central role for inflammation in the pathogenesis of diabetic retinopathy. FASEB J. 2004;18:1450–2.
Esser P, Bresgen M, Fischbach R, Heimann K, Wiedemann P. Intercellular adhesion molecule-1 levels in plasma and vitreous from patients with vitreoretinal disorders. Ger J Ophthalmol. 1995;4:269–74.
Nagai N, Izumi-Nagai K, Oike Y, Koto T, Satofuka S, Ozawa Y, et al. Suppression of diabetes-induced retinal inflammation by blocking the angiotensin II type 1 receptor or its downstream nuclear factor-kappaB pathway. Invest Ophthalmol Vis Sci. 2007;48:4342–50.
Rungger-Brandle E, Dosso AA, Leuenberger PM. Glial reactivity, an early feature of diabetic retinopathy. Invest Ophthalmol Vis Sci. 2000;41:1971–80.
Dyer MA, Cepko CL. Control of Muller glial cell proliferation and activation following retinal injury. Nat Neurosci. 2000;3:873–80.
Yoshida S, Yoshida A, Ishibashi T. Induction of IL-8, MCP-1, and bFGF by TNF-alpha in retinal glial cells: implications for retinal neovascularization during post-ischemic inflammation. Graefes Arch Clin Exp Ophthalmol. 2004;242:409–13.
Barber AJ, Lieth E, Khin SA, Antonetti DA, Buchanan AG, Gardner TW. Neural apoptosis in the retina during experimental and human diabetes. Early onset and effect of insulin. J Clin Invest. 1998;102:783–91.
Villarroel M, Ciudin A, Hernandez C, Simo R. Neurodegeneration: an early event of diabetic retinopathy. World J Diabetes. 2010;1:57–64.
Rios L, Cluzel J, Vennat JC, Menerath JM, Doly M. Comparison of intraocular treatment of DMTU and SOD following retinal ischemia in rats. J Ocul Pharmacol Ther. 1999;15:547–56.
Nayak MS, Kita M, Marmor MF. Protection of rabbit retina from ischemic injury by superoxide dismutase and catalase. Invest Ophthalmol Vis Sci. 1993;34:2018–22.
Szabo ME, Droy-Lefaix MT, Doly M, Carre C, Braquet P. Ischemia and reperfusion-induced histologic changes in the rat retina. Demonstration of a free radical-mediated mechanism. Invest Ophthalmol Vis Sci. 1991;32:1471–8.
Treins C, Giorgetti-Peraldi S, Murdaca J, Van Obberghen E. Regulation of vascular endothelial growth factor expression by advanced glycation end products. J Biol Chem. 2001;276:43836–41.
Satofuka S, Ichihara A, Nagai N, Noda K, Ozawa Y, Fukamizu A, et al. (Pro)renin receptor-mediated signal transduction and tissue renin-angiotensin system contribute to diabetes-induced retinal inflammation. Diabetes. 2009;58:1625–33.
Takeda M, Takamiya A, Yoshida A, Kiyama H. Extracellular signal-regulated kinase activation predominantly in Muller cells of retina with endotoxin-induced uveitis. Invest Ophthalmol Vis Sci. 2002;43:907–11.
Zhou RH, Yan H, Wang BR, Kuang F, Duan XL, Xu Z. Role of extracellular signal-regulated kinase in glutamate-stimulated apoptosis of rat retinal ganglion cells. Curr Eye Res. 2007;32:233–9.
Rosenbaum DM, Rosenbaum PS, Gupta H, Singh M, Aggarwal A, Hall DH, et al. The role of the p53 protein in the selective vulnerability of the inner retina to transient ischemia. Invest Ophthalmol Vis Sci. 1998;39:2132–9.
van Dijk HW, Verbraak FD, Kok PH, Garvin MK, Sonka M, Lee K, et al. Decreased retinal ganglion cell layer thickness in patients with type 1 diabetes. Invest Ophthalmol Vis Sci. 2010;51:3660–5.
Hirata A, Murakami Y, Shoji M, Kadoma Y, Fujisawa S. Kinetics of radical-scavenging activity of hesperetin and hesperidin and their inhibitory activity on COX-2 expression. Anticancer Res. 2005;25:3367–74.
Galati EM, Monforte MT, Kirjavainen S, Forestieri AM, Trovato A, Tripodo MM. Biological effects of hesperidin, a citrus flavonoid. Note I: anti-inflammatory and analgesic activity. Farmaco. 1994;40:709–12.
Kaul TN, Middleton E Jr, Ogra PL. Antiviral effect of flavonoids on human viruses. J Med Virol. 1985;15:71–9.
Zarebczan B, Pinchot SN, Kunnimalaiyaan M, Chen H. Hesperetin, a potential therapy for carcinoid cancer. Am J Surg. 2011;201:329–32 (discussion 33).
Garg A, Garg S, Zaneveld LJ, Singla AK. Chemistry and pharmacology of the Citrus bioflavonoid hesperidin. Phytother Res. 2001;15:655–69.
