Abstract
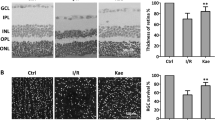
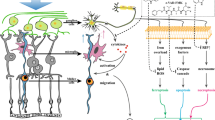
Glaucoma, as an ischemia-reperfusion (I/R) injury disease, leading irreversible blindness through the loss of retinal ganglion cells (RGCs), mediated by various pathways. Resveratrol (Res) is a polyphenolic compound that exerts protective effects against I/R injury in many tissues. This article aimed to expound the underlying mechanisms through which Res protects RGCs and reduces visual dysfunction in vivo. An experimental glaucoma model was created using 6-8-week wild-type male C57BL/6J mice. Res was injected intraperitoneally for 5 days. The mice were then grouped according to the number of days after surgery and whether Res treatment was administered. We applied the Brn3a-labeled immunofluorescence staining and flash electroretinography (ERG) to assess the survival of RGCs and visual function. The expression of components of the NOD-, LRR-, and pyrin domain-containing protein 3 (NLRP3) inflammasome, the interleukin-1-beta (IL-1β), and vital indicators of kelch-like ECH-associated protein 1 (Keap1)/nuclear factor erythroid 2-related factor 2 (Nrf2)/heme-oxygenase 1 (HO-1) pathway at the protein and RNA levels were detected respectively. The survival of RGCs was reduced after surgery compared to controls, whereas Res application rescued RGCs and improved visual dysfunction. In conclusion, our results discovered that Res administration showed neuroprotective effects through inhibition of the NLRP3 inflammasome pathway and activation of Keap1/Nrf2/HO-1 pathway. Thus, we further elucidated the potential of Res in glaucoma therapy.









Similar content being viewed by others
Data Availability
All the data and results used in this study are available from the corresponding author on reasonable request.
References
Schuster AK, Erb C, Hoffmann EM, Dietlein T, Pfeiffer N (2020) The diagnosis and treatment of glaucoma. Dtsch Arztebl Int 117(13):225–234. https://doi.org/10.3238/arztebl.2020.0225
Kang JM, Tanna AP (2021) Glaucoma. Med Clin North Am 105(3):493–510. https://doi.org/10.1016/j.mcna.2021.01.004
Stein JD, Khawaja AP, Weizer JS (2021) Glaucoma in adults-screening, diagnosis, and management: a review. JAMA 325(2):164–174. https://doi.org/10.1001/jama.2020.21899
Huang Y, Xu W, Zhou R (2021) NLRP3 inflammasome activation and cell death. Cell Mol Immunol 18(9):2114–2127. https://doi.org/10.1038/s41423-021-00740-6
Wang L, Hauenstein AV (2020) The NLRP3 inflammasome: mechanism of action, role in disease and therapies. Mol Aspects Med 76:100889. https://doi.org/10.1016/j.mam.2020.100889
Wang M, Pan W, Xu Y, Zhang J, Wan J, Jiang H (2022) Microglia-mediated neuroinflammation: a potential target for the treatment of cardiovascular diseases. J Inflamm Res 15:3083–3094. https://doi.org/10.2147/JIR.S350109
Shen S, Wang Z, Sun H, Ma L (2022) Role of NLRP3 inflammasome in myocardial ischemia-reperfusion injury and ventricular remodeling. Med Sci Monit 28:e934255. https://doi.org/10.12659/MSM.934255
8, Minutoli L, Puzzolo D, Rinaldi M, Irrera N, Marini H, Arcoraci V, Bitto A, Crea G et al (2016) ROS-mediated NLRP3 inflammasome activation in brain, heart, kidney, and testis ischemia/reperfusion injury. Oxid Med Cell Longev 2016:2183026. https://doi.org/10.1155/2016/2183026
Toldo S, Abbate A (2018) The NLRP3 inflammasome in acute myocardial infarction. Nat Rev Cardiol 15(4):203–214. https://doi.org/10.1038/nrcardio.2017.161
Ulasov AV, Rosenkranz AA, Georgiev GP, Sobolev AS (2022) Nrf2/Keap1/ARE signaling: towards specific regulation. Life Sci 291:120111. https://doi.org/10.1016/j.lfs.2021.120111
Saha S, Buttari B, Panieri E, Profumo E, Saso L (2020) An overview of Nrf2 signaling pathway and its role in inflammation. Molecules 25(22). https://doi.org/10.3390/molecules25225474
