Abstract
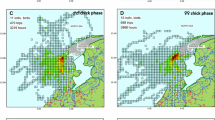
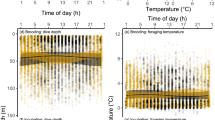
Sexual segregation in the behaviour, morphology or physiology of breeding seabirds can be related to divergent parental roles, foraging niche partitioning or sex-specific nutritional requirements. Here, we combine GPS tracking, dietary and nutritional analysis to investigate sex-specific foraging of Brown Boobies breeding on Raine Island, Great Barrier Reef, Australia. We observed sex-specific segregation in: (1) foraging location: females undertook longer trips, foraging at more distant locations than males; (2) foraging time: male activity and foraging occurred throughout the day, while female activity and foraging increased from midday to an afternoon peak; and (3) prey type, females mostly consumed flying fish, whereas males consumed equal proportions of flying fish and squid. Brown Booby diets contained five tropical prey species that significantly differed in their nutritional composition (Protein, Lipid and Water, wet mass). Despite this variation we found no differences in the overall nutritional content of prey caught by each sex. The observed sex-specific differences in prey type, location and time of capture are likely driven by a combination of a division of labour, risk partitioning and competition. However, Brown Boobies breeding on Raine Island, and other populations, might flexibly partition foraging niches by sex in response to varying competitive and environmental pressures. In light of such potential foraging dynamism, our inconclusive exploration of nutritional segregation between sexes warrants further investigation in the species.
Zusammenfassung
Sexuelle Segregation bei tropischen Seevögeln: Einflussfaktoren geschlechtsspezifischer Nahrungssuche bei Weißbauchtölpeln Sula leucogaster Sexuelle Segregation in Verhalten, Morphologie oder Physiologie brütender Seevögel kann mit unterschiedlichen Elternrollen, Nahrungsnischendifferenzierung oder geschlechtsspezifischem Nährstoffbedarf zusammenhängen. Hier kombinieren wir GPS-Ortung mit Nahrungs- und Nährstoffanalysen, um die geschlechtsspezifische Nahrungssuche bei auf Raine Island im australischen Great Barrier Reef brütenden Weißbauchtölpeln zu untersuchen. Wir haben geschlechtsspezifische Segregation gefunden in Bezug auf (a) den Ort der Nahrungssuche: Weibchen unternahmen längere Suchflüge als Männchen, da sie an weiter entfernten Orten nach Nahrung suchten; (b) den Zeitpunkt der Nahrungssuche: Männchen waren den ganzen Tag über aktiv und suchten nach Nahrung, während bei Weibchen die Aktivität und Nahrungssuche von mittags an zunahmen und am Nachmittag ihren Höhepunkt erreichten; (c) Beutetyp: Weibchen fraßen hauptsächlich fliegende Fische, während Männchen zu gleichen Teilen fliegende Fische und Kalmare verzehrten. Die Weißbauchtölpel nutzten fünf tropische Beutearten, die sich signifikant in ihrer Nährstoffzusammensetzung (Proteine, Lipide und Wasser in der Feuchtmasse) unterschieden. Trotz dieser Variation fanden wir keine Unterschiede im Gesamtnährstoffgehalt der von männlichen und weiblichen Tölpeln gefangenen Beute. Die beobachteten geschlechtsspezifischen Unterschiede im Beutetyp sowie Ort und Zeitpunkt des Beutefangs kommen wahrscheinlich durch eine Kombination von Arbeitsteilung, Risikoaufteilung und Konkurrenz zustande. Bei den auf Raine Island sowie in anderen Populationen brütenden Weißbauchtölpeln könnten die Geschlechter die Nahrungsnische flexibel unter sich aufteilen, abhängig von wechselnden Konkurrenz- und Umweltdrücken. Angesichts von solchem potenziellen Nahrungsdynamismus rechtfertigt unsere nicht beweiskräftige Erforschung der geschlechtsspezifischen Nahrungssegregation weitere Untersuchungen an Weißbauchtölpeln.