Kumar B, Gupta SK, Srinivasan BP, Nag TC, Srivastava S, Saxena R, et al. Hesperetin rescues retinal oxidative stress, neuroinflammation and apoptosis in diabetic rats. Microvasc Res. 2013;87:65–74.
Takumi H, Nakamura H, Simizu T, Harada R, Kometani T, Nadamoto T, et al. Bioavailability of orally administered water-dispersible hesperetin and its effect on peripheral vasodilatation in human subjects: implication of endothelial functions of plasma conjugated metabolites. Food Funct. 2012;3:389–98.
Da T, Verkman AS. Aquaporin-4 gene disruption in mice protects against impaired retinal function and cell death after ischemia. Invest Ophthalmol Vis Sci. 2004;45:4477–83.
Yokota H, Narayanan SP, Zhang W, Liu H, Rojas M, Xu Z, et al. Neuroprotection from retinal ischemia/reperfusion injury by NOX2 NADPH oxidase deletion. Invest Ophthalmol Vis Sci. 2011;52:8123–31.
Shen J, Nakamura H, Fujisaki Y, Tanida M, Horii Y, Fuyuki R, et al. Effect of 4G-alpha-glucopyranosyl hesperidin on brown fat adipose tissue- and cutaneous-sympathetic nerve activity and peripheral body temperature. Neurosci Lett. 2009;461:30–5.
Rojas M, Zhang W, Lee DL, Romero MJ, Nguyen DT, Al-Shabrawey M, et al. Role of IL-6 in angiotensin II-induced retinal vascular inflammation. Invest Ophthalmol Vis Sci. 2010;51:1709–18.
Al-Shabrawey M, Bartoli M, El-Remessy AB, Platt DH, Matragoon S, Behzadian MA, et al. Inhibition of NAD(P)H oxidase activity blocks vascular endothelial growth factor overexpression and neovascularization during ischemic retinopathy. Am J Pathol. 2005;167:599–607.
Miller FJ Jr, Gutterman DD, Rios CD, Heistad DD, Davidson BL. Superoxide production in vascular smooth muscle contributes to oxidative stress and impaired relaxation in atherosclerosis. Circ Res. 1998;82:1298–305.
Kirkeby S, Thomsen CE. Quantitative immunohistochemistry of fluorescence labelled probes using low-cost software. J Immunol Methods. 2005;301:102–13.
Leahy KM, Ornberg RL, Wang Y, Zhu Y, Gidday JM, Connor JR, et al. Quantitative ex vivo detection of rodent retinal ganglion cells by immunolabeling Brn-3b. Exp Eye Res. 2004;79:131–40.
Blasi E, Barluzzi R, Bocchini V, Mazzolla R, Bistoni F. Immortalization of murine microglial cells by a v-raf/v-myc carrying retrovirus. J Neuroimmunol. 1990;27:229–37.
Luo C, Chen M, Xu H. Complement gene expression and regulation in mouse retina and retinal pigment epithelium/choroid. Mol Vis. 2011;17:1588–97.
Tanaka T, Kai S, Matsuyama T, Adachi T, Fukuda K, Hirota K. General anesthetics inhibit LPS-induced IL-1beta expression in glial cells. PLoS One. 2013;8:e82930.
Nagata M. Inflammatory cells and oxygen radicals. Curr Drug Targets Inflamm Allergy. 2005;4:503–4.
Hordijk PL. Regulation of NADPH oxidases: the role of Rac proteins. Circ Res. 2006;98:453–62.
Kaur C, Rathnasamy G, Ling EA. Roles of activated microglia in hypoxia induced neuroinflammation in the developing brain and the retina. J Neuroimmune Pharmacol. 2013;8:66–78.
Arana L, Ordonez M, Ouro A, Rivera IG, Gangoiti P, Trueba M, et al. Ceramide 1-phosphate induces macrophage chemoattractant protein-1 release: involvement in ceramide 1-phosphate-stimulated cell migration. Am J Physiol Endocrinol Metab. 2013;304:E1213–26.
Ling EA, Wong WC. The origin and nature of ramified and amoeboid microglia: a historical review and current concepts. Glia. 1993;7:9–18.
Xu H, Chen M, Forrester JV. Para-inflammation in the aging retina. Prog Retin Eye Res. 2009;28:348–68.
Liu S, Li ZW, Weinreb RN, Xu G, Lindsey JD, Ye C, et al. Tracking retinal microgliosis in models of retinal ganglion cell damage. Invest Ophthalmol Vis Sci. 2012;53:6254–62.
Colton CA, Gilbert DL. Production of superoxide anions by a CNS macrophage, the microglia. FEBS Lett. 1987;223:284–8.
Banati RB, Rothe G, Valet G, Kreutzberg GW. Detection of lysosomal cysteine proteinases in microglia: flow cytometric measurement and histochemical localization of cathepsin B and L. Glia. 1993;7:183–91.
Gehrmann J, Matsumoto Y, Kreutzberg GW. Microglia: intrinsic immuneffector cell of the brain. Brain Res Brain Res Rev. 1995;20:269–87.