Panisello-Roselló GBR, Sanchez-Nuno A, Alva S, Roselló-Catafau N, Carbonell J T (2022) Nrf2 and oxidative stress in liver ischemia/reperfusion injury. Febs J 289(18):5463–5479. https://doi.org/10.1111/febs.16336
Yu C, Xiao JH (2021) The Keap1-Nrf2 system: a mediator between oxidative stress and aging. Oxid Med Cell Longev 2021:6635460. https://doi.org/10.1155/2021/6635460
Loboda A, Damulewicz M, Pyza E, Jozkowicz A, Dulak J (2016) Role of Nrf2/HO-1 system in development, oxidative stress response and diseases: an evolutionarily conserved mechanism. Cell Mol Life Sci 73(17):3221–3247. https://doi.org/10.1007/s00018-016-2223-0
Zhang Q, Liu J, Duan H, Li R, Peng W, Wu C (2021) Activation of Nrf2/HO-1 signaling: an important molecular mechanism of herbal medicine in the treatment of atherosclerosis via the protection of vascular endothelial cells from oxidative stress. J Adv Res 34:43–63. https://doi.org/10.1016/j.jare.2021.06.023
Zhang C, Zhao M, Wang B, Su Z, Guo B, Qin L, Zhang W, Zheng R (2021) The Nrf2-NLRP3-caspase-1 axis mediates the neuroprotective effects of celastrol in Parkinson’s disease. Redox Biol 47:102134. https://doi.org/10.1016/j.redox.2021.102134
Bian H, Wang G, Huang J, Liang L, Zheng Y, Wei Y, Wang H, Xiao L et al (2020) Dihydrolipoic acid protects against lipopolysaccharide-induced behavioral deficits and neuroinflammation via regulation of Nrf2/HO-1/NLRP3 signaling in rat. J Neuroinflammation 17(1):166. https://doi.org/10.1186/s12974-020-01836-y
Wu Y, Qiu G, Zhang H, Zhu L, Cheng G, Wang Y, Li Y, Wu W (2021) Dexmedetomidine alleviates hepatic ischaemia-reperfusion injury via the PI3K/AKT/Nrf2-NLRP3 pathway. J Cell Mol Med 25(21):9983–9994. https://doi.org/10.1111/jcmm.16871
Xiao L, Dai Z, Tang W, Liu C, Tang B (2021) Astragaloside IV alleviates cerebral ischemia-reperfusion injury through NLRP3 inflammasome-mediated pyroptosis inhibition via activating Nrf2. Oxid Med Cell Longev 2021:9925561. https://doi.org/10.1155/2021/9925561
Cheng Y, Cheng L, Gao X, Chen S, Wu P, Wang C, Liu Z (2021) Covalent modification of Keap1 at Cys77 and Cys434 by pubescenoside a suppresses oxidative stress-induced NLRP3 inflammasome activation in myocardial ischemia-reperfusion injury. Theranostics 11(2):861–877. https://doi.org/10.7150/thno.48436
Qi Y, Zhao M, Bai Y, Huang L, Yu W, Bian Z, Zhao M, Li X (2014) Retinal ischemia/reperfusion injury is mediated by toll-like receptor 4 activation of NLRP3 inflammasomes. Invest Ophthalmol Vis Sci 55(9):5466–5475. https://doi.org/10.1167/iovs.14-14380
Su CF, Jiang L, Zhang XW, Iyaswamy A, Li M (2021) Resveratrol in rodent models of Parkinson’s disease: a systematic review of experimental studies. Front Pharmacol 12:644219. https://doi.org/10.3389/fphar.2021.644219
Hecker A, Schellnegger M, Hofmann E, Luze H, Nischwitz SP, Kamolz LP, Kotzbeck P (2022) The impact of resveratrol on skin wound healing, scarring, and aging. Int Wound J 19(1):9–28. https://doi.org/10.1111/iwj.13601
Khorshidi F, Poljak A, Liu Y, Lo JW, Crawford JD, Sachdev PS (2021) Resveratrol: a miracle drug in neuropsychiatry or a cognitive enhancer for mice only? A systematic review and meta-analysis. Ageing Res Rev 65:101199. https://doi.org/10.1016/j.arr.2020.101199
Mahdavi A, Bagherniya M, Mirenayat MS, Atkin SL, Sahebkar A (2021) Medicinal plants and phytochemicals regulating insulin resistance and glucose homeostasis in type 2 diabetic patients: a clinical review. Adv Exp Med Biol 1308:161–183. https://doi.org/10.1007/978-3-030-64872-5_13
Sun ZM, Guan P, Luo LF, Qin LY, Wang N, Zhao YS, Ji ES (2020) Resveratrol protects against CIH-induced myocardial injury by targeting Nrf2 and blocking NLRP3 inflammasome activation. Life Sci 245:117362. https://doi.org/10.1016/j.lfs.2020.117362