Similar content being viewed by others
References
Allen G (2009) Field guide to marine fishes of tropical Australia. Western Australian Museum, Perth
AOAC (2002) Official methods of analysis of AOAC International, 17th edn. Association of Official Analytical Chemists, Washington DC
AOAC (2005) Official methods of analysis of AOAC International, 18th edn. Association of Official Analytical Chemists, Arlington
Ashmole NP, Ashmole MJ (1967) Comparative feeding ecology of sea birds of a Tropical Oceanic Island. Peabody Mus Nat Hist Bull 24:1–131
Bates D, Maechler M, Bolker BM, Walker S (2015) Fitting linear mixed-effects models using lme4. J Stat Softw 67:1–48
Batianoff GN, Cornelius NJ (2005) Birds of Raine Island: population trends, breeding behaviour and nesting habitats. Proc R Soc Qld 112:1–29
Blaber SJM, Milton D, Smith GC, Farmer MJ (1995) Trawl discards in the diets of tropical seabirds of the Northern Great Barrier Reef, Australia. Mar Ecol Prog Ser 127:1–13
Blaber SJM, Milton D, Farmer MJ, Smith GC (1998) Seabird breeding populations on the far Northern Great Barrier Reef, Australia: trends and influences. Emu 98:44–57
Boyd C, Punt AE, Weimerskirch H, Bertrand S (2014) Movement models provide insights into variation in the foraging effort of central place foragers. Ecol Model 286:13–25
Calenge C (2006) The package ‘adehabitat’ for the R software: a tool for the analysis of space and habitat use by animals. Ecol Model 197:516–519
Castillo-Guerrero JA, Lerma M, Mellink E, Suazo-Guillén E, Peñaloza-Padilla EA (2016) Environmentally-mediated flexible foraging strategies in Brown Boobies in the Gulf of California. Ardea 104:33–47
Cleasby IR, Wakefield ED, Bodey TW, Davies RD, Patrick SC, Newton J, Votier SC, Bearhop S, Hamer KC (2015) Sexual segregation in a wide-ranging marine predator is a consequence of habitat selection. Mar Ecol Prog Ser 518:1–12
Clua É, Grosvalet F (2001) Mixed-species feeding aggregation of dolphins, large tunas and seabirds in the Azores. Aquat Living Resour 14:11–18. https://doi.org/10.1016/S0990-7440(00)01097-4
Congdon BC, Preker M (2004) Sex-specific chick provisioning and kleptoparasitism in the Least Frigatebird, Fregata ariel. Emu. 104:347–351. https://doi.org/10.1071/MU03008
Cortés E (1997) A critical review of methods of studying fish feeding based on analysis of stomach contents: application to elasmobranch fishes. Can J Fish Aquat Sci 54:726–738
Cruz SM, Hooten M, Huyvaert KP, Proaño CB, Anderson DJ, Afanasyev V, Wikelski M (2013) At-sea behavior varies with Lunar Phase in a Nocturnal Pelagic Seabird, the Swallow-tailed gull. PLoS One 8:1–8
Denuncio P, Viola M, Machovsky-Capuska GE, Raubenheimer D, Blasina G, Machado R, Polizzi P, Gerpe M, Cappozzo HL, Rodriguez DH (2017) Population variance in prey, diets and their macronutrient composition in an endangered marine predator, the Franciscana dolphin. J Sea Res. https://doi.org/10.1016/j.seares.2017.05.008
Duffy DC, Jackson S (1986) Diet studies of seabirds: a review of methods. Colon Waterbirds 9:1–17
Elliott KH, Gaston AJ, Crump D (2010) Sex-specific behavior by a monomorphic seabird represents risk partitioning. Behav Ecol 21:1024–1032
Gilardi JD (1992) Sex-specific foraging distributions of Brown Boobies in the Eastern Tropical Pacific. Colon Waterbirds 15:148–151
González-Solís J, Croxall JP, Wood AG (2000) Sexual dimorphism and sexual segregation in foraging strategies of Northern giant petrels, Macronectes halli, during incubation. Oikos 90:390–398. https://doi.org/10.1034/j1600-07062000900220x
Guerra M, Drummond H (1995) Reversed size dimorphism and parental care: minimal division of labour in the blue-footed Booby. Behaviour 132:479–797
Ismar SMH, Raubenheimer D, Bury SJ, Millar CD, Hauber ME (2017) Sex-specific foraging during parental care in a size-monomorphic seabird, the Australasian Gannet (Morus serrator). Wilson J Ornithol 129:139–147. https://doi.org/10.1676/1559-4491-1291139
Lascelles B, Taylor P, Miller MGR, Dias MP, Oppel S, Torres L, Hedd A, Le Corre M, Phillips RA, Schaffer S, Weimerskirch H, Small C (2016) Applying global criteria to tracking data to define important areas for marine conservation. Divers Distrib 22:422–431
Lewis S, Sherratt TN, Hamer KC, Wanless S (2001) Evidence of intra-specific competition for food in a pelagic seabird. Nature 412:816–819