Bonne C, Muller A, Villain M. Free radicals in retinal ischemia. Gen Pharmacol. 1998;30:275–80.
Roth S, Shaikh AR, Hennelly MM, Li Q, Bindokas V, Graham CE. Mitogen-activated protein kinases and retinal ischemia. Invest Ophthalmol Vis Sci. 2003;44:5383–95.
Zhang C, Lam TT, Tso MO. Heterogeneous populations of microglia/macrophages in the retina and their activation after retinal ischemia and reperfusion injury. Exp Eye Res. 2005;81:700–9.
Deng Y, Lu J, Sivakumar V, Ling EA, Kaur C. Amoeboid microglia in the periventricular white matter induce oligodendrocyte damage through expression of proinflammatory cytokines via MAP kinase signaling pathway in hypoxic neonatal rats. Brain Pathol. 2008;18:387–400.
Rivera JC, Sitaras N, Noueihed B, Hamel D, Madaan A, Zhou T, et al. Microglia and interleukin-1beta in ischemic retinopathy elicit microvascular degeneration through neuronal semaphorin-3A. Arterioscler Thromb Vasc Biol. 2013;33:1881–91.
Shimazu R, Akashi S, Ogata H, Nagai Y, Fukudome K, Miyake K, et al. MD-2, a molecule that confers lipopolysaccharide responsiveness on Toll-like receptor 4. J Exp Med. 1999;189:1777–82.
Dvoriantchikova G, Barakat DJ, Hernandez E, Shestopalov VI, Ivanov D. Toll-like receptor 4 contributes to retinal ischemia/reperfusion injury. Mol Vis. 2010;16:1907–12.
Produit-Zengaffinen N, Pournaras CJ, Schorderet DF. Retinal ischemia-induced apoptosis is associated with alteration in Bax and Bcl-x(L) expression rather than modifications in Bak and Bcl-2. Mol Vis. 2009;15:2101–10.
Zheng L, Gong B, Hatala DA, Kern TS. Retinal ischemia and reperfusion causes capillary degeneration: similarities to diabetes. Invest Ophthalmol Vis Sci. 2007;48:361–7.
Lam TT, Abler AS, Tso MO. Apoptosis and caspases after ischemia-reperfusion injury in rat retina. Invest Ophthalmol Vis Sci. 1999;40:967–75.
Doonan F, Cotter TG. Apoptosis: a potential therapeutic target for retinal degenerations. Curr Neurovasc Res. 2004;1:41–53.
Zhang Y, Cho CH, Atchaneeyasakul LO, McFarland T, Appukuttan B, Stout JT. Activation of the mitochondrial apoptotic pathway in a rat model of central retinal artery occlusion. Invest Ophthalmol Vis Sci. 2005;46:2133–9.
Parone PA, James D, Martinou JC. Mitochondria: regulating the inevitable. Biochimie. 2002;84:105–11.
Wurm A, Iandiev I, Uhlmann S, Wiedemann P, Reichenbach A, Bringmann A, et al. Effects of ischemia-reperfusion on physiological properties of Muller glial cells in the porcine retina. Invest Ophthalmol Vis Sci. 2011;52:3360–7.
Wurm A, Lipp S, Pannicke T, Linnertz R, Farber K, Wiedemann P, et al. Involvement of A(1) adenosine receptors in osmotic volume regulation of retinal glial cells in mice. Mol Vis. 2009;15:1858–67.
Akiyama H, Nakazawa T, Shimura M, Tomita H, Tamai M. Presence of mitogen-activated protein kinase in retinal Muller cells and its neuroprotective effect in ischemia-reperfusion injury. NeuroReport. 2002;13:2103–7.
Rainey-Smith S, Schroetke LW, Bahia P, Fahmi A, Skilton R, Spencer JP, et al. Neuroprotective effects of hesperetin in mouse primary neurones are independent of CREB activation. Neurosci Lett. 2008;438:29–33.
Acknowledgments
This work is supported by a Grant in aid for Young Scientists (B) Grant 25861609 (HY) from the Ministry of Education, Science and Culture, Tokyo, Japan.
Conflicts of interest
A. Shimouchi, Grant (Ezaki Glico Co., Ltd.); H. Yokota, Grant (Ezaki Glico Co., Ltd.); S. Ono, None; C. Matsumoto, None; T. Tamai, Employee (Ezaki Glico Co., Ltd.); H. Takumi, Employee (Ezaki Glico Co., Ltd.); S. P. Narayanan, None; S. Kimura, None; H. Kobayashi, None; R. B. Caldwell, None; T. Nagaoka, None; A. Yoshida, None.
Author information
Authors and Affiliations
Corresponding author
About this article
Cite this article
Shimouchi, A., Yokota, H., Ono, S. et al. Neuroprotective effect of water-dispersible hesperetin in retinal ischemia reperfusion injury. Jpn J Ophthalmol 60, 51–61 (2016). https://doi.org/10.1007/s10384-015-0415-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10384-015-0415-z