Ji K, Li Z, Lei Y, Xu W, Ouyang L, He T, Xing Y (2021) Resveratrol attenuates retinal ganglion cell loss in a mouse model of retinal ischemia reperfusion injury via multiple pathways. Exp Eye Res 209:108683. https://doi.org/10.1016/j.exer.2021.108683
Sun YY, Zhu HJ, Zhao RY, Zhou SY, Wang MQ, Yang Y, Guo ZN (2023) Remote ischemic conditioning attenuates oxidative stress and inflammation via the Nrf2/HO-1 pathway in MCAO mice. Redox Biol 66:102852. https://doi.org/10.1016/j.redox.2023.102852
Li J, Xu P, Hong Y, Xie Y, Peng M, Sun R, Guo H, Zhang X et al (2023) Lipocalin-2-mediated astrocyte pyroptosis promotes neuroinflammatory injury via NLRP3 inflammasome activation in cerebral ischemia/reperfusion injury. J Neuroinflammation 20(1):148. https://doi.org/10.1186/s12974-023-02819-5
Hu R, Luo H, Ji Y, Wang Z, Zheng P, Ouyang H, Wang X, Wang Y et al (2023) Activation of NLRP3 signaling contributes to cadmium-induced bone defects, associated with autophagic flux obstruction. Sci Total Environ 893:164787. https://doi.org/10.1016/j.scitotenv.2023.164787
Yang J, Yang N, Luo J, Cheng G, Zhang X, He T, Xing Y (2020) Overexpression of S100A4 protects retinal ganglion cells against retinal ischemia-reperfusion injury in mice. Exp Eye Res 201:108281. https://doi.org/10.1016/j.exer.2020.108281
Franke M, Bieber M, Kraft P, Weber A, Stoll G, Schuhmann MK (2021) The NLRP3 inflammasome drives inflammation in ischemia/reperfusion injury after transient middle cerebral artery occlusion in mice. Brain Behav Immun 92:223–233. https://doi.org/10.1016/j.bbi.2020.12.009
Tang TT, Lv LL, Pan MM, Wen Y, Wang B, Li ZL, Wu M, Wang FM et al (2018) Hydroxychloroquine attenuates renal ischemia/reperfusion injury by inhibiting cathepsin mediated NLRP3 inflammasome activation. Cell Death Dis 9(3):351. https://doi.org/10.1038/s41419-018-0378-3
Qin Q, Yu N, Gu Y, Ke W, Zhang Q, Liu X, Wang K, Chen M (2022) Inhibiting multiple forms of cell death optimizes ganglion cells survival after retinal ischemia reperfusion injury. Cell Death Dis 13(5):507. https://doi.org/10.1038/s41419-022-04911-9
Kong P, Cui ZY, Huang XF, Zhang DD, Guo RJ, Han M (2022) Inflammation and atherosclerosis: signaling pathways and therapeutic intervention. Signal Transduct Target Ther 7(1):131. https://doi.org/10.1038/s41392-022-00955-7
Madhu LN, Kodali M, Attaluri S, Shuai B, Melissari L, Rao X, Shetty AK (2021) Melatonin improves brain function in a model of chronic Gulf War illness with modulation of oxidative stress, NLRP3 inflammasomes, and BDNF-ERK-CREB pathway in the hippocampus. Redox Biol 43:101973. https://doi.org/10.1016/j.redox.2021.101973
Sharma BR, Kanneganti TD (2021) NLRP3 inflammasome in cancer and metabolic diseases. Nat Immunol 22(5):550–559. https://doi.org/10.1038/s41590-021-00886-5
Chen H, Deng Y, Gan X, Li Y, Huang W, Lu L, Wei L, Su L et al (2020) NLRP12 collaborates with NLRP3 and NLRC4 to promote pyroptosis inducing ganglion cell death of acute glaucoma. Mol Neurodegener 15(1):26. https://doi.org/10.1186/s13024-020-00372-w
Liu M, Li H, Yang R, Ji D, Xia X (2022) GSK872 and necrostatin-1 protect retinal ganglion cells against necroptosis through inhibition of RIP1/RIP3/MLKL pathway in glutamate-induced retinal excitotoxic model of glaucoma. J Neuroinflammation 19(1):262. https://doi.org/10.1186/s12974-022-02626-4
Liu J, Zhang N, Zhang M, Yin H, Zhang X, Wang X, Wang X, Zhao Y (2021) N-acetylserotonin alleviated the expression of interleukin-1β in retinal ischemia-reperfusion rats via the TLR4/NF-κB/NLRP3 pathway. Exp Eye Res 208:108595. https://doi.org/10.1016/j.exer.2021.108595
Zhao W, Huang X, Han X, Hu D, Hu X, Li Y, Huang P, Yao W (2018) Resveratrol suppresses gut-derived NLRP3 inflammasome partly through stabilizing mast cells in a rat model. Mediators Inflamm 2018:6158671. https://doi.org/10.1155/2018/6158671