Lewis S, Benvenuti S, DallAntonia L, Griffiths R, Money L, Sherratt TN, Wanless S, Hamer KC (2002) Sex-specific foraging behaviour in a monomorphic seabird. Proc R Soc Lond B 269:1687–1693. https://doi.org/10.1098/rspb20022083
Lewis S, Schreiber E, Daunt F, Schenk G, Orr K, Adams A, Wanless S, Hamer KC (2005) Sex-specific foraging behaviour in tropical Boobies: does size matter? Ibis 147:408–414. https://doi.org/10.1111/j1474-919x200500428x
Lormee H, Barbraud C, Chastel O (2005) Reversed sexual size dimorphism and parental care in the Red-Footed Booby Sula sula. Ibis 147:307–315. https://doi.org/10.1111/j1474-919x200500404x
Machovsky-Capuska GE, Priddel D, Leong PH, Jones P, Carlile N, Shannon L, Portelli D, McEwan A, Chaves AV, Raubenheimer D (2016a) Coupling bio-logging with nutritional geometry to reveal novel insights into the foraging behaviour of a plunge-diving marine predator. NZ J Mar Freshwat Res 8330:1–15. https://doi.org/10.1080/0028833020161152981
Machovsky-Capuska GE, Senior AM, Benn EC, Tait AH, Schuckard R, Stockin KA, Cook W, Ogle M, Barna K, Melville D, Wright B, Purvin C, Raubenheimer D (2016b) Sex-specific macronutrient foraging strategies in a highly successful marine predator: the Australasian Gannet. Mar Biol 163:1–14. https://doi.org/10.1007/s00227-016-2841-y
Machovsky-Capuska GE, Senior AM, Simpson SJ, Raubenheimer D (2016c) The multi-dimensional nutritional niche. Trends Ecol Evol 31:355–365. https://doi.org/10.1016/jtree201602009
Michelot T, Langrock R, Patterson TA (2016) moveHMM: an R package for the statistical modelling of animal movement data using hidden Markov models. Methods Ecol Evol 7:1308–1315
Min D, Steensen D (1998) Crude fat analysis. Food Anal 2:201–216
Moltschaniwskyj N, Doherty P (1995) Cross-shelf distribution patterns of tropical juvenile cephalopods sampled with light-traps. Mar Freshw Res 46:707–714
Morris-Pocock JA, Anderson DJ, Friesen VL (2011) Mechanisms of global diversification in the brown booby (Sula leucogaster) revealed by uniting statistical phylogeographic and multilocus phylogenetic methods. Mol Ecol 20:2835–2850
Morse DH (1968) A quantitative study of foraging of male and female spruce-woods warblers. Ecol Soc Am 49:779–784
Nelson B (1978) The Sulidae: gannets and boobies. Oxford University Press, Oxford
Newton I (1979) Population ecology of raptors. T. & A.D. Poyser, Berkhamsted
NRC (1989) Recommended dietary allowances, 10th edn. National Academy Press, Washington DC
Nunes GT, Mancini PL, Bugoni L (2016) When Bergmann’s rule fails: evidences of environmental selection pressures shaping phenotypic diversification in a widespread seabird. Ecography 40:365–375
Oppel S, Beard A, Fox D, Mackley E, Leat E, Henry L, Clingham E, Fowler N, Sim J, Sommerfeld J, Weber N, Weber S, Bolton M (2015) Foraging distribution of a tropical seabird supports Ashmoles hypothesis of population regulation. Behav Ecol Sociobiol 69:915–926
Paiva VH, Pereira J, Ceia FR, Ramos JA (2017) Environmentally driven sexual segregation in a marine top predator. Sci Rep 7:2590
Peck DR, Congdon BC (2006) Sex-specific chick provisioning and diving behaviour in the wedge-tailed shearwater Puffinus pacificus. J Avian Biol 37:245–251
Petit LJ, Petit DR, Petit KE, Fleming W (1990) Intersexual and temporal variation in foraging ecology of Prothonotary warblers during the breeding season. Auk 107:133–145
Phillips RA, Silk JRD, Phalan B, Catry P, Croxall JP (2004) Seasonal sexual segregation in two Thalassarche albatross species: competitive exclusion, reproductive role specialization or foraging niche divergence? Proc R Soc Lond B 271:1283–1291. https://doi.org/10.1098/rspb20042718
Pontón-Cevallos J, Dwyer RG, Franklin CE, Bunce A (2017) Understanding resource partitioning in sympatric seabirds living in tropical marine environments. Emu Austral Ornithology. https://doi.org/10.1080/0158419720161265431
R Core Team (2017) R: a language and environment for statistical computing. R Foundation for Statistical Computing, Vienna
Randall J, Allen G, Steene R (1997) Fishes of the Great Barrier Reef and Coral Sea. University of Hawaii Press, Hawaii
Raubenheimer D (2011) Toward a quantitative nutritional ecology: the right-angled mixture triangle. Ecol Monogr 81:407–427