He Q, Li Z, Wang Y, Hou Y, Li L, Zhao J (2017) Resveratrol alleviates cerebral ischemia/reperfusion injury in rats by inhibiting NLRP3 inflammasome activation through Sirt1-dependent autophagy induction. Int Immunopharmacol 50:208–215. https://doi.org/10.1016/j.intimp.2017.06.029
Li H, Zheng F, Zhang Y, Sun J, Gao F, Shi G (2022) Resveratrol, novel application by preconditioning to attenuate myocardial ischemia/reperfusion injury in mice through regulate AMPK pathway and autophagy level. J Cell Mol Med 26(15):4216–4229. https://doi.org/10.1111/jcmm.17431
Rojo DLVM, Chapman E, Zhang DD (2018) NRF2 and the hallmarks of cancer. Cancer Cell 34(1):21–43. https://doi.org/10.1016/j.ccell.2018.03.022
Qin X, Li N, Zhang M, Lin S, Zhu J, Xiao D, Cui W, Zhang T et al (2019) Tetrahedral framework nucleic acids prevent retina ischemia-reperfusion injury from oxidative stress via activating the Akt/Nrf2 pathway. Nanoscale 11(43):20667–20675. https://doi.org/10.1039/c9nr07171g
Arioz BI, Tastan B, Tarakcioglu E, Tufekci KU, Olcum M, Ersoy N, Bagriyanik A, Genc K et al (2019) Melatonin attenuates LPS-induced acute depressive-like behaviors and microglial NLRP3 inflammasome activation through the SIRT1/Nrf2 pathway. Front Immunol 10:1511. https://doi.org/10.3389/fimmu.2019.01511
Yang H, Lv H, Li H, Ci X, Peng L (2019) Oridonin protects LPS-induced acute lung injury by modulating Nrf2-mediated oxidative stress and Nrf2-independent NLRP3 and NF-κB pathways. Cell Commun Signal 17(1):62. https://doi.org/10.1186/s12964-019-0366-y
Lin Y, Luo T, Weng A, Huang X, Yao Y, Fu Z, Li Y, Liu A et al (2020) Gallic acid alleviates gouty arthritis by inhibiting NLRP3 inflammasome activation and pyroptosis through enhancing Nrf2 signaling. Front Immunol 11:580593. https://doi.org/10.3389/fimmu.2020.580593
Satoh T, Trudler D, Oh CK, Lipton SA (2022) Potential therapeutic use of the rosemary diterpene carnosic acid for Alzheimer’s disease, Parkinson’s disease, and Long-COVID through NRF2 activation to counteract the NLRP3 inflammasome. Antioxid (Basel) 11(1). https://doi.org/10.3390/antiox11010124
Dai Y, Zhang J, Xiang J, Li Y, Wu D, Xu J (2019) Calcitriol inhibits ROS-NLRP3-IL-1β signaling axis via activation of Nrf2-antioxidant signaling in hyperosmotic stress stimulated human corneal epithelial cells. Redox Biol 21:101093. https://doi.org/10.1016/j.redox.2018.101093
Liu Q, Zhang F, Zhang X, Cheng R, Ma JX, Yi J, Li J (2018) Fenofibrate ameliorates diabetic retinopathy by modulating Nrf2 signaling and NLRP3 inflammasome activation. Mol Cell Biochem 445(1–2):105–115. https://doi.org/10.1007/s11010-017-3256-x
Acknowledgements
We would like to thank Editage (www.editage.cn) for English language editing.
Funding
This work was granted by the National Natural Science Foundation of China (No. 81271025 and 81860170).
Author information
Authors and Affiliations
Contributions
Design: Kaibao Ji and Yiqiao Xing. Research conduction: Jiazhen Feng, Kaibao Ji, Yiji Pan and Pingping Huang. Data collection and analysis: Kaibao Ji, Jiazhen Feng and Tao He. Figure preparation: Jiazhen Feng and Kaibao Ji. Study supervision: Tao He and Yiqiao Xing.
Corresponding authors
Ethics declarations
Ethical Approval
This study was performed in line with the principles of the National Institutes of Health Guide for the Care and Use of Laboratory Animals. Approval was granted by the Ethics Committee of the Renmin Hospital of Wuhan University.
Consent to Participate
Not applicable.
Consent to Publish
All authors have given their consent to publish this article.
Competing Interests
All authors declare that there has no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Feng, J., Ji, K., Pan, Y. et al. Resveratrol Ameliorates Retinal Ischemia-Reperfusion Injury by Modulating the NLRP3 Inflammasome and Keap1/Nrf2/HO-1 Signaling Pathway. Mol Neurobiol (2024). https://doi.org/10.1007/s12035-024-04105-8
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s12035-024-04105-8