Raubenheimer D, Machovsky-Capuska GE, Chapman CA, Rothman JM (2015) Geometry of nutrition in field studies: an illustration using wild primates. Oecologia 177:223–234
Schuckard R, Melville D, Cook W, Machovsky-Capuska G (2012) Diet of the Australasian Gannet (Morus serrator) at Farewell Spit, New Zealand. Notornis 59:66–70
Selander RK (1966) Sexual dimorphism and differential niche utilization in birds. Condor 68:113–151
Shaffer SA, Weimerskirch H, Costa DP (2001) Functional significance of sexual dimorphism in wandering albatrosses, Diomedea exulans. Funct Ecol 15:203–210
Soanes LM, Bright JA, Bolton M, Millett J, Mukhida F, Green JA (2015) Foraging behaviour of Brown Boobies Sula leucogaster in Anguilla, Lesser Antilles: preliminary identification of at-sea distribution using a time-in-area approach. Bird Conserv Int 25:87–96. https://doi.org/10.1017/S095927091400001X
Sommerfeld J, Kato A, Ropert-Coudert Y, Garthe S, Hindell MA (2013) The individual counts: within sex differences in foraging strategies are as important as sex-specific differences in Masked Boobies Sula dactylatra. J Avian Biol 44:531–540. https://doi.org/10.1111/j1600-048X201300135x
Spear LB, Ainley DG, Walker WA (2007) Foraging dynamics of seabirds in the eastern tropical Pacific Ocean. Stud Avian Biol 35:1–99
Stahl JC, Sagar PM (2000) Foraging strategies and migration of southern Bullers albatrosses Diomedea b. bulleri breeding on the Solander Is, New Zealand. J R Soc N Z 30:319–334. https://doi.org/10.1080/0301422320009517625
Tait A, Raubenheimer D, Stockin K, Merriman M, Machovsky-Capuska GE (2014) Nutritional geometry and the macronutrient variation in the diet of Gannets: the challenges in marine field studies. Mar Biol 12:2791–2801
Weimerskirch H, Le Corre M, Jaquemet S, Marsac F (2005) Foraging strategy of a tropical seabird, the Red-footed Booby, in a dynamic marine environment. Mar Ecol Prog Ser 288:251–261. https://doi.org/10.3354/meps288251
Weimerskirch H, Le Corre M, Ropert-Coudert Y, Kato A, Marsac F (2006) Sex-specific foraging behaviour in a seabird with reversed sexual dimorphism: the Red-footed Booby. Oecologia 146:681–691. https://doi.org/10.1007/s00442-005-0226-x
Weimerskirch H, Le Corre M, Gadenne H, Pinaud D, Kato A, Ropert-Coudert Y, Bost CA (2009a) Relationship between reversed sexual dimorphism, breeding investment and foraging ecology in a pelagic seabird, the Masked Booby. Oecologia 161:637–649. https://doi.org/10.1007/s00442-009-1397-7
Weimerskirch H, Shaffer S, Tremblay Y, Costa DP, Gadenne H, Kato A, Ropert-Coudert Y, Sato K, Aurioles D (2009b) Species- and sex-specific differences in foraging behaviour and foraging zones in Blue-footed and Brown Boobies in the Gulf of California. Mar Ecol Prog Ser 391:267–278. https://doi.org/10.3354/meps07981
Young HS, McCauley DJ, Dirzo R, Dunbar RB, Shaffer S (2010) Niche partitioning among and within sympatric tropical seabirds revealed by stable isotope analysis. Mar Ecol Prog Ser 416:285–294. https://doi.org/10.3354/meps08756
Zavalaga CB, Benvenuti S, Dall’Antonia L, Emslie SD (2007) Diving behavior of Blue-footed Boobies Sula nebouxii in Northern Peru in relation to sex, body size and prey type. Mar Ecol Prog Ser 336:291–303. https://doi.org/10.3354/meps336291
Acknowledgements
We would like to thank Damon Shearer, Andrew Dunstan and crew of the QPWS Reef Ranger for transport, accommodation and logistical support on and off of Raine Island. We also thank two anonymous reviewers, whose comments significantly improved the manuscript. This research was funded by the Australian Research Council (ARC) LP 0562157, the Marine and Tropical Sciences Research Facility (MTSRF), the Great Barrier Reef Marine Park Authority (GBRMPA) and National Environmental Research Program (NERP). Fieldwork procedures were authorised under James Cook University Ethics Approval A1992. GEMC is supported by the Loxton research fellowship from The University of Sydney.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by C. Barbraud.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Miller, M.G.R., Silva, F.R.O., Machovsky-Capuska, G.E. et al. Sexual segregation in tropical seabirds: drivers of sex-specific foraging in the Brown Booby Sula leucogaster . J Ornithol 159, 425–437 (2018). https://doi.org/10.1007/s10336-017-1512-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10336-017-1512-1